Clear Sky Science · en
The perforant pathway and CA3-Schaffer collateral afferents coordinate to regulate spatial learning
Why finding your way matters
Whether a mouse searching for a hidden platform or a person navigating a new city, the brain constantly turns sights, sounds and self-motion into an internal map. This study explores how two key brain regions, the entorhinal cortex and the hippocampus, work together to support this ability. By watching live activity in tiny nerve fibers and precisely stimulating or silencing specific pathways in mice, the researchers uncover how these areas coordinate to form and stabilize spatial memories—insights that may eventually help us understand and treat memory disorders.
Two key hubs of the brain’s inner GPS
The hippocampus and the nearby entorhinal cortex sit deep within the brain’s temporal lobe and form the core of our navigation system. Within the hippocampus, an internal circuit links an area called CA3 to another called CA1 through fibers known as Schaffer collaterals. At the same time, the entorhinal cortex sends a separate stream of information directly into CA1 via the so‑called perforant pathway. The authors set out to determine how these two input streams cooperate when an animal learns the layout of a space, and how changes at their connections—known broadly as “plasticity”—support the building of a stable spatial map.
Watching learning unfold fiber by fiber
To follow this process in behaving animals, the team used fiber photometry, a technique that reports nerve activity using flashes of fluorescent light. They engineered mice so that CA3 neurons projecting to CA1 glowed when active, then trained the animals on the Morris water maze, a classic test in which a mouse must learn the hidden location of a small platform in a pool. During early trials, the CA3–CA1 fibers were strongly active as the animals searched; as the mice became proficient and found the platform more quickly, this activity gradually declined. The pattern suggests that these connections are especially engaged while the brain is first encoding the layout of the environment, and then settle into a more efficient, stable representation once the memory is established.
How upstream input boosts or blunts learning
The scientists next asked how signals from the entorhinal cortex influence this hippocampal circuit. Using light‑sensitive proteins to activate entorhinal neurons while recording from CA3–CA1 fibers, they showed that driving input from the medial entorhinal cortex reliably increased activity in those hippocampal connections. Conversely, when they chemogenetically dampened entorhinal signals arriving in CA1 during water‑maze training, CA3–CA1 activity weakened and the mice learned the platform location more slowly and less precisely. In a complementary set of experiments, the authors recorded directly from entorhinal neurons that project to CA1 and observed that their activity ramped up across training days, mirroring the animals’ improving performance. Together, these results indicate that strong, well‑timed entorhinal signals are needed to properly tune hippocampal circuits for navigation.
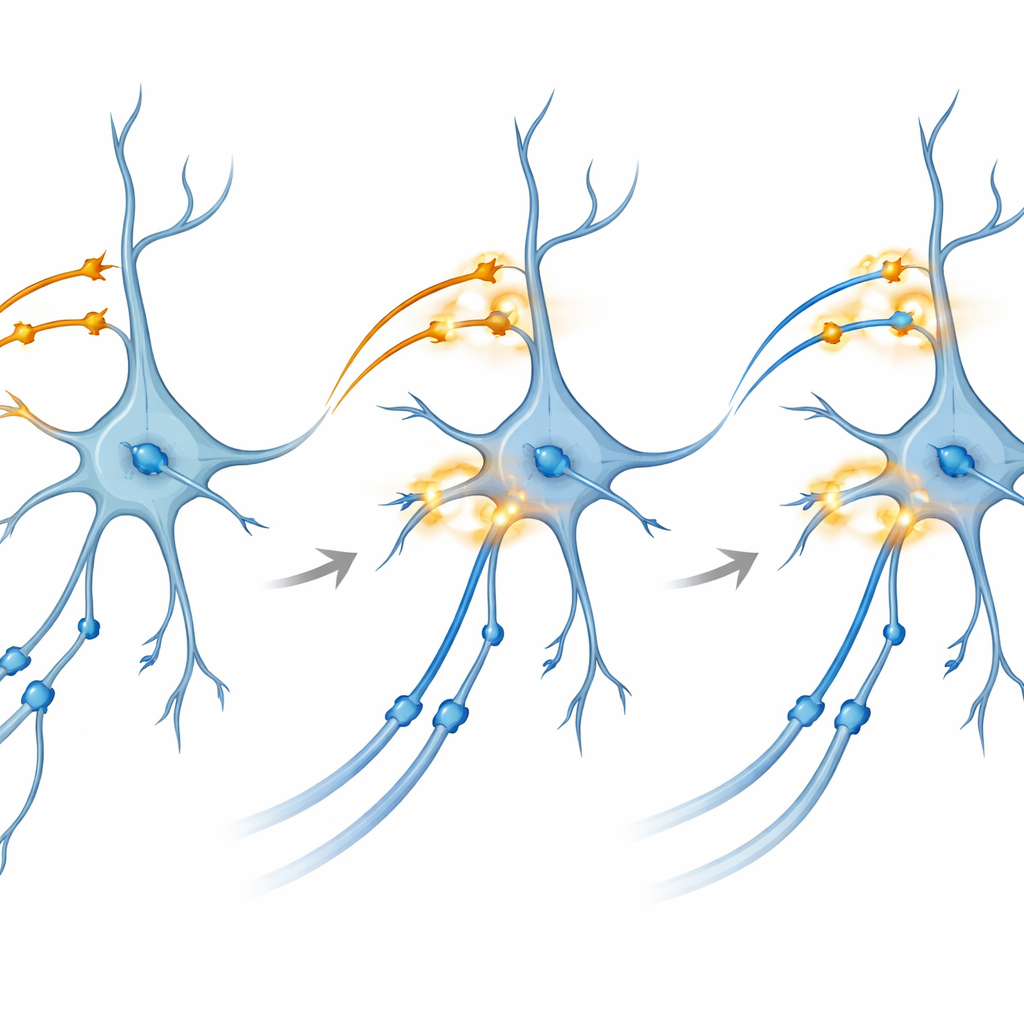
Zooming in on how connections strengthen
To probe the underlying mechanism, the team turned to brain slices kept alive in a dish. Here they could separately control light‑activated proteins in CA3 and in entorhinal inputs to CA1. Surprisingly, theta‑burst light stimulation—patterns that mimic natural rhythmic firing—applied to either pathway alone did not reliably produce long‑term potentiation, the lasting strengthening of synapses thought to underlie memory. However, when they delivered a carefully timed dual‑color burst that co‑activated both pathways onto CA1 at the same time, the CA3–CA1 connections showed robust and long‑lasting enhancement. Blocking NMDA receptors or specific calcium channels, two well‑known molecular gates for plasticity, prevented this effect, tying the phenomenon to classic biochemical routes of memory formation.
What this means for memory and disease
Overall, the study paints a picture of spatial learning as a coordinated dance between incoming entorhinal signals and internal hippocampal wiring. Entorhinal inputs not only carry information about position and context; they also act as a powerful control knob that can amplify or dampen activity in CA3–CA1 fibers and, when co‑activated, drive long‑term strengthening of those synapses. For a lay reader, the key takeaway is that the brain’s “GPS” does not rely on a single area, but on a partnership in which one region sets the scene and another locks in the details. Disruptions to this partnership—whether by aging, injury or neurodegenerative disease—could therefore help explain why people lose their way in familiar places, and targeting these pathways might offer new avenues for preserving or restoring spatial memory.
Citation: Huang, F., Temitayo Bello, S., Lau, S.H. et al. The perforant pathway and CA3-Schaffer collateral afferents coordinate to regulate spatial learning. Commun Biol 9, 364 (2026). https://doi.org/10.1038/s42003-026-09577-z
Keywords: spatial memory, hippocampus, entorhinal cortex, synaptic plasticity, navigation