Clear Sky Science · en
Functional remodeling of iNKT cells by sulfatide-reactive type II NKT cells reprograms alveolar macrophages to alleviate lung ischemia-reperfusion injury
Why protecting donor lungs matters
When surgeons transplant a lung or restore blood flow after a blockage, the sudden return of oxygen can paradoxically injure the tissue. This problem, called lung ischemia–reperfusion injury, is a major cause of early failure after lung transplantation and other critical procedures. The study summarized here explores how a rare set of immune cells can be “reprogrammed” to calm inflammation, offering a potential new way to protect fragile lungs when they are needed most.
Immune cells that can heal or harm
Our lungs are patrolled by alveolar macrophages, immune cells that sit in the air sacs and respond quickly to danger. These cells can shift between two main modes. In a “fight” mode (often called M1), they pump out inflammatory substances that help kill germs but can also damage tissue. In a “repair” mode (often called M2), they release soothing molecules that limit inflammation and aid healing. In lung ischemia–reperfusion injury, the balance tilts toward the damaging fight mode. The researchers reasoned that if they could gently push macrophages toward the repair mode at the right moment, they might soften the blow of this injury.
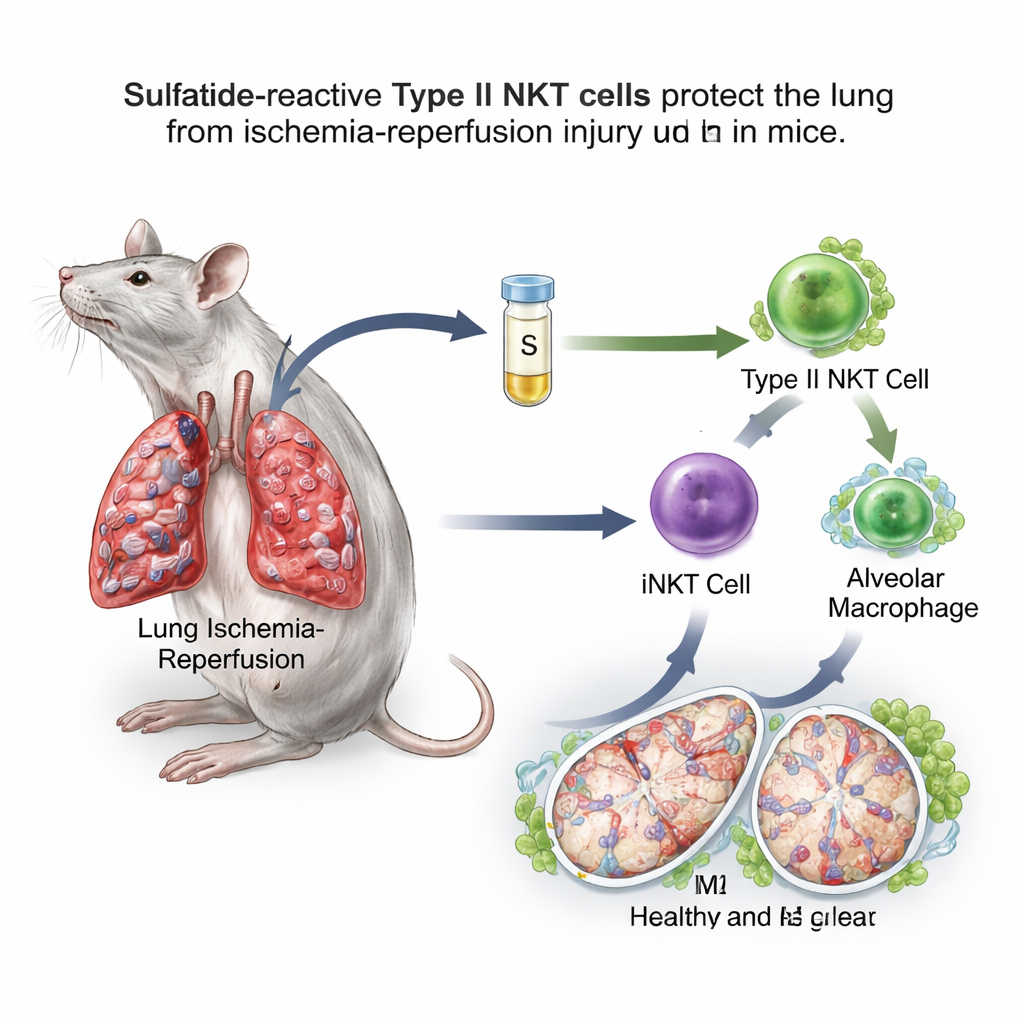
A sugar–fat molecule flips an immune switch
The team focused on natural killer T (NKT) cells, unusual immune cells that sit between fast-acting innate defenses and slower, targeted responses. NKT cells come in two main types. Type I, also called invariant NKT (iNKT) cells, are known to worsen lung ischemia–reperfusion in mice. Type II NKT cells, however, can restrain immune reactions in other organs. A fatty molecule called sulfatide is recognized specifically by type II NKT cells. In a mouse model where blood flow to one lung was clamped for an hour and then restored, the researchers gave sulfatide several hours before the insult. They found that sulfatide-treated mice had less lung swelling, fewer leaky blood vessels, lower oxidative stress, better oxygen levels, and reduced inflammatory substances in lung wash fluid compared with untreated animals.
Reprogramming macrophages through a relay of signals
Closer examination showed that sulfatide shifted alveolar macrophages toward the repair-oriented M2 state. These macrophages displayed more surface markers and genes linked to healing and were better at dampening inflammation when transferred into other mice. When the scientists selectively removed alveolar macrophages, lung injury worsened and sulfatide largely lost its protective effect, showing that these cells are a crucial hub in the pathway. Importantly, the benefit of sulfatide disappeared in mice lacking either type II NKT cells or type I iNKT cells, and it could be restored by transferring back iNKT cells that had been pre-exposed to sulfatide. Together, these experiments revealed a relay: sulfatide activates type II NKT cells, which in turn reshape the behavior of iNKT cells, and these reprogram alveolar macrophages.
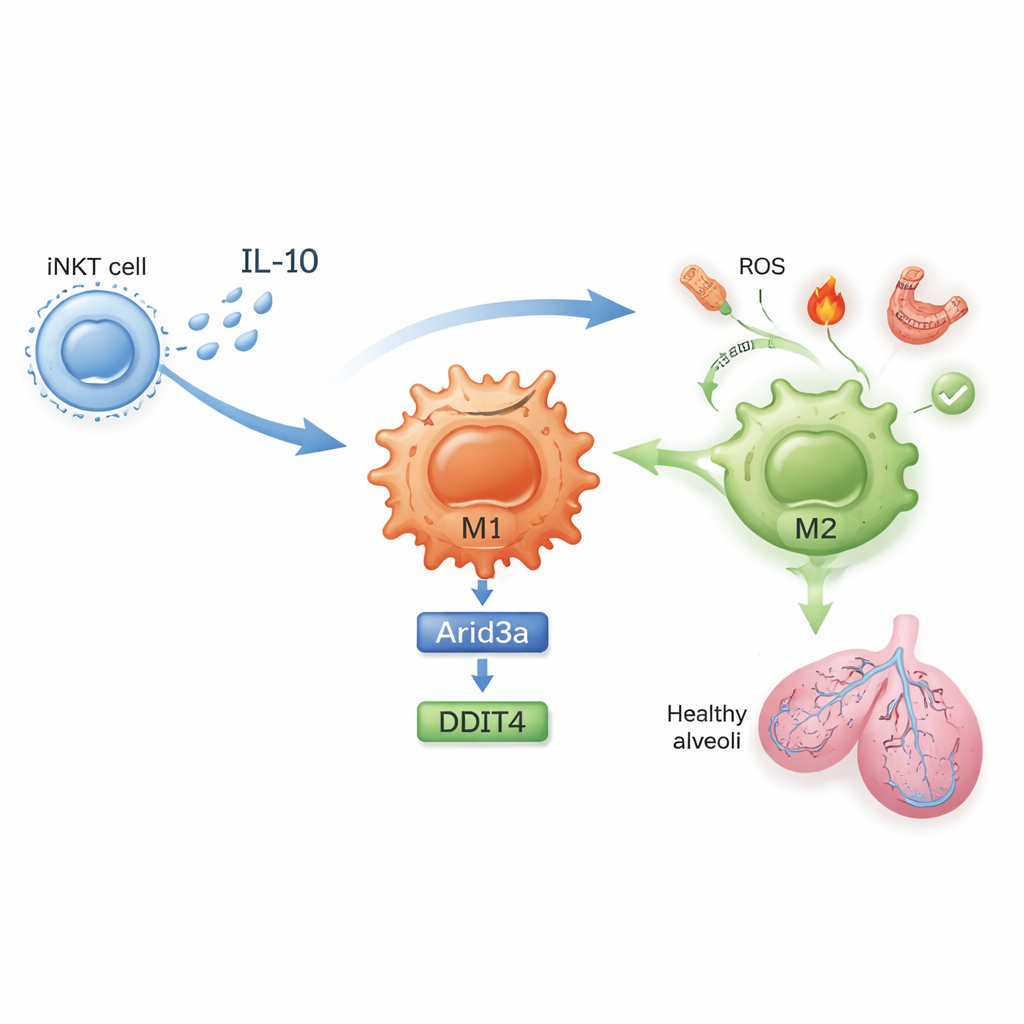
A molecular chain that calms inflammation
What exactly changes inside these cells? In untreated mice, lung iNKT cells proliferated during ischemia–reperfusion and produced high levels of interferon-gamma, a potent inflammatory signal. After sulfatide exposure, iNKT cells instead made more interleukin‑10 (IL‑10), a powerful anti-inflammatory molecule, and less interferon-gamma. The study shows that IL‑10 from iNKT cells acts directly on macrophages to drive their M2 polarization. Using gene expression profiling and carefully designed cell culture experiments, the authors uncovered a molecular chain inside macrophages: IL‑10 boosts a transcription factor called Arid3a, which in turn turns up a stress-response gene named DDIT4. This Arid3a–DDIT4 axis helps shut down a growth-control pathway that otherwise keeps macrophages in an inflammatory state, nudging them toward the protective M2 program.
From mouse lungs to future therapies
To test how central this molecular chain is, the researchers used small interfering RNAs to silence Arid3a or DDIT4 in macrophages before transferring them into mice. When either gene was knocked down, sulfatide no longer pushed macrophages fully into the repair mode, and lung protection was weakened. Additional analysis suggests that a signaling protein called YES1 helps sulfatide-activated type II NKT cells rewire iNKT cells toward IL‑10 production. While these findings come from mouse models and from engineered cells in the lab, they outline a clear story: by briefly activating a specific NKT cell subset with sulfatide, it may be possible to convert otherwise damaging immune responses into ones that protect transplanted or injured lungs. In the future, drugs that mimic this pathway could help more patients survive and thrive after life-saving lung procedures.
Citation: Li, Q., Yin, J., Lin, Q. et al. Functional remodeling of iNKT cells by sulfatide-reactive type II NKT cells reprograms alveolar macrophages to alleviate lung ischemia-reperfusion injury. Commun Biol 9, 289 (2026). https://doi.org/10.1038/s42003-026-09572-4
Keywords: lung ischemia reperfusion injury, alveolar macrophages, natural killer T cells, interleukin-10, immune modulation