Clear Sky Science · en
Differential GABA dynamics across brain functional networks in autism
Why this brain study matters
Many people on the autism spectrum describe the world as overwhelming, with sounds, lights, and emotions feeling too intense or strangely muted. Scientists suspect that part of the reason lies in how brain cells balance "go" and "stop" signals. This study looks at one key "stop" chemical, GABA, and asks a practical question: how does the autistic brain respond when we nudge this system with a drug? The answer could help explain why some medications work unpredictably in autism and why getting the dose right is so challenging.
Signals that keep brain activity in balance
Our brains run on a constant tug-of-war between excitation (neurons firing) and inhibition (neurons calming things down). GABA is the main chemical that provides this braking function. In autism, years of research suggest that this balance is off, especially in brain systems that handle sensory information such as vision, hearing, and touch. But most previous work has been static: measuring brain chemistry or structure at rest and comparing autistic and non-autistic people. What has been missing is a dynamic test of how the GABA system actually responds when pushed by a drug challenge, especially across the large-scale networks that support sensation, movement, attention, and emotion.
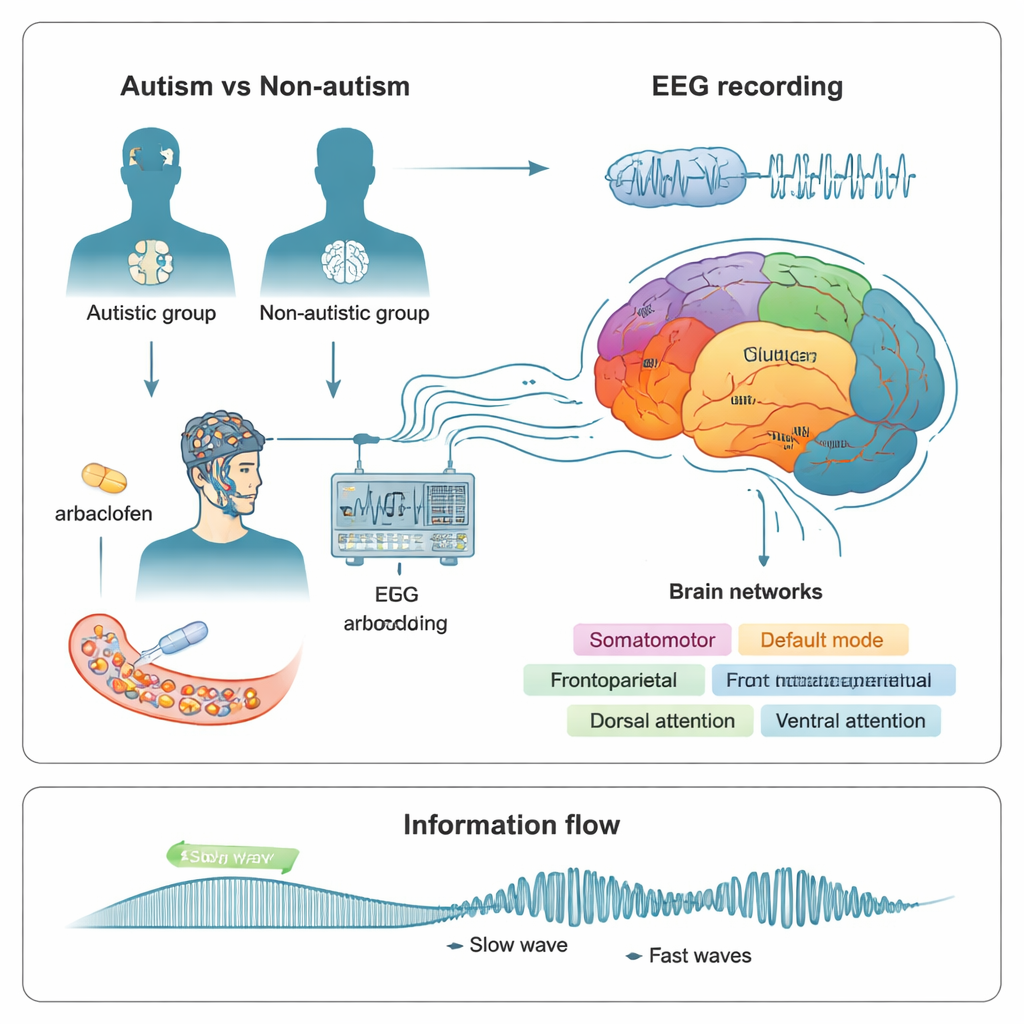
Listening in on brain rhythms
To probe this, the researchers recorded resting brain activity using EEG, which measures tiny electrical signals from the scalp. They studied 24 non-autistic adults and 15 autistic adults over multiple visits. Each time, participants swallowed either a placebo or one of two doses (15 mg or 30 mg) of arbaclofen, a drug that activates a specific type of GABA receptor called GABAB. About three hours later, when the drug was active, the team recorded EEG with eyes open and closed and used computer models to trace the signals back to 400 locations across the cortex. These locations were then grouped into seven major functional networks, including visual, somatomotor (movement and touch), limbic (emotion and memory), and several higher-order thinking and attention networks.
How slow and fast brain waves talk
Rather than looking only at how strong certain brain waves were, the team focused on how different frequencies interact. In healthy brains, slow waves that span large regions often help coordinate faster, more local bursts of activity. This interaction, called phase–amplitude coupling, can be thought of as a slow rhythm opening and closing "windows" in which fast activity is more likely. The authors measured how strongly slow rhythms in ranges like theta and alpha were locking together with faster beta and gamma activity, both within and between the seven networks. Stronger coupling may sometimes be useful, but if it becomes too tight or inflexible it can signal an imbalance in how information flows through the brain.
Autistic brains show tighter coupling at rest
Under placebo, autistic participants showed consistently higher coupling between theta and beta rhythms across most brain networks when their eyes were closed, compared with non-autistic volunteers. The limbic system stood out: there, all four coupling measures the team examined were elevated, suggesting unusually strong links between slow, widespread rhythms and fast local activity in regions involved in emotion and memory. The somatomotor network also showed heightened coupling between theta and gamma rhythms. These patterns support the idea that excitation–inhibition balance is altered at the level of dynamic brain rhythms in autism, especially in sensory and emotional networks.
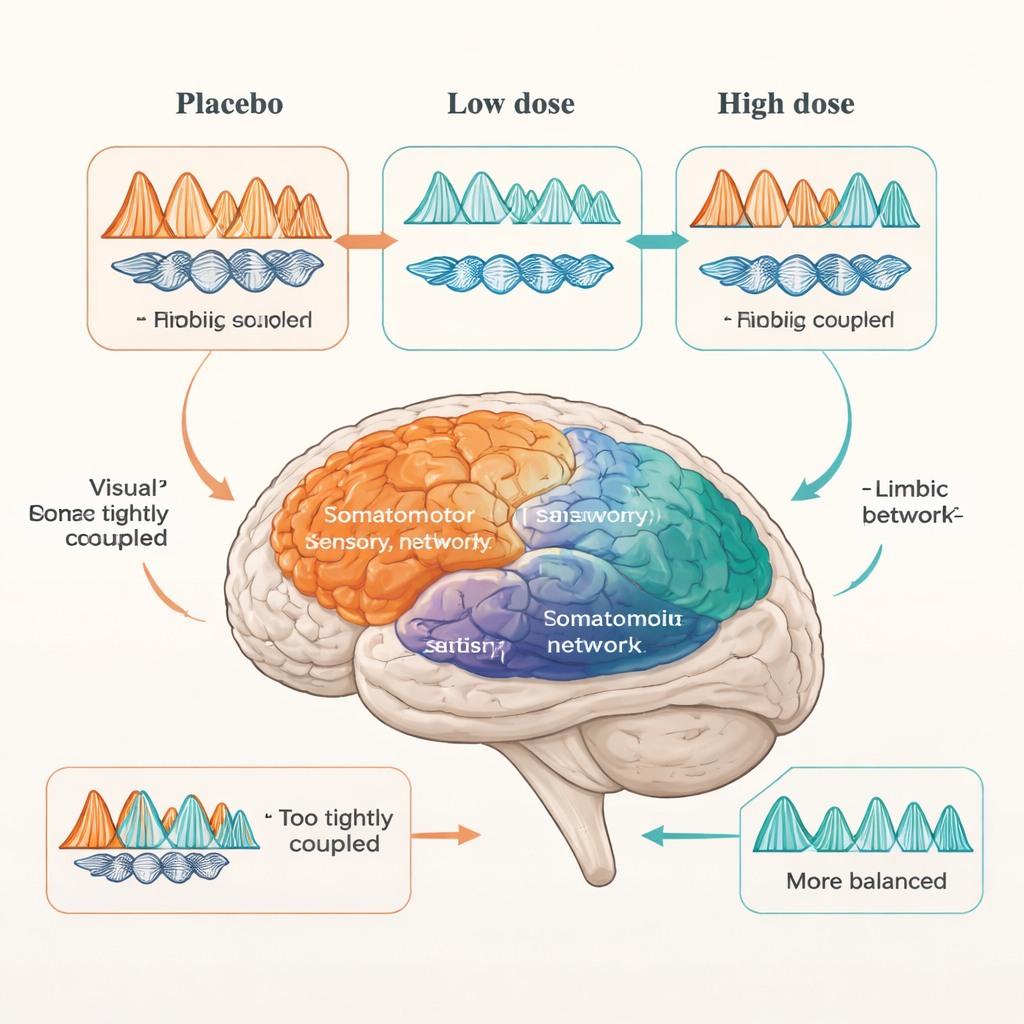
Dose matters—and different networks behave differently
When the researchers introduced arbaclofen, the picture became more complex and dose-dependent. In autistic participants, the higher 30 mg dose shifted the elevated theta–beta coupling in visual and somatomotor networks toward the non-autistic range, suggesting a more typical pattern of sensory information flow. However, higher-order networks supporting planning, self-referential thought, and attention changed very little. The limbic system behaved differently again: a low 15 mg dose brought its exaggerated coupling—both within the network and in its connections to other networks—closer to control levels. But at 30 mg, many of these abnormalities reappeared, and atypical limbic links to other networks, such as the somatomotor system, resurfaced. In other words, some emotional circuits seemed to respond best to a lower dose and could be over-driven at higher doses.
What this means for real-world treatments
For a layperson, the main message is that the autistic brain does not respond to GABA-acting drugs in a simple, uniform way. Different brain networks—sensory, emotional, and higher-thinking systems—show distinct patterns of sensitivity, and some circuits are especially dose-sensitive. This helps explain why medications that target inhibition can sometimes have paradoxical or mixed effects in autism, helping in one domain while disturbing another. While this study does not test whether arbaclofen improves everyday symptoms, it shows that carefully chosen doses can nudge certain autistic brain networks toward a more typical balance of activity. Future work may use this kind of brain-based "stress test" to personalize treatments, aiming to restore more flexible, better-tuned communication among the brain’s networks.
Citation: Huang, Q., Chen, D., Pereira, A.C. et al. Differential GABA dynamics across brain functional networks in autism. Commun Biol 9, 283 (2026). https://doi.org/10.1038/s42003-026-09563-5
Keywords: autism, GABA, brain networks, EEG, arbaclofen