Clear Sky Science · en
Entrained cortical delta–spindle activity, not periodic synchrony, prevents arousal by NREM thalamic bursts
Why sleepy brains can ignore powerful signals
When we sleep, our brains are far from quiet: deep inside, cells fire in rapid bursts that, during the day, can help grab our attention. Yet at night, these same bursts usually fail to wake us. This study asks a surprisingly simple question with big implications for sleep, consciousness, and disorders like insomnia and Parkinson’s disease: why don’t these strong signals from the thalamus, a key relay hub in the brain, rouse the sleeping cortex?
Meet the brain’s night‑shift relay station
The thalamus sits near the center of the brain and helps route information between the senses, deeper structures, and the cortex. In this work, researchers recorded from two specific thalamic nuclei in non‑human primates: the ventral anterior (VA) and centromedian (CM) nuclei. These areas talk both to movement‑related regions and to circuits that control arousal. At the same time, the team monitored standard sleep signals (EEG, eye movements, muscle activity) while the monkeys cycled naturally through wakefulness, non‑REM (NREM) sleep, and REM (dream) sleep. 
Bursts that are stronger, but not rhythmically locked
Thalamic cells have two main firing styles. In tonic mode, they emit a fairly steady stream of spikes; in burst mode, they fire short, rapid volleys of spikes. During wakefulness and REM sleep, VA and CM neurons mostly fired tonically at similar rates. In NREM sleep, their overall firing rate dropped, but bursting dramatically increased: more than two‑thirds of 10‑second windows were dominated by burst firing. Despite this, the timing of bursts was surprisingly irregular. Careful analyses of the intervals between bursts and their frequency content showed no strong periodic peaks—bursts clustered in time but did not form a clock‑like rhythm. This challenges the textbook idea that sleep bursts are neatly periodic “null messages” to the cortex.
Not marching in lockstep
If many neurons burst together, their combined impact on the cortex could be huge. The authors therefore examined how tightly different thalamic neurons synchronized their bursts, whether they were recorded from the same microelectrode or from opposite hemispheres. Cross‑correlation measures revealed only very small peaks around zero time lag, indicating that bursts from different cells tend to be only loosely coordinated and spread over long time windows. Even when the analysis was adjusted to capture slower, broader co‑fluctuations, synchrony remained weak. In other words, during NREM sleep the thalamus does not operate as a tightly beating metronome, but rather as many semi‑independent relays.
State‑dependent conversations with the cortex
If periodicity and tight synchrony are not the explanation, why don’t these powerful bursts wake the brain? To probe this, the researchers aligned each burst to EEG activity at the scalp and field potentials in the thalamus itself. During NREM sleep, the EEG began to dip toward a negative phase about a second before each burst, then flipped into a positive wave and was followed by slow oscillations and sleep spindles—hallmark features of deep sleep. Spectral analyses showed that NREM bursts were tightly linked to delta waves and spindles, reinforcing the ongoing sleep pattern rather than disrupting it. In wake and REM, the same bursts produced much smaller, differently shaped responses, more in line with active processing. Importantly, bursts did not systematically precede awakenings or brief “micro‑arousals”; if anything, they tended to favor staying in, or returning to, NREM sleep. 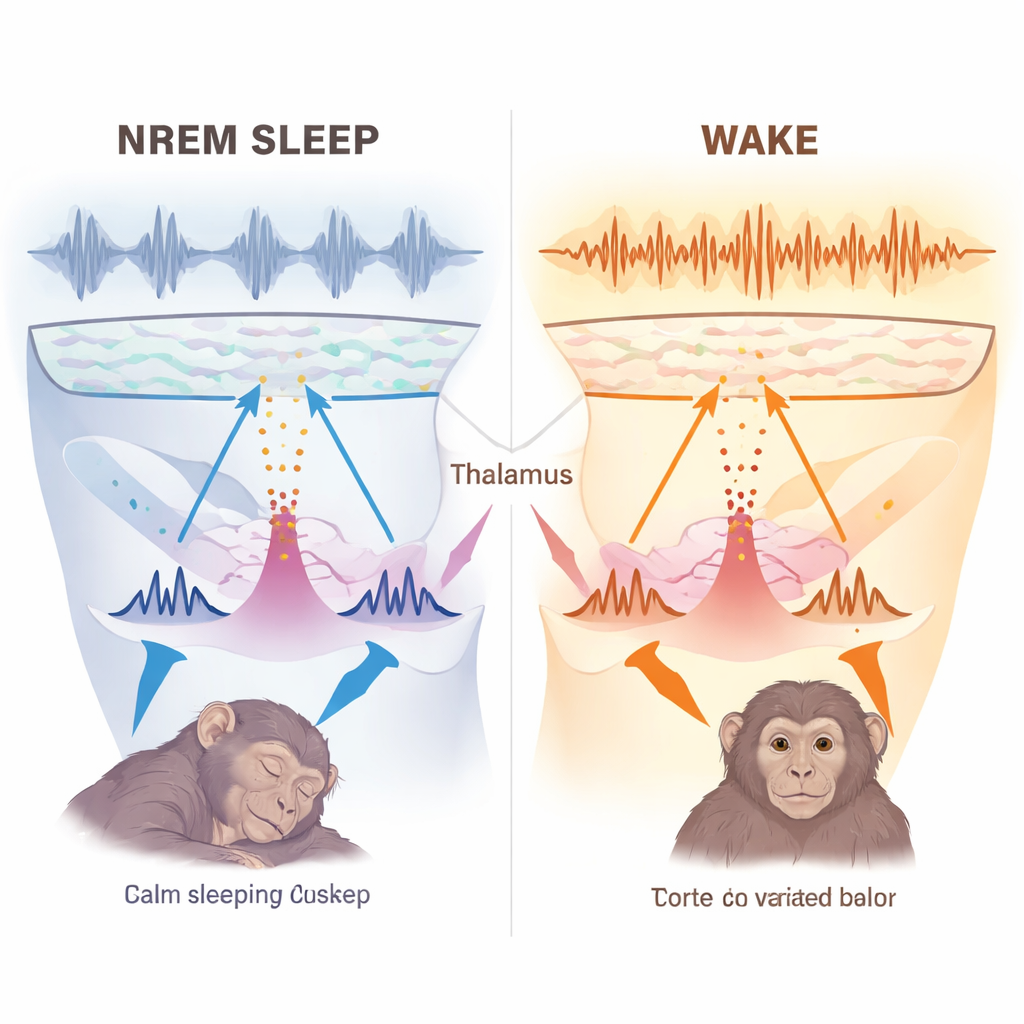
Rethinking who drives whom at night
These findings support a new picture of the sleeping brain. The authors argue that the basal ganglia, which send inhibitory signals to VA and CM, modulate these thalamic nuclei rather than fully controlling them during NREM sleep. At night, the thalamus and cortex appear to form a self‑sustaining loop: cortical slow waves help set the conditions for thalamic bursts, and those bursts in turn help build the familiar delta waves and spindles that define deep sleep. Under these special state‑dependent dynamics, the same type of thalamic burst that can serve as a vivid "wake‑up call" during active states instead becomes part of the machinery that keeps the cortex asleep.
What this means for understanding sleep
To a non‑specialist, the key message is that it is not the mere presence of strong thalamic bursts, nor their regular timing or perfect synchrony, that determines whether we wake up. Rather, what matters is the broader context: in NREM sleep, the cortex and thalamus are wired and chemically tuned so that bursts are absorbed into ongoing delta and spindle rhythms instead of breaking through to consciousness. This shift in perspective may help explain why deep sleep feels so disconnected from the outside world, and could guide future work on sleep disorders and on therapies that tap into thalamocortical circuits without disturbing restorative sleep.
Citation: Liu, X., Guang, J., Israel, Z. et al. Entrained cortical delta–spindle activity, not periodic synchrony, prevents arousal by NREM thalamic bursts. Commun Biol 9, 285 (2026). https://doi.org/10.1038/s42003-026-09565-3
Keywords: sleep, thalamus, NREM, brain rhythms, arousal