Clear Sky Science · en
HBP1 enhances progesterone receptor activity and IGFBP1 expression driving endometrial decidualization
Why this matters for fertility
When a pregnancy begins, the embryo can only settle into the womb if the uterine lining has gone through a crucial makeover called decidualization. Many cases of infertility and repeated failure of embryo transfer remain unexplained, and growing evidence suggests that something goes wrong in this makeover rather than in the embryo itself. This study uncovers a previously underappreciated molecular "switch," a transcription factor named HBP1, that helps prepare the uterine lining for implantation and may offer new ways to diagnose and treat implantation problems.
A hidden switch in the uterine lining
The inner lining of the uterus, the endometrium, changes rhythmically with each menstrual cycle. Under the influence of estrogen it grows, and under progesterone it matures into a receptive bed for an embryo. In this work, researchers focused on human endometrial stromal cells, the structural cells that transform into larger, more secretory decidual cells at the time of implantation. By mining existing gene activity data and performing new experiments, they found that HBP1 levels rise sharply as these stromal cells begin decidualizing, suggesting that HBP1 is part of the built-in timing system that readies the uterus.
Helping cells change shape and slow down
To test whether HBP1 is merely present or truly needed, the team dialed its levels up and down in cultured human endometrial stromal cells. When HBP1 was reduced, classic decidualization markers such as IGFBP1, FOXO1, and prolactin dropped at both the RNA and protein level. The cells also failed to undergo the typical shape change from slim, spindle-like cells to broader, polygonal ones and instead kept proliferating. When HBP1 was increased, IGFBP1 levels rose and cell division slowed. Together these findings show that HBP1 helps cells exit the growth mode and enter the specialized decidual state that supports early pregnancy.
Fine-tuning hormone signals inside the cell
Progesterone and its receptor are central to preparing the uterus, but not all tissues respond equally well to the hormone. The researchers discovered that progesterone signaling itself boosts HBP1 levels, creating a positive feedback loop. Strikingly, lowering HBP1 did not change how much progesterone receptor the cells made, but it did blunt the activity of important progesterone-responsive genes, including FKBP4, FKBP5, FOSL2, and coactivator SRC1. Using genome-wide approaches, they showed that HBP1 binds near many of these genes and is associated with an increase in a specific histone mark, H3K4me3, which flags DNA as active. In essence, HBP1 does not turn the receptor on or off; it makes the receptor’s target genes easier to read.
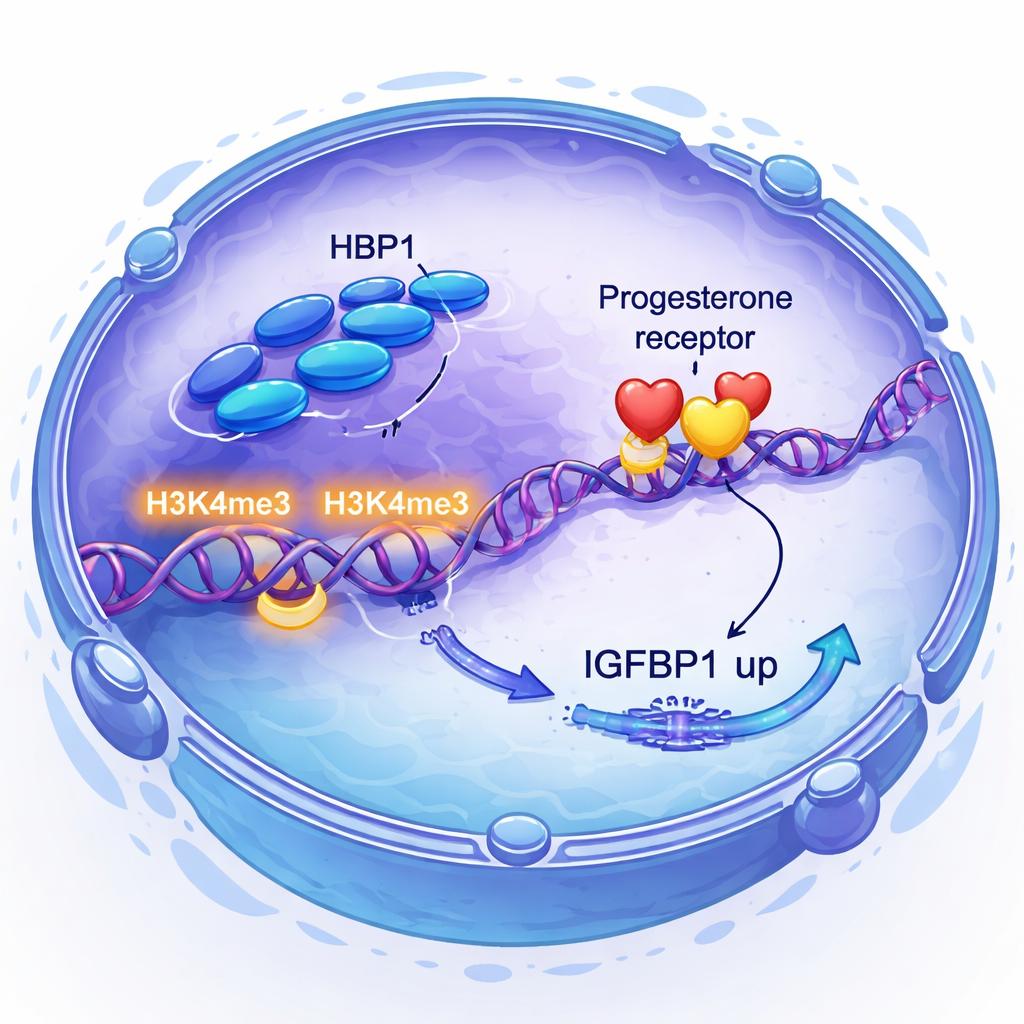
Direct control of a key implantation signal
The team then asked how HBP1 connects to IGFBP1, a protein long used as a marker of healthy decidualization and known to influence embryo–uterus communication. By combining RNA sequencing with chromatin immunoprecipitation sequencing, they showed that HBP1 sits directly on the IGFBP1 promoter region and increases H3K4me3 there, driving higher IGFBP1 production. They also found that reducing HBP1 activated the PI3K–AKT pathway, a growth-related signal that, when overactive, suppressed IGFBP1. Blocking this pathway rescued IGFBP1 levels. Thus HBP1 supports decidualization in two ways: by directly turning on IGFBP1 and by damping a growth signal that would otherwise hold decidual genes back.
Linking a molecular defect to failed implantation
Finally, the researchers looked at endometrial samples from women experiencing recurrent implantation failure and compared them with tissues from fertile controls during the mid-secretory, implantation-ready phase. Women with repeated failure had notably lower levels of HBP1 and its downstream partners IGFBP1, FKBP5, and FOSL2, even though progesterone receptor levels were similar between groups. This pattern fits with the idea of "progesterone resistance"—the hormone is present, but the tissue cannot mount a full response because key support factors like HBP1 are missing.
What this means for patients
In accessible terms, this study suggests that HBP1 acts like a master setting on the uterine lining’s control panel. When properly turned up, it helps progesterone signals be heard, encourages cells to stop dividing and specialize, and boosts the production of implantation-friendly molecules such as IGFBP1. When HBP1 is too low, the lining may look normal under a microscope but function poorly, leading to embryos that simply cannot gain a foothold. Understanding and measuring HBP1 and its network could, in the future, help doctors identify women at risk for implantation problems and inspire new treatments that restore the womb’s receptivity.
Citation: Guo, Y., Tian, W., Nie, C. et al. HBP1 enhances progesterone receptor activity and IGFBP1 expression driving endometrial decidualization. Commun Biol 9, 286 (2026). https://doi.org/10.1038/s42003-026-09567-1
Keywords: endometrial decidualization, embryo implantation, progesterone signaling, infertility, HBP1