Clear Sky Science · en
Local cation-clamping distorts and softens RNA duplex
Why this tiny twist in RNA matters
Inside every cell, DNA and RNA are constantly bent, stretched, and twisted as they store and use genetic information. These motions are not just mechanical details—they control how genes are read, how viruses copy themselves, and how new RNA-based medicines and nanodevices will work. This study shows that simple salts made of positively and negatively charged ions can dramatically change how stiff or flexible RNA is, and that RNA responds very differently from DNA. Understanding this subtle “softening and stiffening” dance could help scientists better design RNA drugs, vaccines, and molecular tools.
Salty surroundings and genetic molecules
DNA and RNA carry a strong negative electrical charge, so they are surrounded by clouds of oppositely charged ions in water. Most earlier work focused on positively charged ions, especially multivalent ones that carry two or three positive charges, such as magnesium or spermidine. These ions are known to hold DNA strands together, condense them, or change how easily they bend. RNA, though chemically similar to DNA, behaves differently: it is usually harder to bend but easier to stretch. The new work asks a deeper question: what happens when both the positive ions and their accompanying negative partners—called anions—interact with DNA and RNA at very high salt levels, much higher than in typical cells?
Pulling on single molecules to measure stiffness
The researchers used a technique called magnetic tweezers to stretch individual DNA or RNA double helices one at a time. Each molecule was anchored to a glass surface at one end and to a tiny magnetic bead at the other. By moving magnets above the sample, they could pull with controlled forces and record how far each molecule extended, and how it twisted under tension. From these force–extension curves, they extracted four key mechanical traits: how hard the molecule is to bend, how hard it is to stretch, its effective length per base pair, and how twisting changes its length. They repeated these measurements across a wide range of multivalent salt concentrations for several different salts, including spermidine and calcium chloride.
DNA stiffens again, but RNA suddenly softens
For DNA, the behavior largely matched expectations. As the concentration of multivalent positive ions increased, DNA first became easier to bend—its stiffness dropped—because the ions neutralized its negative charge. At even higher levels, the excess positive ions overcompensated, effectively reversing the net charge on DNA. This “charge inversion” made DNA harder to bend again, so its stiffness rose. Surprisingly, RNA showed the opposite and more dramatic pattern. At low to moderate salt levels, its bending stiffness increased: the RNA duplex became straighter and more rigid. But at very high concentrations, the bending stiffness of RNA dropped by more than half, and other properties such as its stretchiness and twist–stretch behavior flipped in unexpected ways.
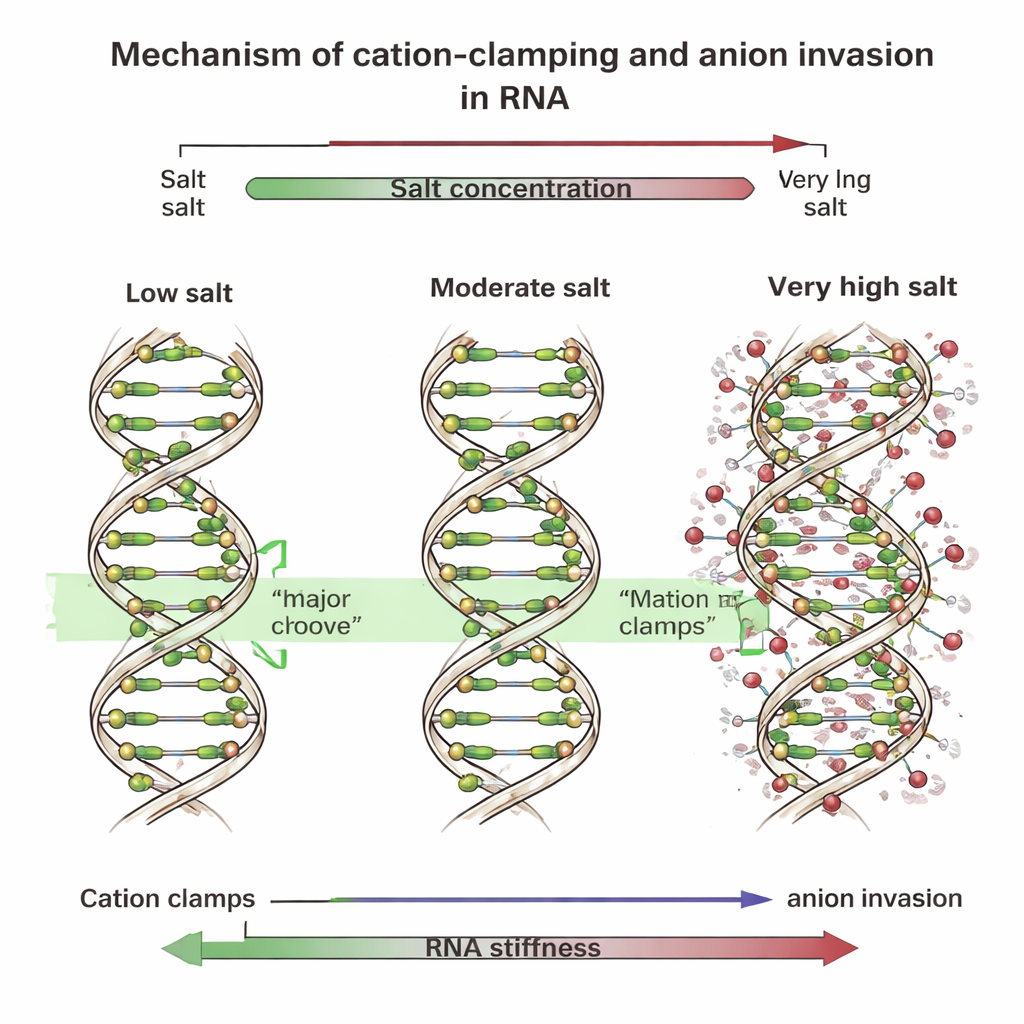
Clamps, grooves, and invading ions
To uncover the cause, the team ran detailed computer simulations that tracked every atom of short DNA and RNA fragments in salty water. These simulations revealed that at low and moderate concentrations, multivalent positive ions settle into a wide surface on RNA called the major groove. There they form “cation clamps” that bridge across the groove, pulling its sides together, straightening the helix, and making it stiffer. DNA, whose grooves are shaped differently, mainly binds these ions along its outer backbone instead. At very high salt levels, however, many negative ions—such as chloride—push in close to the RNA backbone and even into the major groove. Their presence disrupts the orderly clamps, breaking them into irregular, patchy “local clamps” that distort the helix. When the researchers mimicked this effect by adding artificial springs or locking extra negative ions near the RNA in simulations, the RNA backbone bent more and its overall stiffness plummeted, just as seen in experiments.
What this means for future RNA technologies
In plain terms, the study shows that RNA can be made either straighter and stiffer or more kinked and flexible simply by changing the type and concentration of surrounding salts. At modest levels of multivalent ions, positive charges neatly clamp the RNA’s outer surface and brace it; at extreme levels, invading negative ions break that uniform bracing and create warped, softer regions. DNA does not show this sharp softening because it binds ions in a different way and undergoes net charge reversal instead. These insights highlight that not only positive ions but also their negative partners are crucial for controlling RNA shape. That knowledge will help researchers deliberately tune RNA mechanics in the lab—for example, to stabilize RNA drugs, control how RNA folds in biosensors, or design more reliable RNA-based nanostructures.
Citation: Zhang, C., Dong, HL., Zhang, JH. et al. Local cation-clamping distorts and softens RNA duplex. Commun Biol 9, 308 (2026). https://doi.org/10.1038/s42003-026-09580-4
Keywords: RNA mechanics, multivalent ions, cation clamping, DNA versus RNA, salt effects