Clear Sky Science · en
Epigenetic and metabolic rewiring in metastatic pheochromocytomas and paragangliomas driven by SDHB mutations
Why these rare tumors matter
Pheochromocytomas and paragangliomas are rare tumors that grow from hormone‑producing nerve cells, often near the adrenal glands. Most stay slow‑growing and can be cured with surgery, but about one in five eventually spreads to other organs, becoming life‑threatening. This study asks a simple but crucial question: what makes some of these tumors turn dangerous? By looking deep into how these cancers switch their genes on and off and how they fuel themselves, the researchers uncover a hidden weakness that might one day be targeted with new treatments.
From quiet growth to deadly spread
The team focused on tumors carrying changes in a gene called SDHB. SDHB helps mitochondria—the cell’s power plants—run part of the Krebs cycle, a core energy‑producing pathway. When SDHB is damaged, a chemical called succinate builds up, and cells start behaving as if they live in low oxygen, even when they do not. The researchers studied tissue from 34 patients, comparing tumors that stayed localized with those that had already spread. Using a high‑resolution DNA “methylation” map, which shows how chemical tags turn genes up or down, they found that metastatic tumors had a stronger pattern of gene silencing than benign ones, especially when SDHB was mutated.
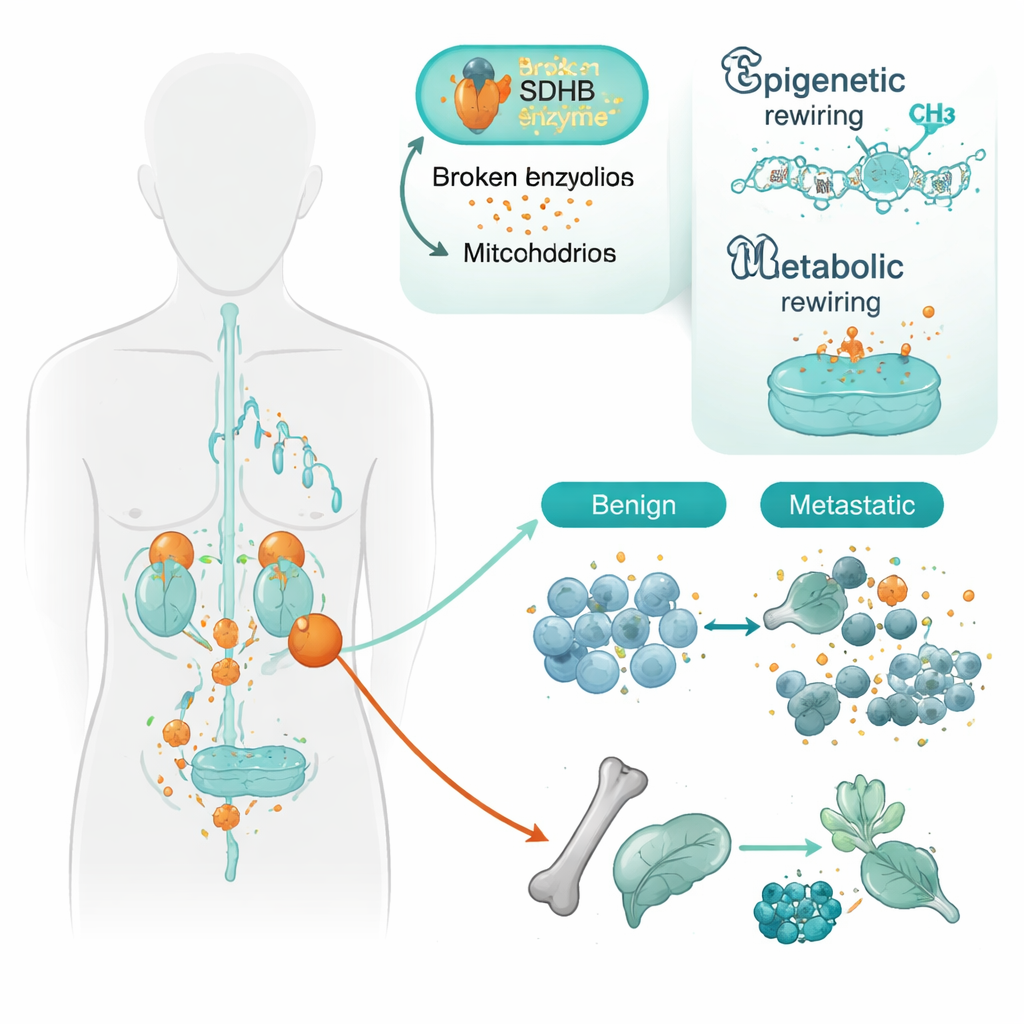
Reprogramming cell identity
Many of the genes silenced in aggressive tumors were those that help nerve‑like cells mature and maintain their specialized identity. These include families of genes that guide how cells stick together, organize their DNA, and decide what type of cell to become. One stand‑out example was a cell‑adhesion gene called PCDHGC3. Even in non‑spreading SDHB‑mutant tumors, this gene’s control region was already partly switched off; in metastatic tumors, the silencing was even stronger. Another gene, SATB2, involved in organizing large stretches of DNA, was silenced only in metastatic tumors, regardless of SDHB status. Together, these patterns suggest that SDHB loss “primes” cells for a less mature, more mobile state, and additional epigenetic changes then help them break away and spread.
Rewiring how tumors eat sugar
Surprisingly, not all changes involved shutting genes down. A smaller set of genes became less methylated and more active, and these were heavily involved in moving sugars into cells. Among them, the fructose transporter gene SLC2A5 stood out. Its product, known as GLUT5, shuttles fructose—a sugar common in fruit and many processed foods—into cells. In lab models of tumor‑like cells exposed to low oxygen, SLC2A5 steadily switched on, while other sugar transporters responded less consistently. The authors then created patient‑derived cell cultures from PPGL tumors and showed that when glucose was scarce, adding fructose helped these cells keep dividing, especially under low‑oxygen conditions that mimic the tumor environment.
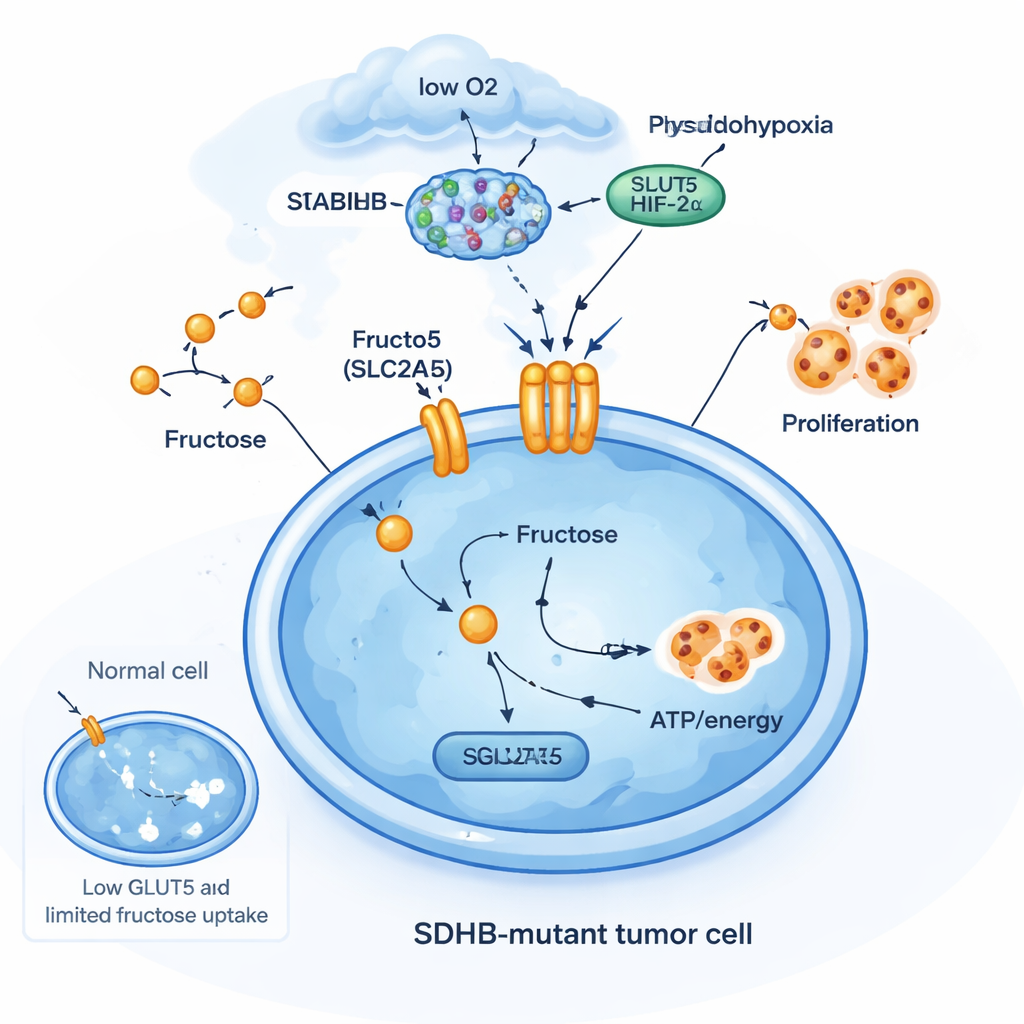
A fructose‑powered survival trick
To understand why SDHB mutations matter here, the team edited several cell types to remove SDHB. In chromaffin‑like cells from the adrenal gland and in a kidney cancer cell line with constant activation of the oxygen‑sensing factor HIF2α, loss of SDHB boosted SLC2A5 levels. But in more common cancer cell lines that lacked these neuroendocrine or HIF2α features, the same SDHB knockout did not turn on SLC2A5. This shows that the fructose transporter is not part of a generic stress response; it is a highly specific adaptation in certain cell types that already live in a “pseudo‑hypoxic” state. In these cells, SDHB loss, succinate buildup, and HIF2α activity cooperate to open a new metabolic door: fructose uptake to fuel growth when oxygen and glucose are limited.
What this means for patients
Put simply, the study reveals a double shift in SDHB‑mutant metastatic PPGLs. First, their DNA control system is rewired to push cells into a less specialized, more plastic state that is prone to invasion. Second, their energy system is rewired to tap into fructose as an alternative fuel in harsh, low‑oxygen, low‑glucose niches. By identifying the fructose transporter SLC2A5/GLUT5 as a key player in this process, the work points to a possible Achilles’ heel: blocking fructose uptake or metabolism could selectively starve these tumors while sparing most normal tissues. Although such therapies are not yet available, mapping this epigenetic and metabolic landscape is a critical step toward more precise, metabolism‑focused treatments for patients with high‑risk SDHB‑mutant PPGLs.
Citation: Cubiella, T., Alba-Linares, J.J., San-Juan-Guardado, J. et al. Epigenetic and metabolic rewiring in metastatic pheochromocytomas and paragangliomas driven by SDHB mutations. Commun Biol 9, 266 (2026). https://doi.org/10.1038/s42003-026-09543-9
Keywords: pheochromocytoma, paraganglioma, SDHB mutation, epigenetics, fructose metabolism