Clear Sky Science · en
Glycophagy is an ancient bilaterian pathway supporting metabolic adaptation through STBD1 structural evolution
How oysters reveal hidden energy tricks
Animals survive tough times by tapping into stored fuel, but they don’t all rely on the same kind of “battery.” This study looks at Pacific oysters and shows that, unlike many other animals that lean on fat, oysters depend heavily on stored sugar, in the form of glycogen. The work uncovers a little-known cellular recycling route—called glycophagy—that helps oysters rapidly drain and refill their sugar reserves, and traces how this system evolved across the animal kingdom.
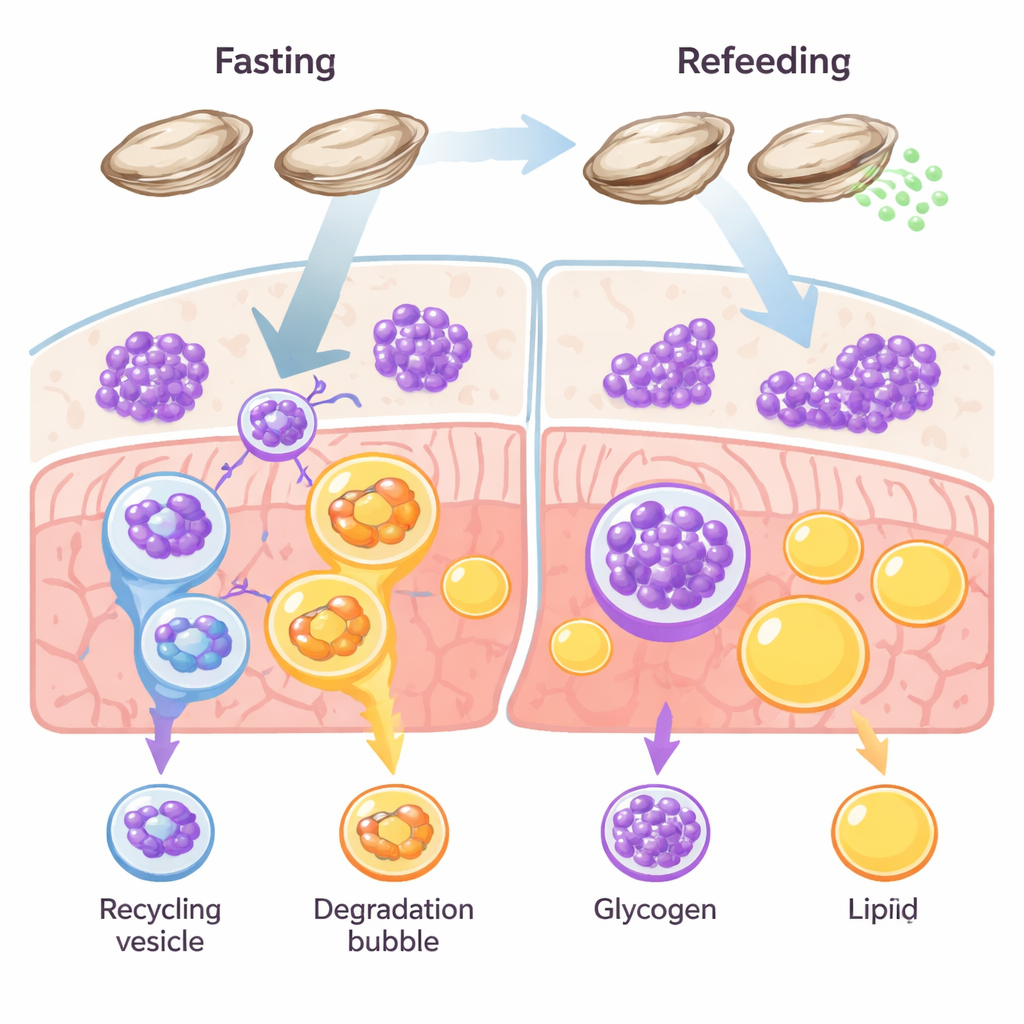
A sugar-first survival strategy
Most animals juggle several energy stores, mainly fat and glycogen. In vertebrates such as fish and mammals, fat usually dominates as the long-term reserve. Oysters break this rule. Their tissues are packed with glycogen, and earlier research suggested that these shellfish burn through sugar, rather than fat, when food becomes scarce or when they are making eggs and sperm. The authors set out to test whether glycophagy—a targeted breakdown of glycogen inside cells—acts as a primary survival pathway in oysters during starvation and recovery.
Watching cells switch from storage to spending
The team starved Pacific oysters for two weeks and then refed them, tracking how their tissues responded over time. During fasting, microscopic stains showed a surge in autophagy markers—cellular “cleanup” signals—exactly where glycogen was stored, while the glycogen itself dwindled. Fat stores, in contrast, barely changed. When the oysters were fed again, glycogen quickly returned and autophagy signals fell. At the same time, levels of a key receptor protein called STBD1, which specifically connects glycogen to the autophagy machinery, rose and fell in step with glycogen use. Another receptor linked to fat recycling, p62, stayed largely flat. Together, these patterns point to a dedicated, sugar-focused recycling route: glycophagy, not fat-burning lipophagy, is doing the heavy lifting in oyster energy balance.
The molecular hook that grabs glycogen
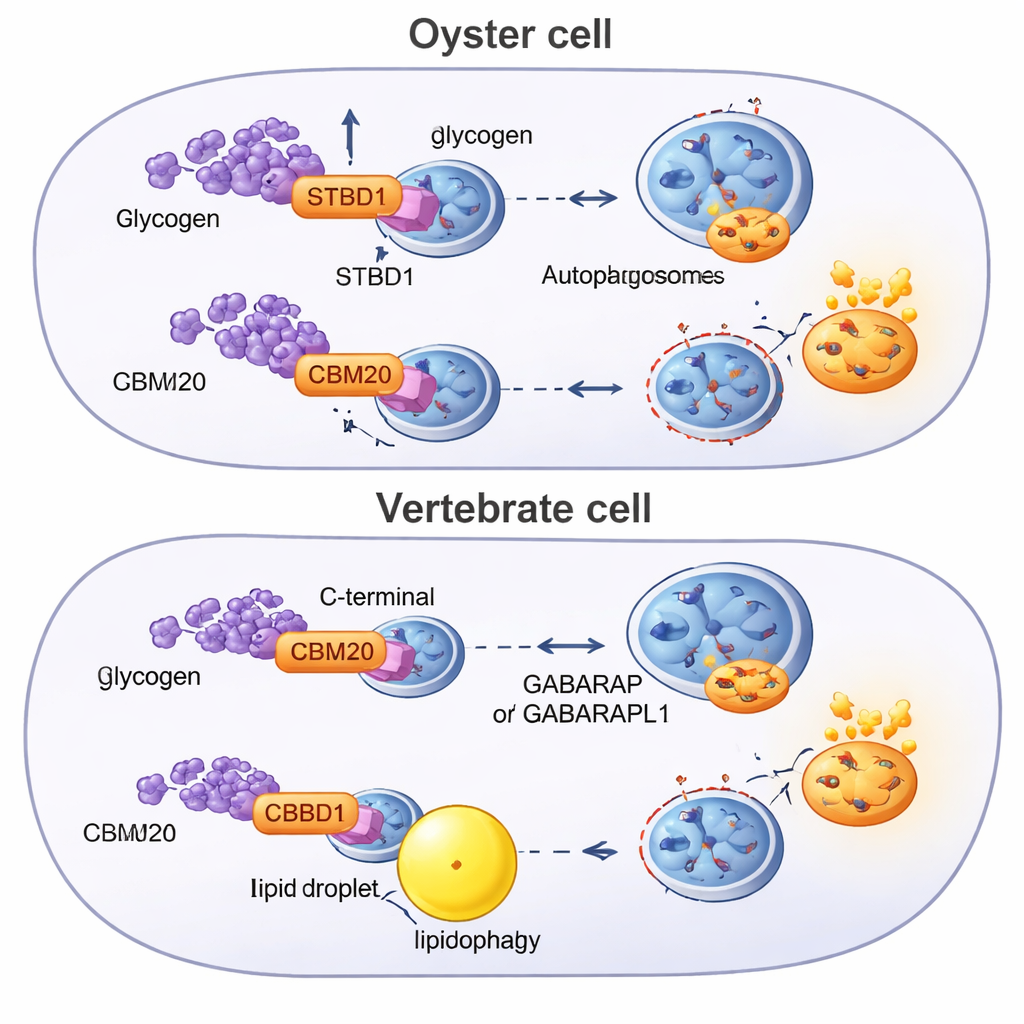
Glycophagy depends on STBD1 acting as a molecular hook: it binds glycogen and hands it over to small adapter proteins that build recycling vesicles. In oysters, STBD1 turned out to be wired differently from its vertebrate counterpart. The oyster protein carries its glycogen-grabbing region, known as the CBM20 domain, at the front end (N-terminus), whereas vertebrate STBD1 places this same module at the back (C-terminus) and includes an extra greasy tail that anchors it to cellular membranes. Computer models and simulations suggested that the oyster’s N-terminal layout gives CBM20 a stronger, more versatile grip on branched sugar chains. Lab experiments backed this up: purified oyster STBD1 bound glycogen more tightly than fish or mouse STBD1, and when all versions were expressed in human cells, the oyster protein drove a sharper drop in glycogen during induced autophagy.
Rewiring an ancient pathway across animals
To understand where this system came from, the researchers scanned genomes from dozens of animals for CBM20-bearing proteins and built evolutionary trees. They found that STBD1 is a bilaterian invention—appearing in the common ancestor of animals with two-sided body plans—but its structure has been reshuffled in different lineages. Oysters and other lophotrochozoans tend to keep the ancestral N-terminal CBM20 design, sometimes even duplicating the sugar-binding module. Chordates, the group that includes vertebrates, show a distinct version in which CBM20 has been moved to the protein’s tail. This rearrangement correlates with weaker glycogen binding and with a metabolic strategy that leans more heavily on fat breakdown through lipophagy, supported by other autophagy receptors and adapter proteins that oysters lack.
What this means for life’s energy choices
For non-specialists, the take-home message is that animals have evolved more than one way to ride out energy shortages. Oysters showcase an old, sugar-centered strategy: a high-affinity version of STBD1 grabs glycogen quickly and feeds it into cellular recycling units, making glycophagy a main power source during stress. Vertebrates, by contrast, seem to have traded some of that strong sugar grip for a more balanced or fat-skewed approach, supported by different protein partners and domain layouts. By tying detailed protein structure to whole-organism fuel choices, this study illustrates how small molecular “rewirings” can help different branches of the animal tree adapt to their own environments and lifestyles.
Citation: Ren, L., Bai, Y., Shi, C. et al. Glycophagy is an ancient bilaterian pathway supporting metabolic adaptation through STBD1 structural evolution. Commun Biol 9, 268 (2026). https://doi.org/10.1038/s42003-026-09546-6
Keywords: glycophagy, glycogen metabolism, oyster biology, autophagy, evolution of metabolism