Clear Sky Science · en
Multi-omic analysis of human PHACTR1 signaling networks
Why this blood vessel gene matters to you
Heart attacks, strokes, and high blood pressure all trace back to the health of our blood vessels. A single gene called PHACTR1 has repeatedly turned up in large genetic studies of these conditions, but scientists have not understood why. This study used a sweeping, “all-levels” look inside human cells to map what PHACTR1 actually does, revealing how it can influence cell growth, energy use, and iron handling—processes that ultimately shape the health of arteries.
A big-picture scan inside cells
Instead of focusing on one molecule at a time, the researchers used a strategy called multi-omics—simultaneously measuring thousands of RNAs, proteins, small metabolites, and fats (lipids) in cells. They engineered human cells to either make extra PHACTR1 or have the gene dialed down, mimicking natural genetic differences seen in people. By comparing these altered cells with normal controls across four molecular layers, then feeding the data into sophisticated pathway software, they built a global map of how changing PHACTR1 ripples through the cell’s inner machinery. 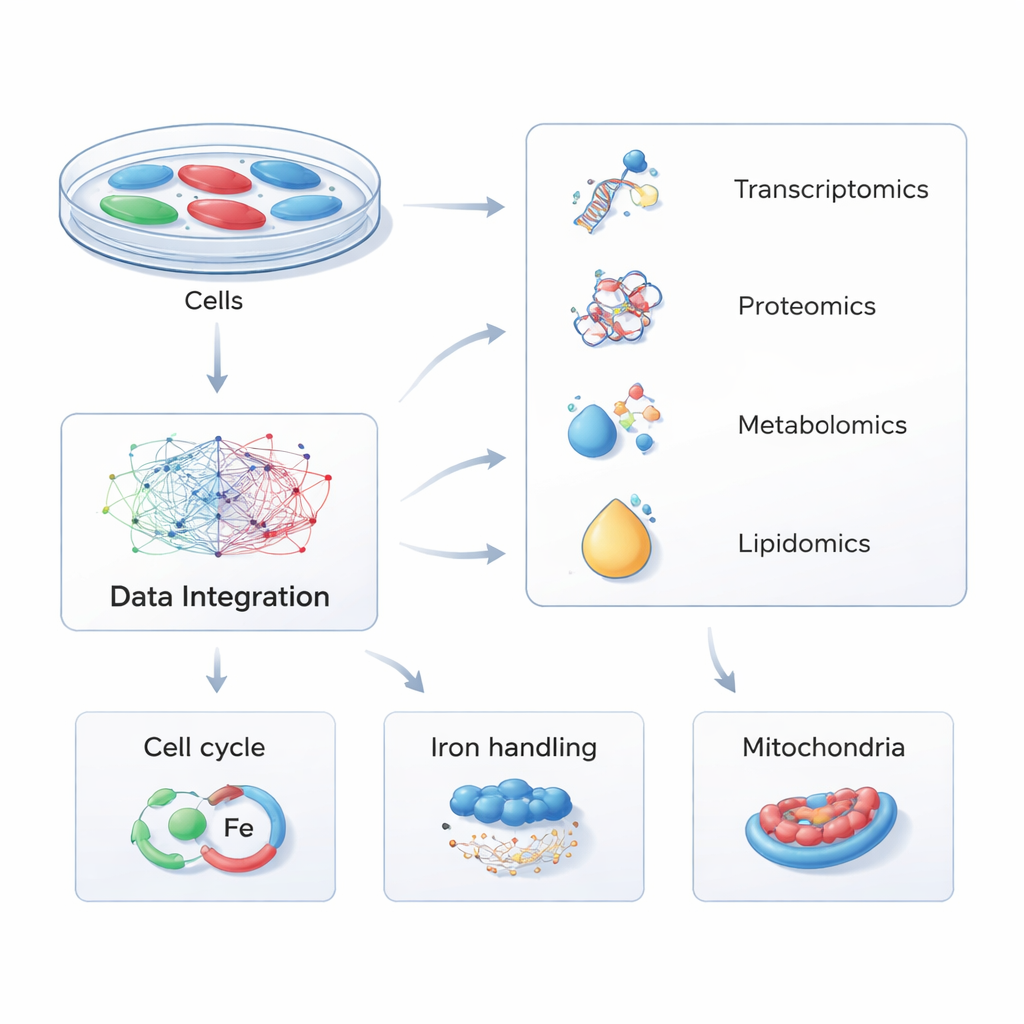
Beyond the cell skeleton: controlling growth cycles
PHACTR1 was known mainly for helping organize the cell’s internal scaffolding of actin fibers. The new data showed it also has a strong hand in controlling the cell cycle—the choreography by which cells copy their DNA and divide. Cells with extra PHACTR1 shifted out of the resting G1 phase and piled up in DNA-copying and pre-division phases, with fewer cells successfully entering full mitosis. Key regulators such as Cyclin B1, Cdt1, and other cell-cycle proteins were altered. When the team repeated the experiments in primary human endothelial cells—the cells that line blood vessels—they again saw changes in core cell-cycle regulators. This suggests PHACTR1 helps decide when vascular cells rest, divide, or stall, a balance that is crucial in both repairing vessel walls and forming dangerous tissue overgrowths.
Managing iron and guarding against damage
The multi-omics integration also pointed to iron-related pathways and a specialized form of iron-driven cell death called ferroptosis. Although the cells were not actively dying by ferroptosis, increasing PHACTR1 reduced levels of major iron-handling proteins, including ferritin heavy chain (the main iron-storage shell) and heme oxygenase 1 (which recycles iron from heme). In primary endothelial cells, PHACTR1 similarly altered these proteins and key defenders against oxidative damage. Because mismanaged iron can fuel inflammation, oxidative stress, and unstable artery plaques, these results suggest that PHACTR1 may tune how vascular cells store and detoxify iron, potentially affecting who is more vulnerable to certain vessel diseases.
Mitochondria, energy, and artery health
Another surprise was PHACTR1’s role in mitochondria, the cell’s power plants. The team found PHACTR1 protein within isolated mitochondria and saw that changing its levels reshaped the mitochondrial network. When PHACTR1 was high, mitochondria were more elongated, and a protein called Drp1 was chemically modified at a site that reduces mitochondrial splitting. Levels of a scaffold protein, AKAP1, which helps organize signaling on the mitochondrial surface, rose and fell in step with PHACTR1. These structural shifts matched changes in how cells made energy: high PHACTR1 reduced mitochondrial ATP production and was linked to a buildup of certain fatty-acid–derived molecules that signal sluggish fat burning, while low PHACTR1 pushed cells to rely more on sugar-burning glycolysis. Analyses of human artery samples showed that PHACTR1 and AKAP1 tend to be switched on together, tying this mitochondrial control circuit directly to real blood vessels. 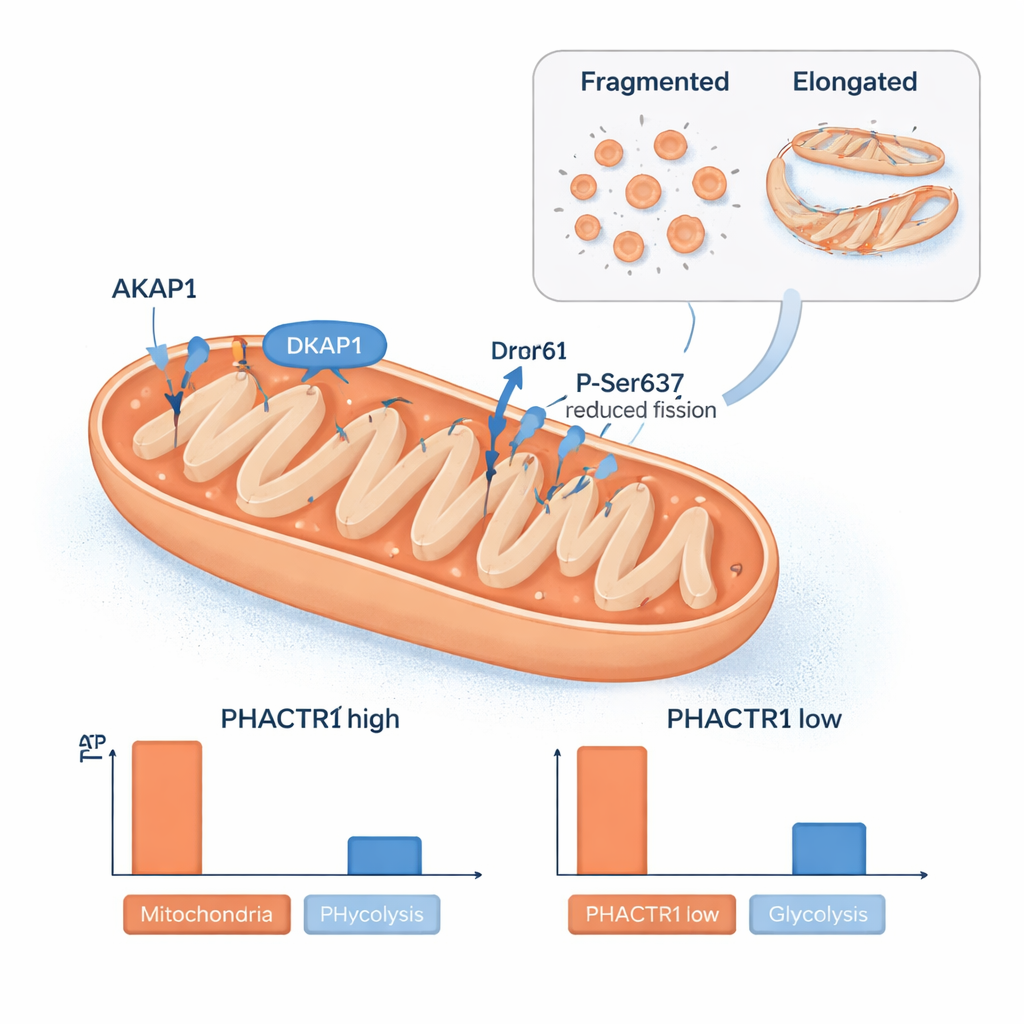
What it all means for blood vessels
Taken together, this work shows that PHACTR1 is not a single-purpose gene but a master coordinator touching how vascular cells divide, store iron, and fuel themselves. By mapping these connections across thousands of molecules, the study helps explain why natural differences in PHACTR1 are linked to such a wide range of vessel-related diseases—from coronary artery disease to spontaneous artery tears and migraine. For non-specialists, the message is that a gene flagged by human genetics is now being traced to concrete cellular behaviors that influence plaque growth, vessel stability, and energy balance. In the long run, understanding these PHACTR1-controlled networks may open the door to more precise therapies that nudge cell cycles, iron handling, or mitochondrial function back toward a healthier state in people at risk of cardiovascular disease.
Citation: Wolhuter, K., Ma, L., Bryce, N.S. et al. Multi-omic analysis of human PHACTR1 signaling networks. Commun Biol 9, 265 (2026). https://doi.org/10.1038/s42003-026-09542-w
Keywords: PHACTR1, vascular disease, multi-omics, mitochondria, iron metabolism