Clear Sky Science · en
ZFP42 maintains stemness and rhythmic transcription in human epidermal stem and progenitor cells via CRY1
Why your skin’s internal clock matters
Every night while you sleep, your skin doesn’t rest—it repairs, renews, and prepares for the next day’s onslaught of sun and wear. This study explores how tiny biological clocks inside skin stem cells keep that renewal process on schedule, and how a lesser-known factor called ZFP42 teams up with a clock protein, CRY1, to keep those stem cells youthful and functional. Understanding this link between timekeeping and skin renewal could one day inform better treatments for aging skin, wound healing, and skin diseases.
The skin’s timekeepers
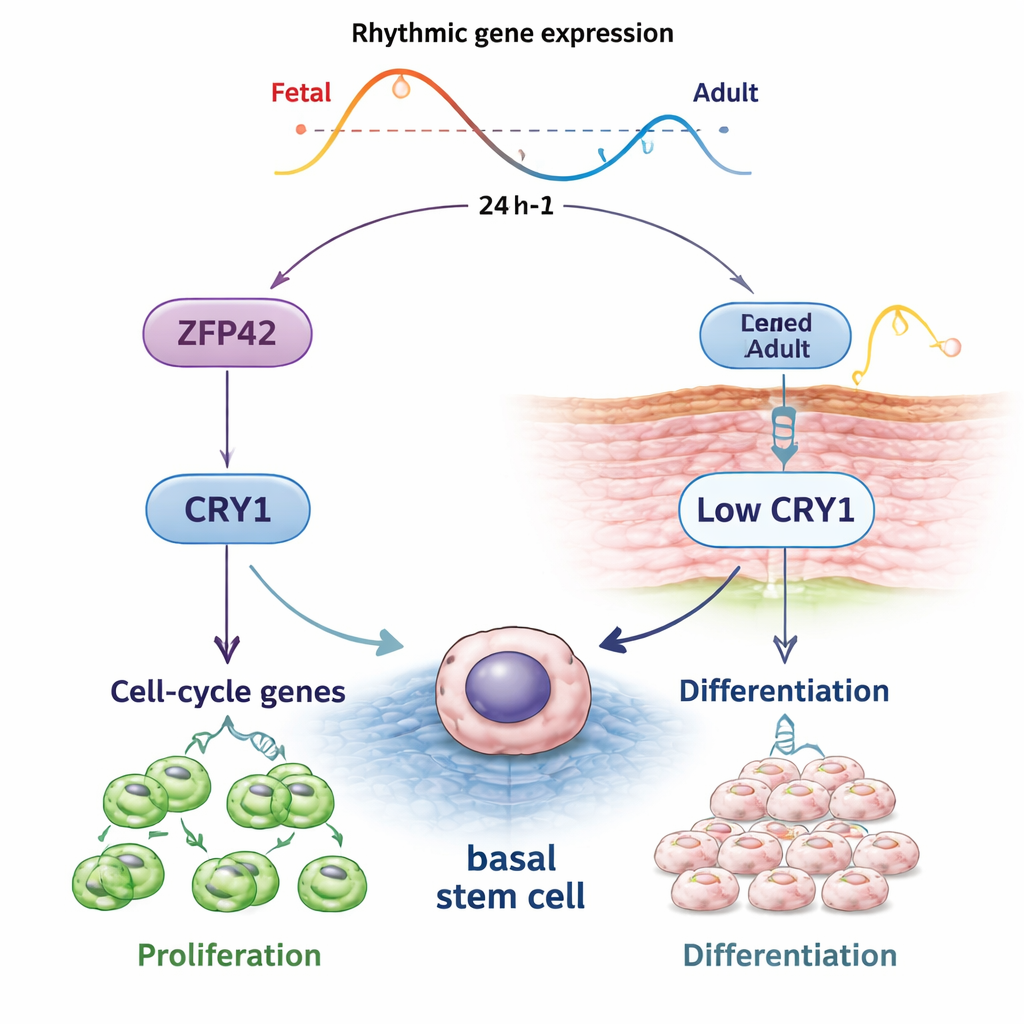
Like the brain’s master clock that responds to light, nearly every tissue in the body has its own “peripheral” clock. In the outer skin layer, the epidermis, stem and progenitor cells at the base constantly divide, push upward, and form the protective barrier we depend on. The authors studied these epidermal stem and progenitor cells (EPSCs) from both human fetuses and adults to see how many genes turn on and off with a roughly 24-hour rhythm. They found that about one in ten active genes in these cells shows daily oscillation, and that the timing patterns of many key genes change between fetal and adult skin, suggesting that our skin’s cellular clocks age along with us.
Fetal vs. adult skin: stronger rhythms in youth
By synchronizing EPSCs in lab dishes and measuring gene activity every three hours, the team discovered that core clock genes—such as BMAL1, PER1, and CRY1—cycle robustly in both fetal and adult cells. However, the overall “loudness” of rhythmic gene activity, known as amplitude, was higher in fetal cells and dampened in adult cells. Many shared rhythmic genes were tied to DNA replication, cell division, and repair, processes that are central to keeping stem cells healthy. The timing of gene peaks also shifted: adult cells showed delayed and altered rhythms, especially in genes related to viral defense, cell aging, and programmed cell death, hinting that the daily program of protection and renewal is re-tuned as skin matures and ages.
Daily timing of growth and maturation
When the researchers looked at genes that were rhythmic only in fetuses or only in adults, they saw striking differences. In fetal EPSCs, waves of genes involved in wound response, metabolism, and intense cell division tended to peak earlier in the daily cycle, fitting with their rapid growth and regenerative capacity. In adult EPSCs, genes linked to building the skin barrier and driving cells toward full differentiation often peaked earlier than genes controlling DNA replication and division. This separation suggests that adult skin stem cells may use time of day to alternate between preparing cells to harden into a protective surface and fueling cell proliferation later, perhaps to balance barrier maintenance with protection from daytime stress like ultraviolet radiation.
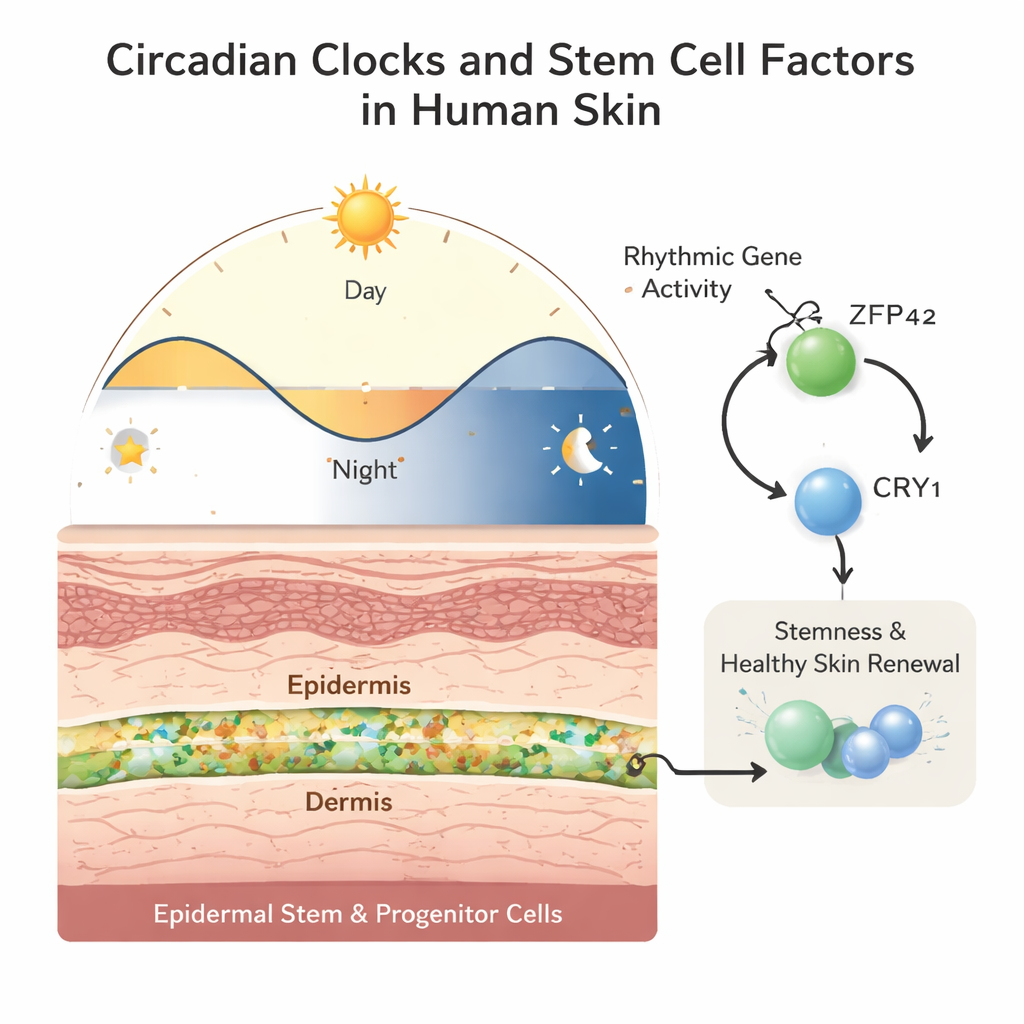
ZFP42 and CRY1: keeping stem cells youthful
Digging deeper, the team searched for regulatory molecules that might sit above these rhythmic genes. They identified ZFP42, better known as a marker of very early stem cells, as highly active in fetal EPSCs and strongly associated with promoters of fetal rhythmic genes. When ZFP42 levels were reduced in young EPSCs, the cells divided less, accumulated in an early cell-cycle phase, and showed more signs of differentiation and cell death. The reverse was also true: boosting ZFP42 in EPSCs from older adults improved their colony-forming ability, making them behave more like younger stem cells. Crucially, ZFP42 directly activated the clock gene CRY1 and was required for its normal rhythmic behavior. Lowering CRY1 alone mimicked many effects of ZFP42 loss—slower proliferation and increased differentiation—while restoring CRY1 partly rescued the growth defects, indicating that a ZFP42–CRY1 axis is central to maintaining stem-like behavior.
What this means for skin health
For non-specialists, the takeaway is that skin stem cells don’t just respond to damage and aging passively—they follow a daily schedule choreographed by molecular clocks, and this schedule changes over the course of life. ZFP42 acts as a youth-associated switch that keeps the clock gene CRY1 active and rhythmic, helping epidermal stem cells renew themselves while preventing premature maturation. As ZFP42-linked programs weaken with age, the clock-controlled renewal cycles in skin appear to fade. Although this work was done in cultured cells and needs confirmation in living tissue, it points to the possibility that tuning the ZFP42–CRY1 pathway or aligning treatments with the skin’s internal time may one day improve therapies for aging skin, chronic wounds, and other disorders of the body’s largest organ.
Citation: Gao, S., Tan, H., Xu, S. et al. ZFP42 maintains stemness and rhythmic transcription in human epidermal stem and progenitor cells via CRY1. Commun Biol 9, 291 (2026). https://doi.org/10.1038/s42003-026-09576-0
Keywords: circadian rhythm, skin stem cells, epidermis, CRY1, ZFP42