Clear Sky Science · en
Cell-based and isoform-selective G protein-coupled receptor kinase assays for comprehensive inhibitor evaluation
Why turning down cellular “volume knobs” matters
Many of our medicines work by dialing up or down the activity of cell-surface receptors that sense hormones, neurotransmitters, and drugs. These receptors must be carefully switched off again so cells do not stay overstimulated, a process partly controlled by enzymes called GRKs. When GRKs are too active, as seen in heart failure and some cancers, signaling goes awry. This study develops practical, cell-based tests to measure how well experimental molecules can block specific GRKs, helping scientists design smarter drugs that fine-tune these crucial cellular volume knobs.
Gatekeepers on the cell surface
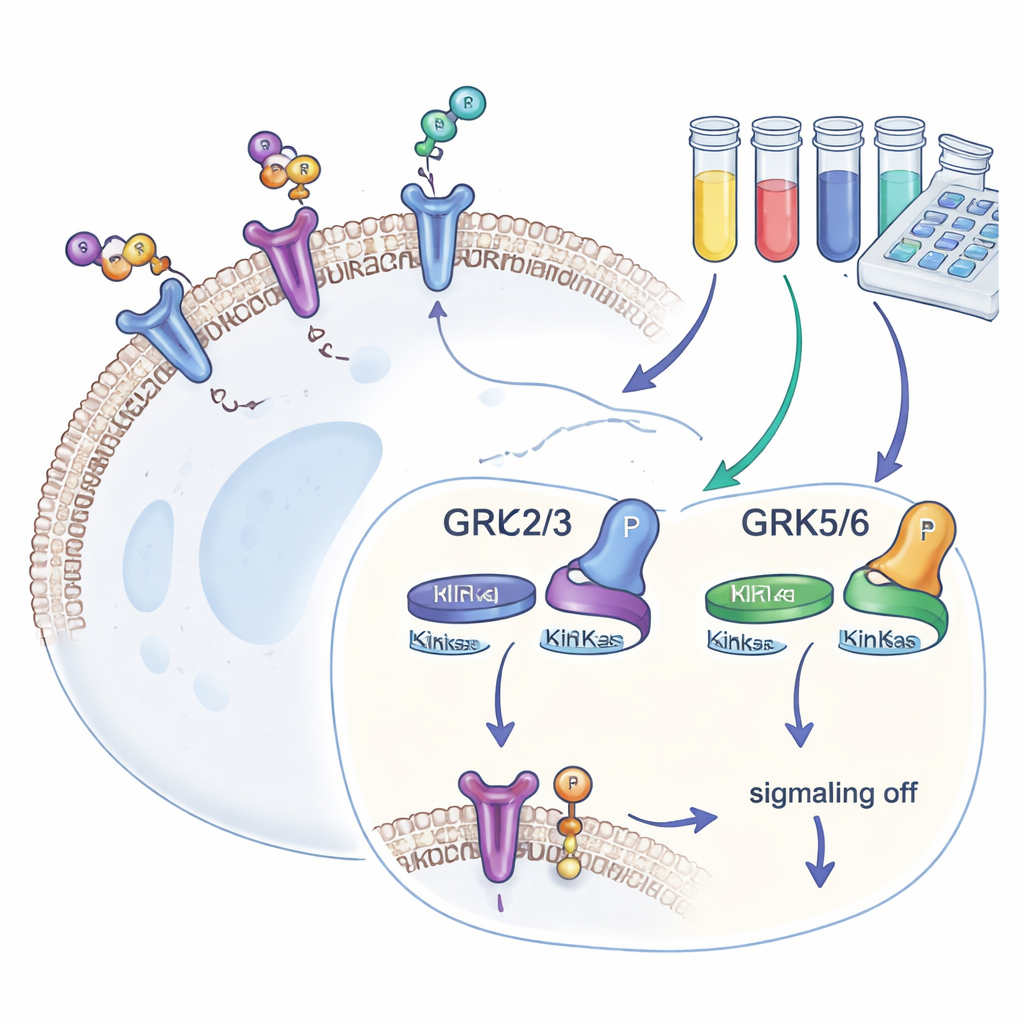
Our cells carry hundreds of types of G protein-coupled receptors (GPCRs), which detect outside signals and translate them into internal responses. After a GPCR has been triggered, GRKs attach small phosphate “flags” to its tail. These flags attract another protein, beta-arrestin, which stops further signaling and often pulls the receptor inside the cell. Four GRK versions—GRK2, GRK3, GRK5, and GRK6—are found in many tissues. Because they shape how strongly GPCRs respond, and because their levels are altered in diseases such as heart failure, cancer, and addiction, drug developers are keen to find GRK blockers that are both potent and selective.
Building a clean test bed inside cells
Most previous GRK studies relied on computer modeling or test-tube chemistry, which reveal how tightly an inhibitor can bind but not how it performs in the crowded interior of a living cell. To bridge this gap, the authors engineered human HEK293 cells that lack all four common GRKs and then reintroduced only one GRK isoform at a time. Each cell line also carried a well-studied receptor, the beta-2 adrenergic receptor, tagged so its phosphorylation at a specific tail site (called T360/S364) could be read out using a sensitive antibody-based assay. Because this site is modified only by GRKs, the amount of phosphate present serves as a direct, quantitative measure of how active each GRK isoform is inside living cells.
Sorting the good, the weak, and the nonspecific
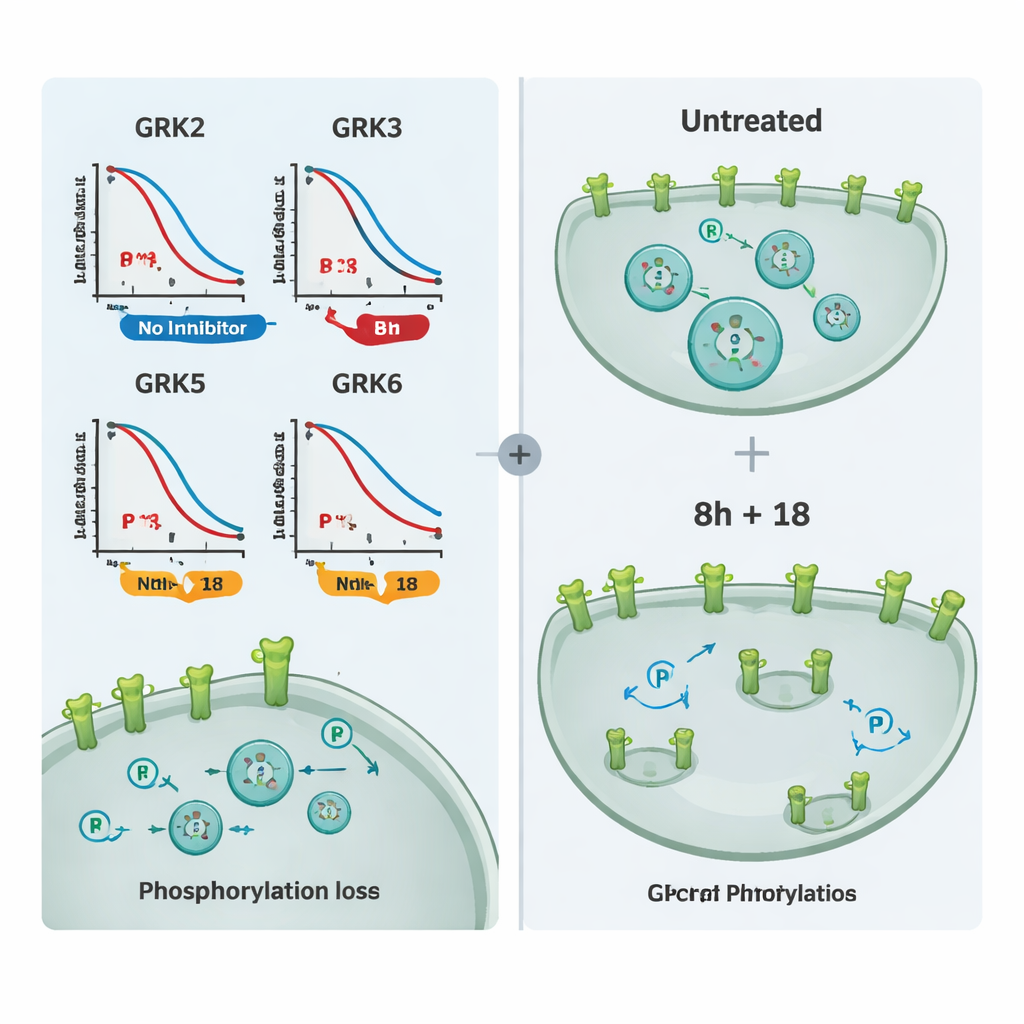
Using this toolkit, the team tested a panel of commercially available GRK blockers. They first grouped compounds that mainly target GRK2 and GRK3, and another set aimed at GRK5 and GRK6. By comparing how much each molecule reduced receptor phosphorylation in cells expressing only one GRK subtype, they could map real-world selectivity. One compound, called 8h, emerged as the most potent blocker of GRK2/3, while compound 18 stood out for selectively inhibiting GRK5/6. Some widely used molecules showed little effect at the tested doses, likely because they did not enter cells efficiently, and a very powerful covalent inhibitor disrupted cell health, making it unsuitable for imaging experiments.
From chemical fingerprints to receptor behavior
To show that these inhibitors affect not just a single test receptor but GPCR biology more broadly, the authors examined several medically important receptors, including the mu-opioid receptor and the vasopressin V2 receptor. They measured both phosphorylation and receptor internalization by microscopy. Compound 8h or 18 alone partially reduced phosphorylation and the inward movement of receptors for many targets, but the combination of 8h and 18 almost completely prevented these changes and kept receptors on the cell surface. Additional experiments tracking recruitment of beta-arrestin confirmed that the same compounds could tune signaling at other receptors regulated by overlapping sets of GRKs.
What this means for future medicines
For non-specialists, the key message is that the study delivers a reliable set of cell-based tests—and two especially useful tool compounds, 8h and 18—that let researchers see, in living cells, exactly how different GRK isoforms are turned down. Rather than guessing from simplified test-tube data, scientists can now compare candidate inhibitors side by side and decide whether they mainly affect GRK2/3, GRK5/6, or all four at once. This clarity should speed the development of drugs that more precisely modulate GPCR signaling, with potential benefits for treating heart disease, cancer, pain disorders, and other conditions where signaling balance has gone off-kilter.
Citation: Blum, N.K., Kiefer, M.C., Decker, A. et al. Cell-based and isoform-selective G protein-coupled receptor kinase assays for comprehensive inhibitor evaluation. Commun Biol 9, 287 (2026). https://doi.org/10.1038/s42003-026-09568-0
Keywords: GPCR signaling, GRK inhibitors, beta-adrenergic receptor, cell-based assay, drug discovery