Clear Sky Science · en
A pathogenic mutation in α-SNAP impairs membrane lipid binding by concealing a critical hydrophobic loop
How a Tiny Protein Change Can Disrupt Brain Development
Our brain cells constantly ship cargo in tiny bubbles called vesicles, and a helper protein named alpha-SNAP is central to making those deliveries happen. A rare mutation in alpha-SNAP, known from a mouse line called “hyh,” causes severe problems in brain formation and fluid balance. This study asks a deceptively simple question with big consequences: does that single-letter change in alpha-SNAP stop it from latching onto cell membranes, and could that be a key reason the brain goes awry?
A Cellular Dock Worker with Many Jobs
Alpha-SNAP normally works as a kind of dock worker for vesicles, helping them fuse with cellular membranes so that cargo can be released or recycled. It cooperates with a set of proteins called SNAREs and an energy-using machine called NSF to pull membranes together and then reset the system for the next round. Beyond this classic role, alpha-SNAP also helps control processes like cell self-cleaning (autophagy), cell death, calcium entry, and energy sensing. All of these functions depend, in one way or another, on alpha-SNAP’s ability to touch and partially insert into the oily surface of membranes.
A Hidden Loop and a Troublesome Mutation
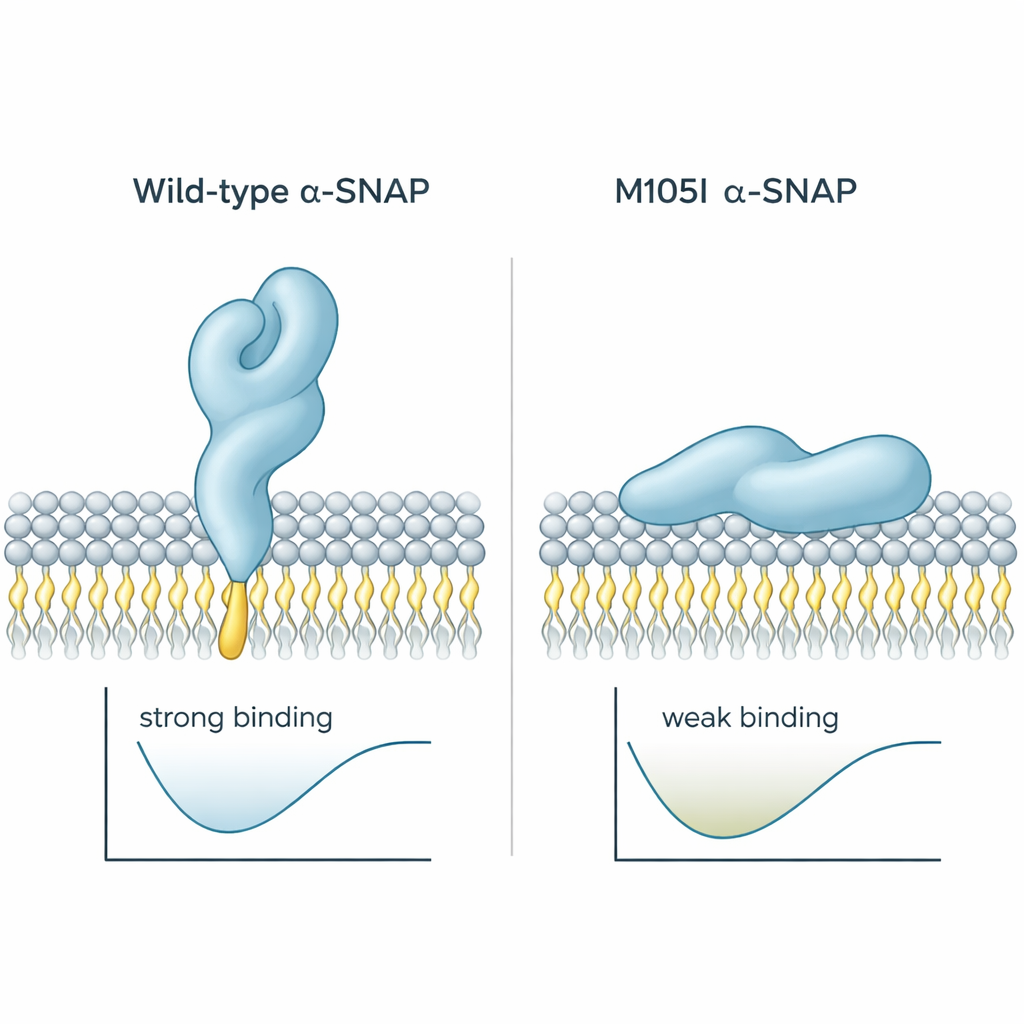
Earlier work had shown that alpha-SNAP uses a short greasy “loop” near one end of the protein to hook into membranes. In healthy protein, this loop sticks out and can dive into the outer layer of the membrane. In the hyh mouse, a single building block at position 105 is swapped, creating the M105I mutation. The authors used computer simulations to see how this change alters the protein’s shape. They found that the mutant protein becomes slightly more compact and twists in a way that tucks the hydrophobic loop inward, away from water and away from membranes. As a result, the mutant protein approaches membranes at a flatter angle and tends to press against them with a less sticky region, reducing both contact time and depth of insertion. Calculations of binding energy supported this: the wild-type protein settled into a low-energy, strongly bound state, while the mutant preferred weaker, shallower contacts.
Putting the Prediction to the Test in the Lab
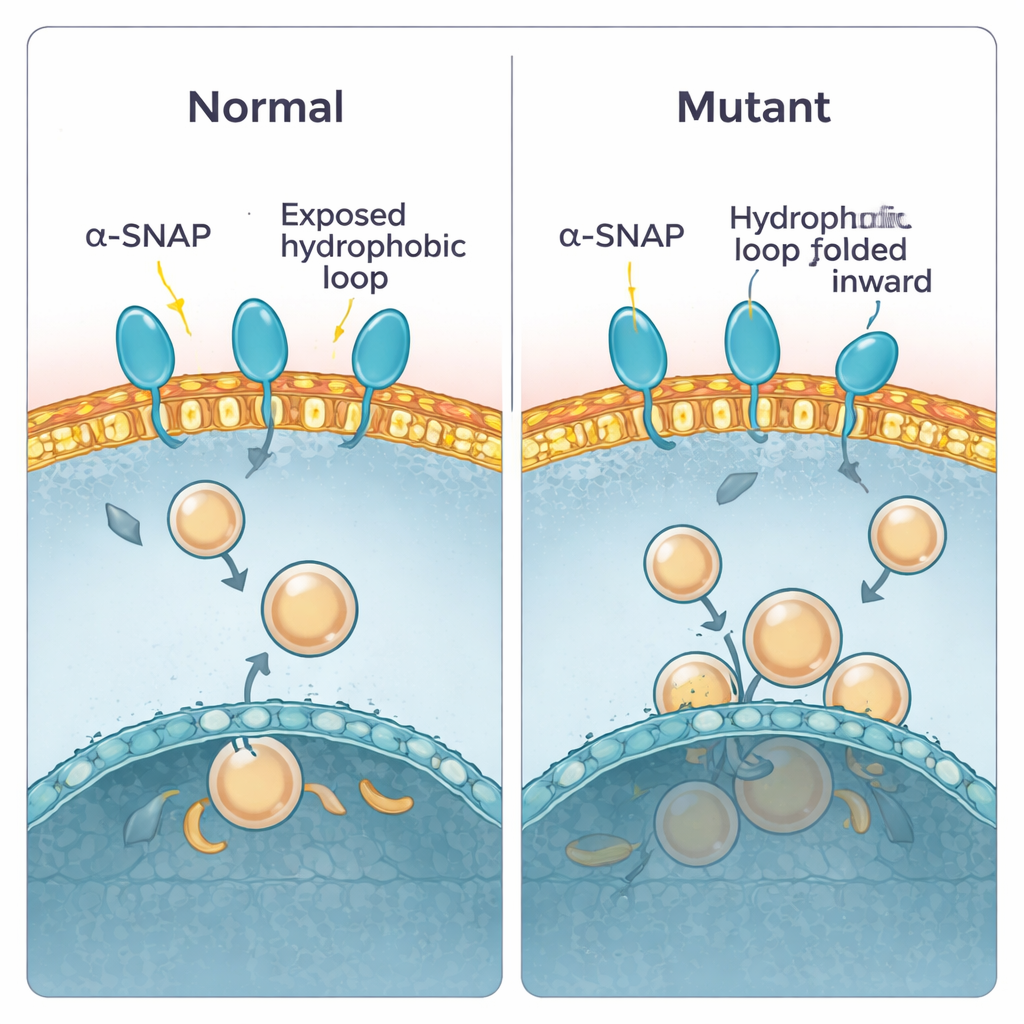
To check these predictions, the team purified both normal and mutant alpha-SNAP and tested how they behaved in different experimental setups. First, they used a detergent that separates into watery and oily phases, mimicking the choice between water and membrane. Normal alpha-SNAP partitioned evenly, consistent with a balanced, partly oily surface. The mutant version was less willing to enter the oily phase, suggesting that its greasy parts are indeed more buried. Next, they let the proteins interact with flat plasma-membrane “sheets” prepared from cells and imaged how many binding spots appeared. Again, normal alpha-SNAP dotted the membrane, while the mutant bound much less. Finally, they floated artificial membrane bubbles (liposomes) made from brain lipids through sugar gradients. Wild-type alpha-SNAP rode upward with vesicles rich in plasma-membrane lipids, whereas the mutant lagged behind, binding poorly unless the vesicles were made from a different, more internal membrane mix.
Consequences in the Developing Brain
The researchers then turned to brains of developing mouse embryos, focusing on a stage when new nerve cells are being born. By separating cell contents into soluble and membrane-bound pools, they showed that total alpha-SNAP levels were already reduced in hyh brains, but more importantly, the fraction attached to membranes was disproportionately low compared with normal mice. When they further separated different membrane types, they found that the biggest loss was at the plasma membrane, the outer surface of the cell, while internal membranes were much less affected. Microscopy told the same story: in normal tissue, alpha-SNAP outlined cells in a honeycomb pattern that matched a known surface marker. In hyh brains, this crisp border staining faded into a more diffuse, internal glow, indicating that the mutant protein fails to stay anchored at the cell edge where vesicle fusion and signaling are most active.
Why This Matters for Disease
Taken together, the work shows that the M105I mutation does more than simply lower the amount of alpha-SNAP; it changes how the protein folds so that its key membrane-grabbing loop is hidden. This makes it harder for alpha-SNAP to grip the plasma membrane and to adopt the proper orientation for organizing fusion machinery. In a developing brain, where precise delivery of signals and building materials at the cell surface is essential, this subtle structural flaw likely contributes to the hydrocephalus, miswired cortex, and other defects seen in hyh mice. For non-specialists, the message is clear: even a tiny change in the shape of a single protein can reshape how cells communicate and, in turn, how a brain is built.
Citation: Méndez-Ruette, M., Bedoya, M., Hinrichsen, B. et al. A pathogenic mutation in α-SNAP impairs membrane lipid binding by concealing a critical hydrophobic loop. Commun Biol 9, 294 (2026). https://doi.org/10.1038/s42003-026-09578-y
Keywords: alpha-SNAP, membrane binding, protein mutation, brain development, vesicle fusion