Clear Sky Science · en
Development of capsaicin-derived prohibitin ligands to modulate the Aurora kinase A/PHB2 interaction and mitophagy in cancer cells
Why a chili pepper chemical matters for cancer
Chili peppers owe their burn to capsaicin, a small molecule that latches onto proteins in our cells. This study explores a surprising twist on that familiar spice: researchers redesigned capsaicin to create new drug-like molecules that home in on cancer cell “power plants” — the mitochondria — and temper a process those cells use to keep their energy factories in top shape. Because cancer cells rely heavily on efficient mitochondria to fuel runaway growth, being able to selectively disrupt this support system could open a new path for anticancer therapies.
Cell power plants and their internal cleanup crew
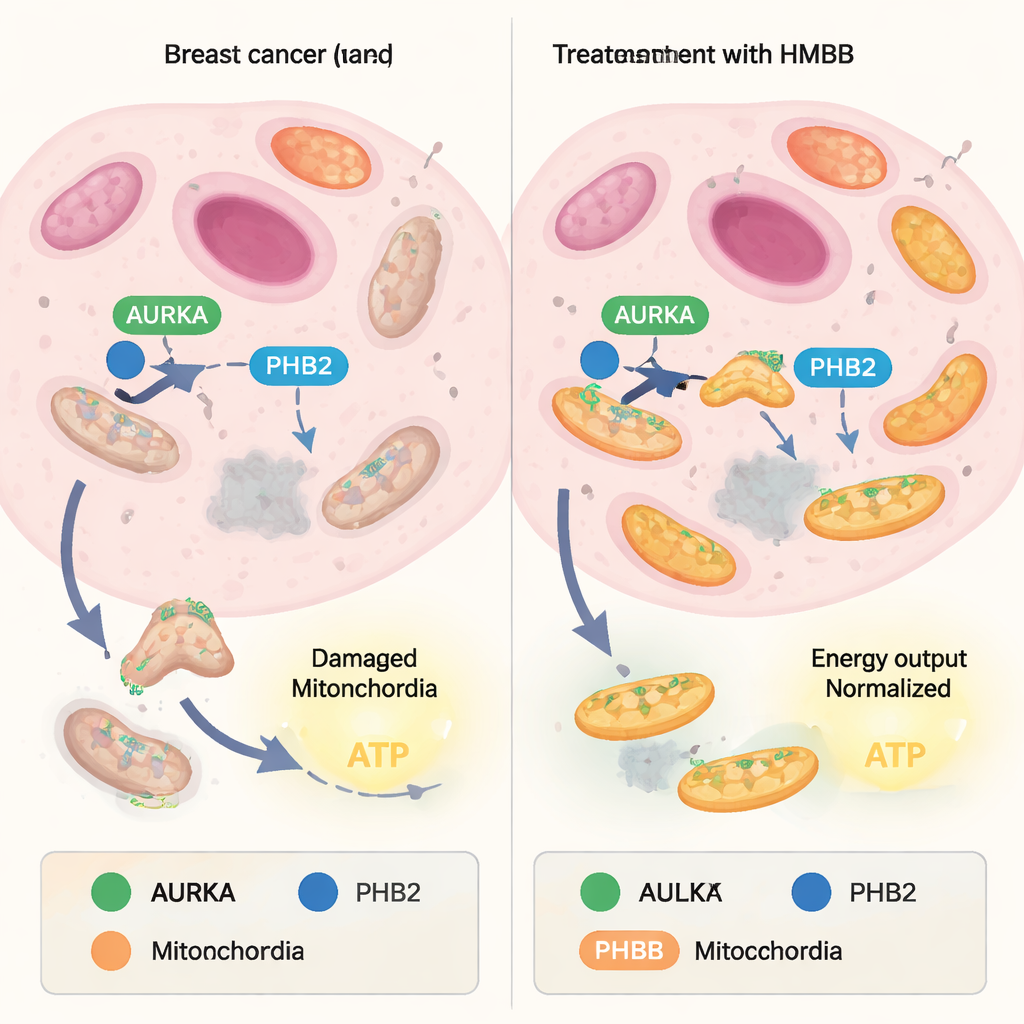
Mitochondria constantly balance building new units with recycling damaged ones. The recycling side, called mitophagy, acts like a quality-control system: worn-out mitochondria are tagged and broken down, while healthier ones remain. Many cancer cells hijack this balance, using mitophagy to weed out weak mitochondria and enrich for “super-performers” that churn out large amounts of ATP, the cell’s energy currency. A protein called Aurora kinase A (AURKA), often overproduced in tumors, was recently found inside mitochondria where it teams up with another protein, Prohibitin‑2 (PHB2), and the autophagy adaptor LC3. Together they form a complex that promotes mitophagy and supports high energy output in cancer cells.
From spicy capsaicin to tailored molecular tools
Earlier work had shown that a natural compound called xanthohumol can latch onto PHB2 and disrupt the AURKA–PHB2–LC3 complex, blocking mitophagy and dialing back mitochondrial ATP production. However, xanthohumol’s chemical structure is not ideal for drug development. In this study, the authors turned to capsaicin, another natural PHB ligand, and asked whether it could be reshaped into more practical tools. They first confirmed that capsaicin slightly weakens the interaction between AURKA and PHB2 in live breast cancer cells, using an imaging technique (FRET/FLIM) that measures how closely two tagged proteins sit next to each other. They then synthesized 16 capsaicin-inspired molecules by modifying the “tail” and the aromatic “head” of the original structure and systematically tested how each version affected the AURKA–PHB2 partnership inside mitochondria.
Discovering a molecular ‘glue’ that locks proteins together
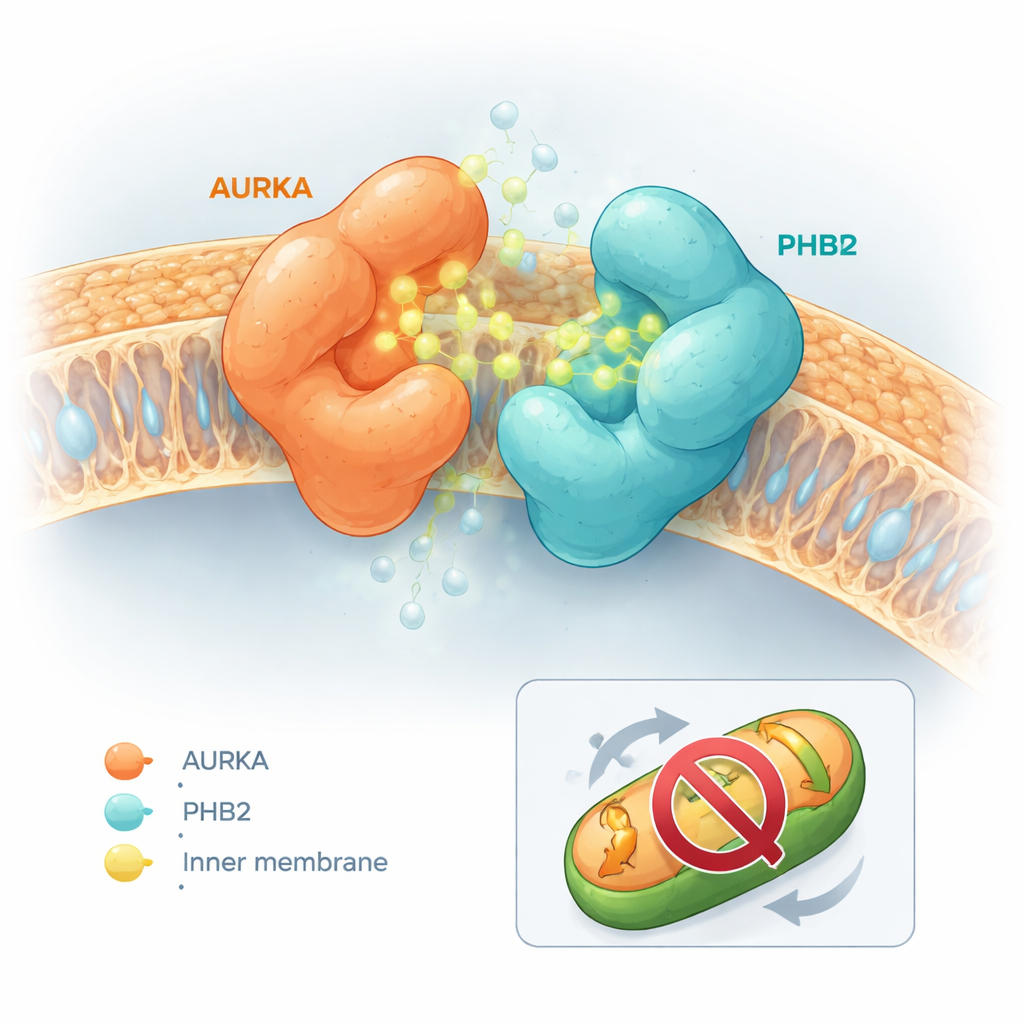
The modified molecules had strikingly different effects. Some, like capsaicin itself or a derivative called compound 5, made AURKA and PHB2 separate more easily. Others did almost nothing. A subset, however — especially a molecule named compound 13, later called HMBB — had the opposite effect and significantly tightened the bond between AURKA and PHB2. Computer-based docking simulations helped explain this behavior. Capsaicin and compound 5 tended to wedge into the active site of AURKA in ways that created physical clashes with PHB2, nudging the two proteins apart. In contrast, compounds 12 and 13 nestled into the AURKA active site and simultaneously contacted a known inhibitory pocket on PHB2 without causing a clash. Instead, their shapes allowed them to act as a “molecular glue,” bridging AURKA and PHB2 and stabilizing the complex.
Blocking mitochondrial cleanup without shutting down the cell’s control center
A crucial question was whether this molecular glue behavior simply disabled AURKA everywhere in the cell, which would risk broad side effects, or whether it acted more selectively. Using a separate fluorescent biosensor that reports on AURKA activation at centrosomes — the structures that organize the cell’s division machinery — the team observed that HMBB did not dampen AURKA activity at these sites. Yet in mitochondrial assays, HMBB reversed the loss of mitochondrial mass normally caused by AURKA overexpression in breast cancer cells. In cells that naturally make high levels of AURKA, HMBB increased mitochondrial content, indicating that AURKA-driven mitophagy was blocked. In cells with low AURKA levels, HMBB had little impact, underscoring its dependence on the AURKA–PHB2 pathway.
What this could mean for future cancer treatments
Taken together, the findings show that carefully designed capsaicin derivatives can fine‑tune the way AURKA and PHB2 interact in mitochondria. By acting as molecular glue, HMBB and related compounds lock AURKA and PHB2 together in a configuration that prevents the usual mitophagy program from proceeding, without shutting down AURKA’s essential roles elsewhere in the cell. For a lay reader, the key idea is that the authors have created prototype molecules that selectively interfere with how cancer cells keep their power plants in top condition, potentially weakening their energy supply while sparing normal cell functions. While these compounds are still at an early, laboratory stage, they illustrate a promising strategy: using small, drug-like molecules to target specific protein partnerships inside mitochondria and, in doing so, to undermine the metabolic advantages that many tumors depend on.
Citation: Djehal, A., Caron, C., Giordano, D. et al. Development of capsaicin-derived prohibitin ligands to modulate the Aurora kinase A/PHB2 interaction and mitophagy in cancer cells. Commun Biol 9, 320 (2026). https://doi.org/10.1038/s42003-026-09573-3
Keywords: mitochondria, mitophagy, Aurora kinase A, capsaicin derivatives, cancer metabolism