Clear Sky Science · en
Radionuclide-stimulated dynamic therapy induces complementary immunogenic necroptosis and apoptosis cancer cell death pathways
Turning Cancer’s Tricks Against Itself
Cancer is notorious for dodging our best treatments, especially in aggressive breast tumors that spread throughout the body. This study explores an inventive approach called radionuclide-stimulated dynamic therapy (RaST), which uses medical imaging tracers and light-activated nanoparticles to kill cancer cells in ways that also wake up the immune system. For readers, this work matters because it suggests how existing imaging tools and smart materials might be repurposed to create longer‑lasting, immune‑driven cancer control rather than temporary tumor shrinkage.
A New Way to Light Up Tumors From Within
Traditional light-based cancer treatments work only where an external beam can reach, such as skin or surface tumors. RaST gets around this limitation by using a radioactive sugar, similar to the tracer already used in PET scans, that naturally gathers in fast-growing cancer cells. As this tracer travels through the tumor, it emits a faint blue glow known as Cerenkov radiation. The researchers designed tiny titanium dioxide nanoparticles coated with a cancer-targeting protein so they, too, home in on the tumor. When the tracer and nanoparticles meet, the internal light switches on the particles, which in turn generate bursts of reactive oxygen species—highly reactive molecules that damage cancer cells from within.
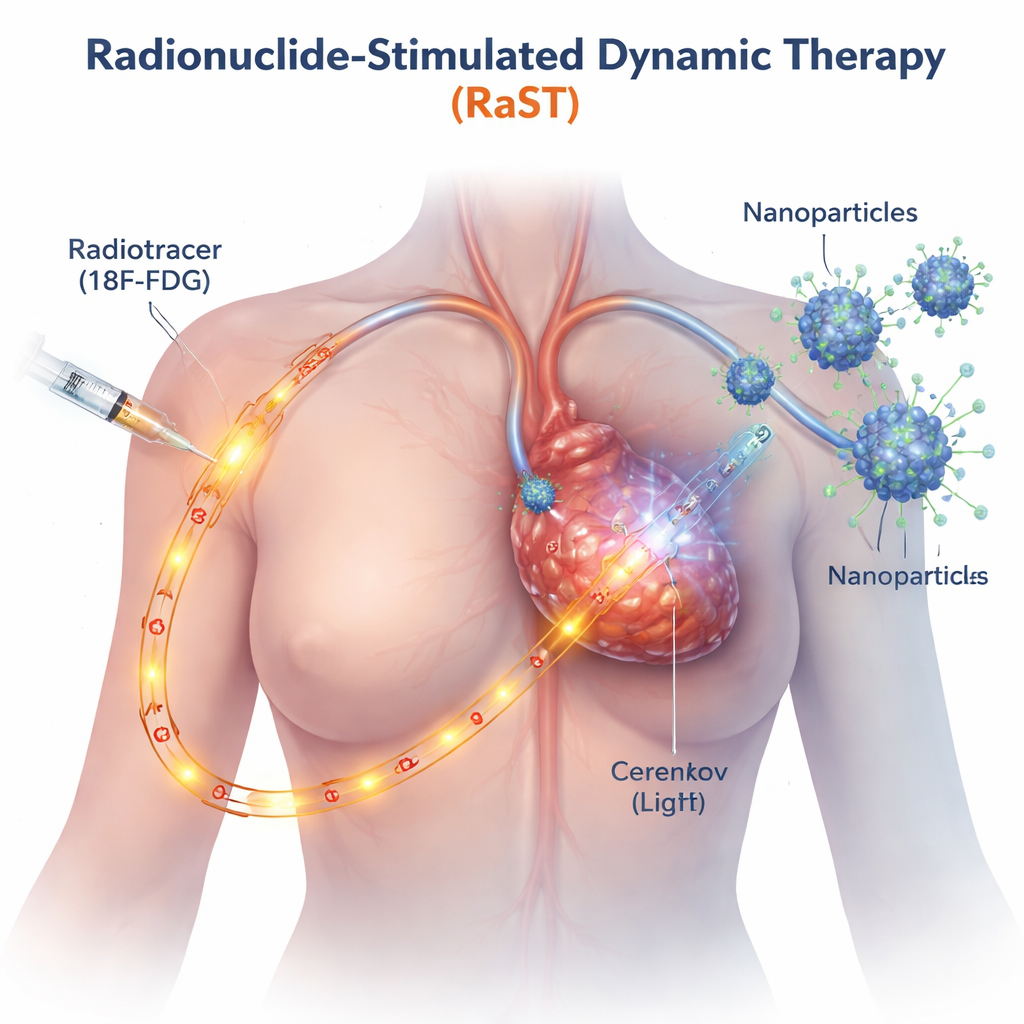
Triggering Two Distinct Paths to Cell Death
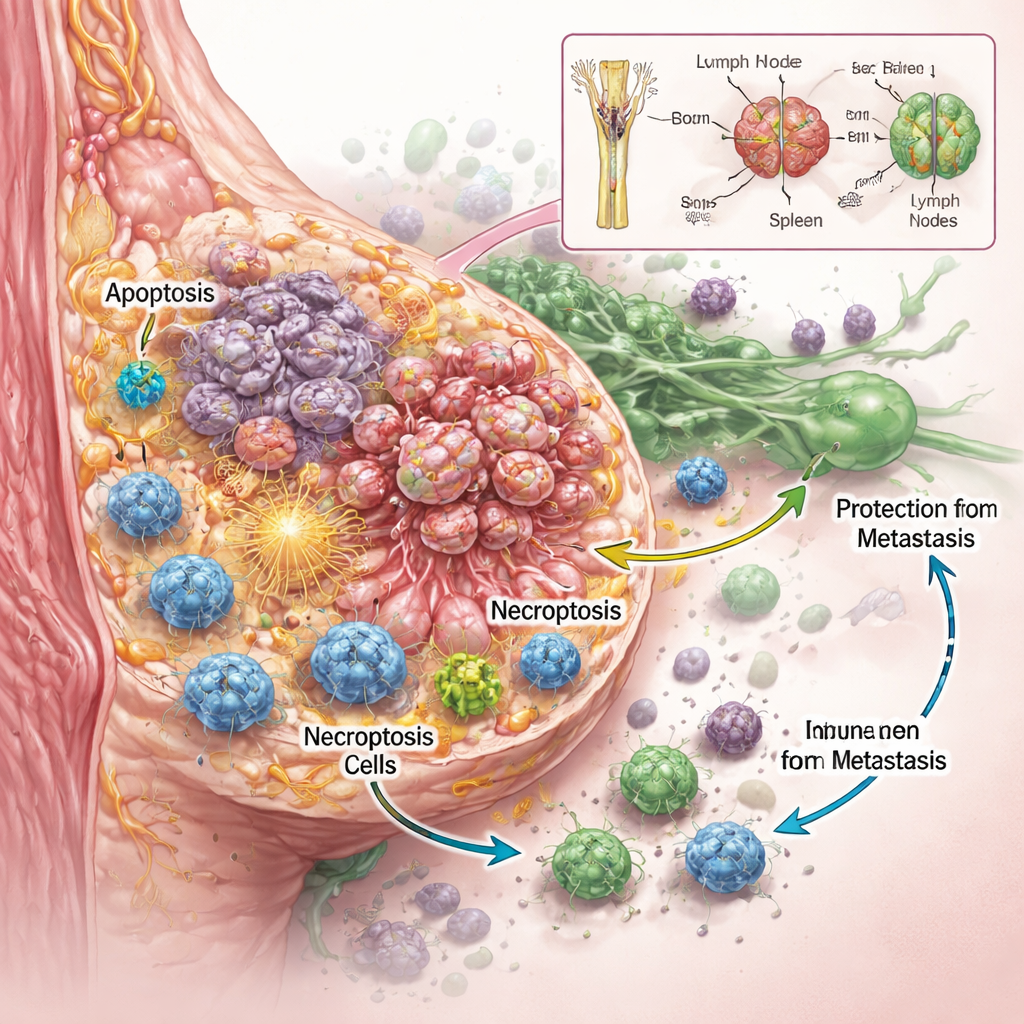
The team wanted to know not just whether RaST killed cancer cells, but how. Using several aggressive breast cancer cell lines in the lab, they found that carefully tuned doses of radioactive tracer and nanoparticles worked together to damage tumor cells more effectively than either component alone, even in types known to resist standard chemotherapy. Detailed imaging and protein measurements revealed that RaST activated two regulated cell death programs at once. One was apoptosis, often described as cellular “suicide,” where cells dismantle themselves in an orderly fashion. The other was necroptosis, a more explosive form of cell death in which cells swell, rupture, and spill molecular distress signals into their surroundings. These signals, known as damage-associated molecules and inflammatory cytokines, can serve as flares that attract and activate immune cells.
Enlisting the Immune System for Lasting Control
To see whether this inflammatory style of tumor killing mattered in living organisms, the researchers tested RaST in mouse models of breast cancer. In mice lacking key immune defenses, RaST did little to stop tumor growth or lung spread, despite generating plenty of local cell death. But in mice with intact immune systems, the same treatment produced partial and complete tumor responses and prevented metastasis to the lungs. Tumors exposed to RaST became packed with immune cells—especially myeloid cells such as macrophages and dendritic cells—that are skilled at engulfing dead material and presenting tumor fragments to T cells. Blood tests showed waves of immune-stimulating and immune-regulating signals over time, suggesting that RaST first fires up an attack on the cancer and later shifts toward a more balanced state as tumors shrink or stabilize.
Hunting Hidden Remnants and Guarding Against Relapse
Even in mice where tumors seemed completely cured by RaST, a highly sensitive imaging dye later revealed tiny pockets of residual cancer cells tucked inside lymph nodes. Remarkably, these animals still did not develop new tumors or distant metastases over many months. Chemical analyses showed that the titanium dioxide nanoparticles, initially abundant in the bones and liver, gradually redistributed to the spleen and lymph nodes—precisely the hubs where immune cells patrol. This slow migration likely keeps a local source of reactive oxygen available to immune cells, helping them recognize and control these hidden cancer remnants before they can regrow or spread.
What This Could Mean for Future Cancer Care
In simple terms, this work shows that RaST is more than just another way to poison tumor cells. By combining radioactive tracers, light-responsive nanoparticles, and the body’s own defenses, it pushes cancer cells into two forms of programmed death that loudly alert the immune system. In immune-competent animals, this leads to lasting control of aggressive breast tumors and blocks metastasis, even when a few cancer cells linger in lymph nodes. While much remains to be done before this approach can be tested widely in people, the study points toward future treatments that do not simply aim to shrink tumors, but to reshape the relationship between cancer, cell death, and immunity so the body can help keep the disease in check over the long term.
Citation: Egbulefu, C., Black, K., Su, X. et al. Radionuclide-stimulated dynamic therapy induces complementary immunogenic necroptosis and apoptosis cancer cell death pathways. Commun Biol 9, 275 (2026). https://doi.org/10.1038/s42003-026-09555-5
Keywords: breast cancer, nanoparticles, immunotherapy, radionuclide therapy, necroptosis