Clear Sky Science · en
Gut metabolite indole-3-acetic acid aggravates neuropsychiatric lupus via the AHR/STAT3 pathway in microglia
How Gut Germs Can Affect the Mind
People with lupus, an autoimmune disease, sometimes develop serious problems with thinking, mood, and memory—a condition called neuropsychiatric lupus. These brain-related symptoms can be disabling, yet doctors still struggle to explain exactly why they occur or how to treat them. This study explores an unexpected culprit: certain gut bacteria and the chemicals they produce, which may travel from the intestine to the brain and fan the flames of inflammation there.
From Gut Microbes to Troubled Behavior
The researchers began by asking whether gut microbes from patients with neuropsychiatric lupus differ in their effects from those of patients with lupus who lack brain symptoms. They transferred stool bacteria from each group of patients into healthy mice that had first been cleared of their own gut microbes. Mice receiving microbes from neuropsychiatric lupus patients developed more anxiety-like behavior, signs of depression, and problems with learning and memory than mice that received microbes from other lupus patients. Their brains showed increased activation of microglia—the immune cells of the brain—and higher levels of inflammatory molecules. The protective seal around the brain, known as the blood–brain barrier, was also leakier in these animals, suggesting that something in the transplanted microbiota was damaging this critical barrier.
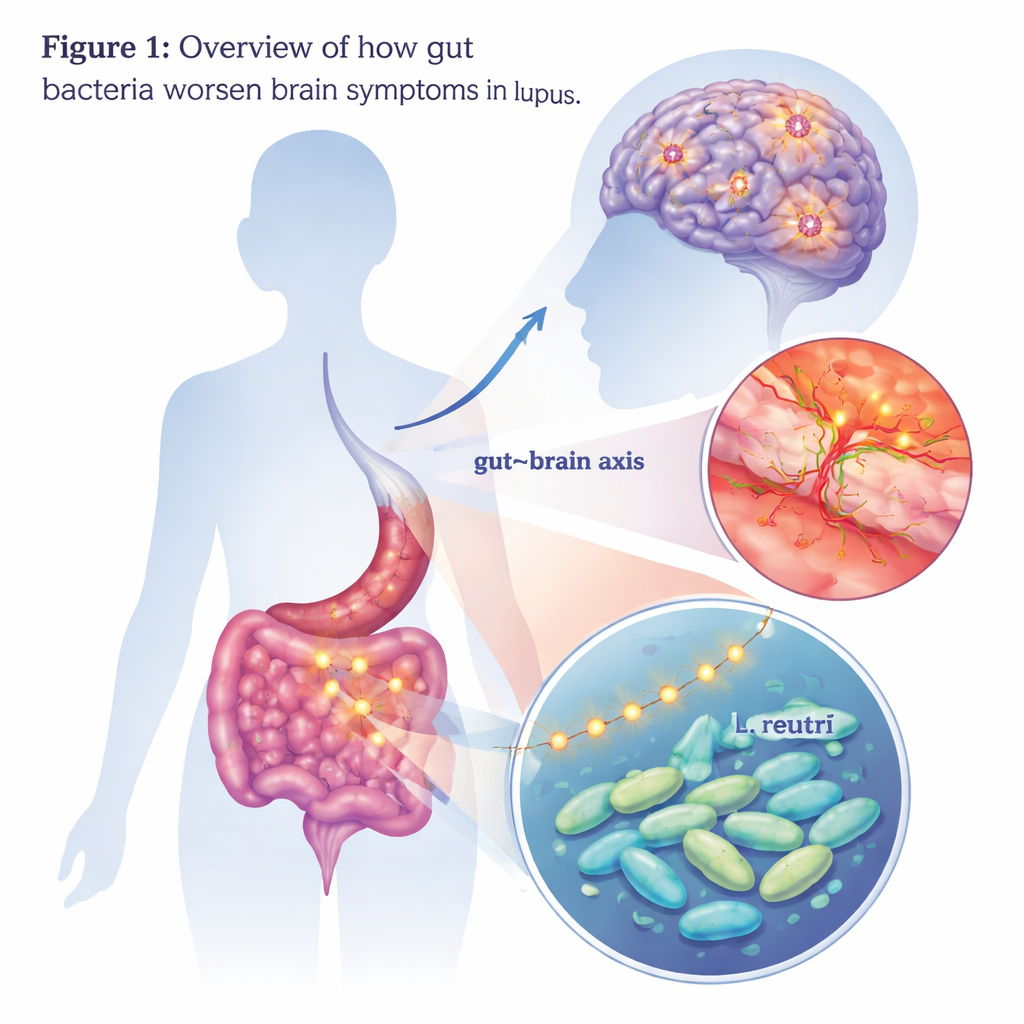
Zooming In on a Single Bacterium
To pinpoint which microbes might be responsible, the team compared the gut bacterial communities of patients and mice. One group stood out: Lactobacillus, and especially a species called Lactobacillus reuteri, was more abundant in patients with neuropsychiatric lupus and in mice that showed brain changes. When the scientists fed L. reuteri alone to lupus-prone mice, the animals developed stronger anxiety- and depression-like behaviors and did worse on memory tests. Their brains showed more activated microglia, greater loss of neurons, and further disruption of the blood–brain barrier. These findings suggested that, at least in the context of lupus, this normally probiotic-like bacterium can take on a more harmful role.
A Small Molecule with Big Effects
L. reuteri is known to produce a chemical called indole‑3‑acetic acid (IAA) from dietary tryptophan. Using sensitive chemical testing, the team found higher levels of IAA in the stool of neuropsychiatric lupus patients than in other lupus patients, and they confirmed that L. reuteri cultures make much more IAA than common gut bacteria such as Escherichia coli. In patients with neuropsychiatric lupus, IAA levels in the fluid surrounding the brain and spinal cord correlated with higher levels of inflammatory cytokines, hinting that this molecule can reach the central nervous system and stir up inflammation. In lupus-prone mice, feeding L. reuteri raised IAA levels in the gut, blood, and brain. Giving IAA alone to these mice reproduced many of the same changes: abnormal behaviors, microglial activation, higher inflammatory signals, and a weaker blood–brain and intestinal barrier.
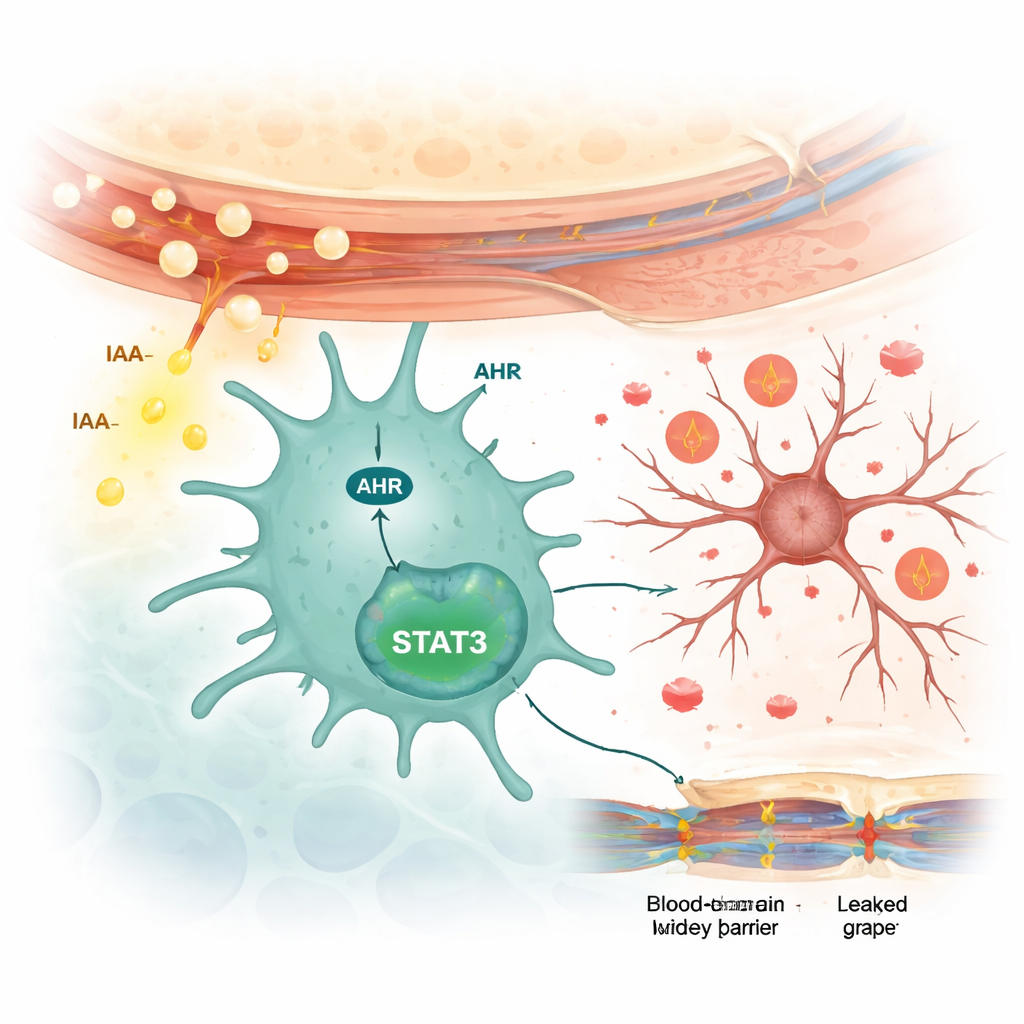
How Brain Immune Cells Are Switched On
The scientists then examined how IAA communicates with brain cells. They found that IAA increased the activity of a sensor protein called the aryl hydrocarbon receptor (AHR) specifically in microglia, not in neurons or star-shaped support cells called astrocytes. In cultured microglial cells, IAA boosted AHR levels and switched on a downstream signaling protein named STAT3, which in turn drove the production of inflammatory cytokines. Blocking AHR with a drug prevented STAT3 activation and cut down the release of these inflammatory signals. When lupus-prone mice were treated with the AHR-blocking drug, their anxiety- and depression-like behaviors improved, microglial activation decreased, inflammatory molecules dropped, and the blood–brain barrier became less leaky.
What This Means for Patients
Altogether, the study outlines a chain of events that links the gut to the brain in neuropsychiatric lupus. An overabundance of L. reuteri in the intestine produces extra IAA, which weakens the intestinal barrier and enters the bloodstream. IAA then reaches the brain, where it turns on microglial AHR and STAT3 signaling, triggering inflammation that harms neurons and loosens the blood–brain barrier. For patients, these findings suggest that altering gut bacteria, reducing harmful metabolites like IAA, or blocking the AHR/STAT3 pathway in microglia might one day help protect the brain from the devastating effects of neuropsychiatric lupus.
Citation: Feng, Y., Zheng, L., Tang, W. et al. Gut metabolite indole-3-acetic acid aggravates neuropsychiatric lupus via the AHR/STAT3 pathway in microglia. Commun Biol 9, 281 (2026). https://doi.org/10.1038/s42003-026-09561-7
Keywords: neuropsychiatric lupus, gut microbiota, Lactobacillus reuteri, microglia inflammation, blood–brain barrier