Clear Sky Science · en
HCN channels reveal conserved and divergent physiology in supragranular pyramidal neurons in primate species
Why brain wiring differences matter
Humans clearly think and behave differently from mice, but neuroscientists are still working out what that means for the individual nerve cells that make up our brains. This study focuses on a special set of neurons in the upper layers of the cortex—cells that help link different brain areas—and asks whether a key electrical feature that sets human neurons apart from rodents is uniquely human, or instead shared across primates. The answer helps explain how primate brains handle slow rhythmic activity that supports perception, memory, and attention.
Special channels that shape neural rhythm
Neurons communicate using tiny electrical signals. These signals are strongly shaped by pores in the cell membrane called ion channels. The authors concentrate on HCN channels, which quietly open when a neuron’s voltage drifts more negative and then gently pull it back toward its resting level. This self-correcting current makes neurons respond best to inputs in the slow "delta" and "theta" ranges (about 1–8 cycles per second), frequencies commonly seen in brain waves during sleep, navigation, and focused attention. Earlier work showed that human upper-layer pyramidal neurons have especially strong HCN-related properties compared with mice, raising the possibility that these channels are part of what makes the human cortex special.
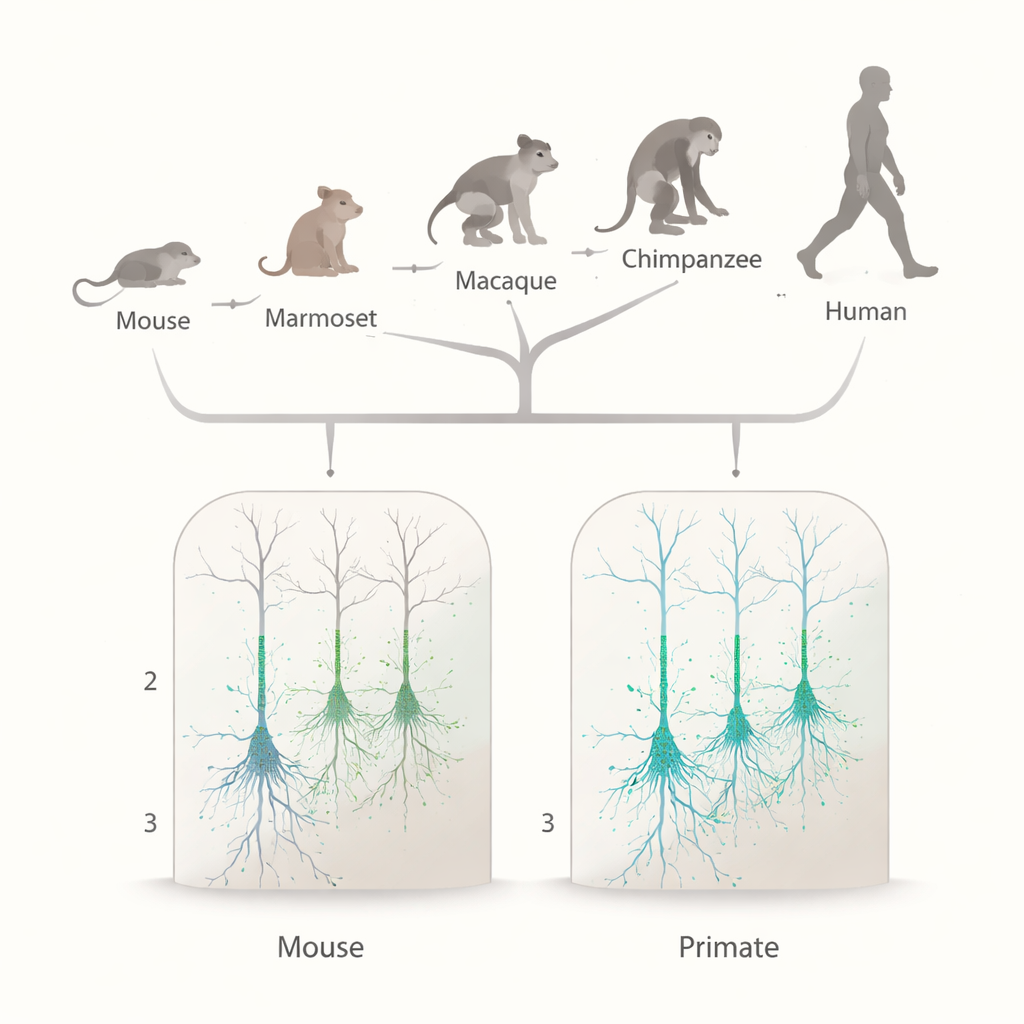
Scanning primate brains for the same electrical signature
To find out if this HCN enrichment is uniquely human or shared across primates, the team compared gene activity and electrical behavior in neurons from several species. Using single-nucleus RNA sequencing datasets, they first measured expression of the HCN1 gene and an auxiliary protein called TRIP8b (encoded by PEX5L) in excitatory neurons in the upper cortical layers of New World monkeys, Old World monkeys, great apes, and humans, and contrasted these with mouse data. In all primate species, HCN1 and TRIP8b were widely expressed in upper-layer excitatory neurons, at levels similar to a deep-layer neuron class already known to rely heavily on HCN channels. In mice, by contrast, HCN1 was much less common in these upper-layer cells. This pointed to a broad primate pattern rather than a human one-off.
Testing live neurons from monkeys
The authors then obtained living brain slices from two macaque species and squirrel monkeys and recorded from more than 500 upper-layer pyramidal neurons in temporal and motor cortex. They used clever current stimuli to probe whether the cells showed "membrane resonance"—a preference for oscillations in a particular frequency band—which is a hallmark of active HCN conductance. Many neurons in all three monkey species resonated above 2 Hz, especially in motor cortex, indicating strong HCN involvement. Other measurements, such as a characteristic "sag" in voltage during negative current injection and a faster cutoff for low-pass filtering, also supported widespread HCN activity. In pig-tailed macaque temporal cortex, HCN-related effects grew stronger in neurons located deeper within the upper layers, mirroring earlier findings in human middle temporal gyrus.
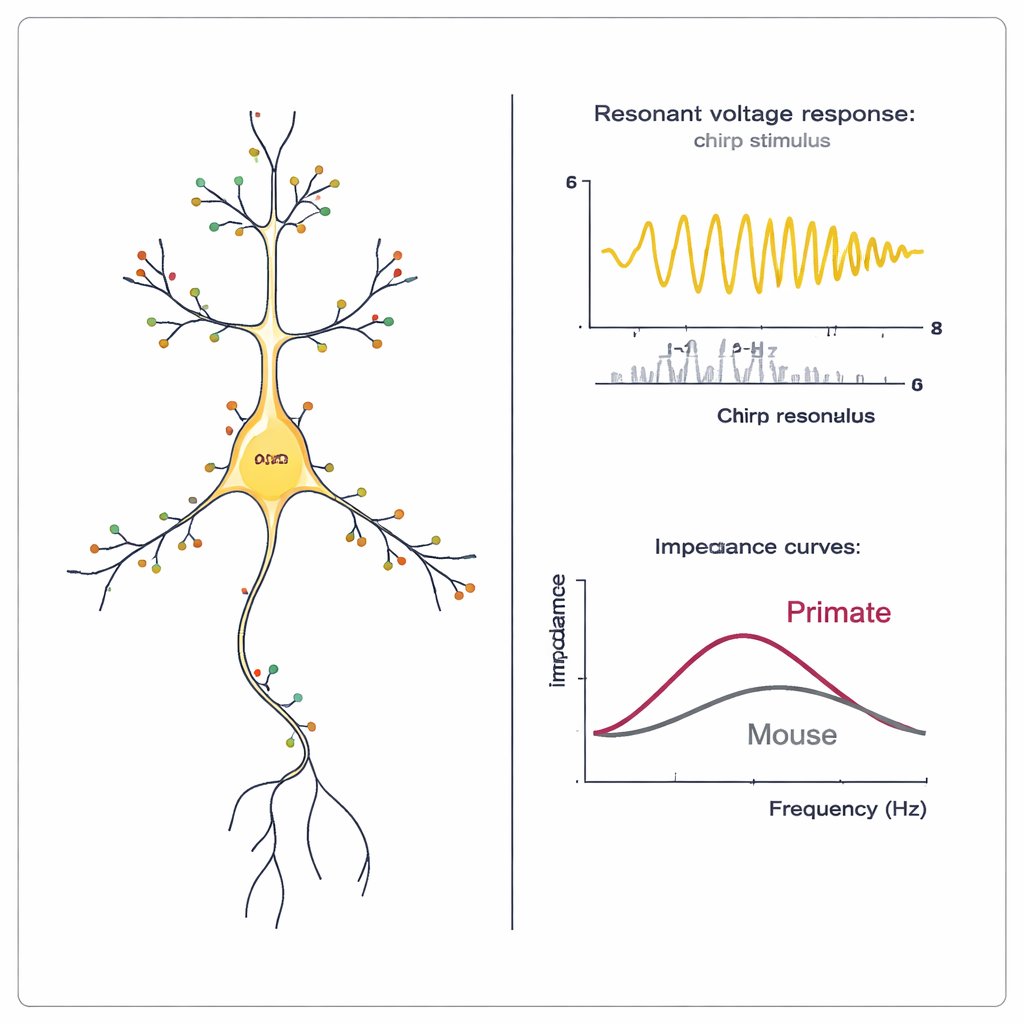
Blocking channels and comparing humans to macaques
To confirm that HCN channels actually cause these effects, the researchers applied a specific blocker, ZD7288, to macaque temporal cortex slices. When HCN channels were blocked, neurons became more electrically resistant, their resting voltage shifted more negative, and both sag and resonance essentially disappeared. The size of the change in resonance and sag tracked how much the input resistance changed, implying that neurons with the strongest HCN signatures also had the most HCN conductance. Finally, by combining electrical recordings and gene expression from the same cells using a method called Patch-seq, the team could align macaque and human neurons to matching transcriptomic types. In one major upper-layer type (L2/3 IT_1), HCN-related properties—including resonance and sag—increased with depth from the brain surface in both species and correlated with HCN1 expression. Interestingly, within this cell type, macaque neurons showed even stronger HCN-dependent behavior than their human counterparts, whereas a second upper-layer type (L2/3 IT_3) showed only mild differences between species.
What this means for how primate brains process information
Overall, the study shows that enhanced HCN channel expression and function in upper-layer pyramidal neurons is a conserved feature across primates, not a uniquely human adaptation. Compared with rodents, primates have thicker upper cortical layers and neurons with longer, more branched dendrites. Strong HCN conductance helps these large cells integrate inputs more evenly across their dendritic trees and tune them to slow delta/theta rhythms that dominate primate cortical activity. Subtle variations across cell types, brain areas, and species—such as the particularly strong HCN effects in one macaque neuron type—may provide additional flexibility for fine-tuning cognition. But the basic message is clear: the electrical specializations once thought to distinguish human cortical neurons instead appear to be a shared primate strategy for handling complex, rhythmic information flow.
Citation: Radaelli, C., Schmitz, M., Liu, XP. et al. HCN channels reveal conserved and divergent physiology in supragranular pyramidal neurons in primate species. Commun Biol 9, 279 (2026). https://doi.org/10.1038/s42003-026-09558-2
Keywords: HCN channels, primate cortex, pyramidal neurons, delta theta rhythms, Patch-seq