Clear Sky Science · en
Targeting PAK4 promotes Gemcitabine-induced pyroptosis in pancreatic cancer via NLRP1/caspase-3/GSDME axis
Why killing cancer cells the “fiery” way could matter
Pancreatic cancer is among the deadliest cancers, and even our best chemotherapy drugs often stop working as tumors become resistant. This study explores a surprising twist: the same drug, gemcitabine, can kill cancer cells in a more explosive, inflammatory way called pyroptosis—if a key molecular “brake” called PAK4 is switched off. Understanding and targeting this brake could make existing treatments work better for many patients.
A stubborn cancer that shrugs off treatment
Pancreatic ductal adenocarcinoma is usually diagnosed late and responds poorly to therapy, so gemcitabine remains a cornerstone drug despite its limits. Gemcitabine typically kills cells through apoptosis, a tidy, quiet form of programmed cell death. But more than half of the pancreatic tumors examined in this study produced high levels of a protein called GSDME, which can flip this quiet death into pyroptosis—a rapid, “bursting” form of cell death that damages the cell membrane and releases inflammatory signals. This raised a key question: if tumors can undergo pyroptosis, why do so many still resist gemcitabine?
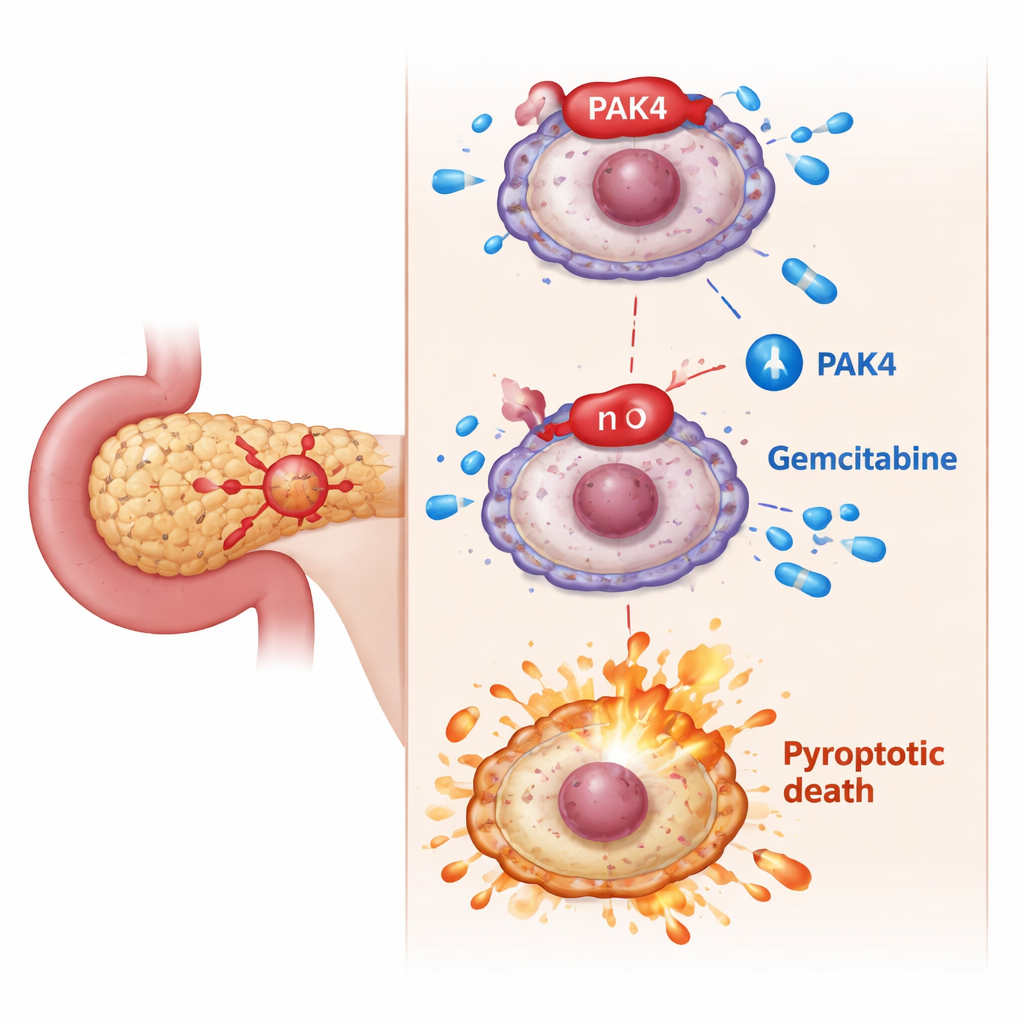
The hidden brake: PAK4 blocks fiery cell death
The researchers homed in on PAK4, a growth-promoting protein known to be overactive in pancreatic cancer and linked to poor response to gemcitabine. In cell experiments, cancer cells with high PAK4 levels were harder to kill with gemcitabine. When the team reduced PAK4, gemcitabine suddenly became much more lethal: cells showed classic pyroptotic features—swelling, balloon-like bubbles, ruptured membranes, and higher release of inner-cell contents. At the molecular level, this shift depended on GSDME being cut into an active fragment by another protein called caspase-3. Blocking caspase-3 stopped GSDME from triggering pyroptosis, confirming that PAK4 normally acts to hold this death pathway in check.
How PAK4 dismantles the cell’s death alarm
Diving deeper, the scientists found that PAK4 does not attack GSDME directly. Instead, it sabotages an upstream “alarm sensor” called NLRP1. In patients, higher NLRP1 levels were linked to better survival, suggesting it helps the body keep tumors in check. In cancer cells, when NLRP1 was removed, gemcitabine-induced cell death and GSDME activation dropped sharply, showing that NLRP1 feeds into the pyroptosis machinery. PAK4 and NLRP1 levels were inversely related in tumor samples: where PAK4 was high, NLRP1 was low. The study showed that active PAK4 works through another protein, the E3 ligase Mdm2, to tag NLRP1 for destruction by the cell’s waste-disposal system. By marking NLRP1 in this way, PAK4 prevents activation of a chain reaction—through caspase-9 and caspase-3—that would normally culminate in GSDME-driven pyroptosis.
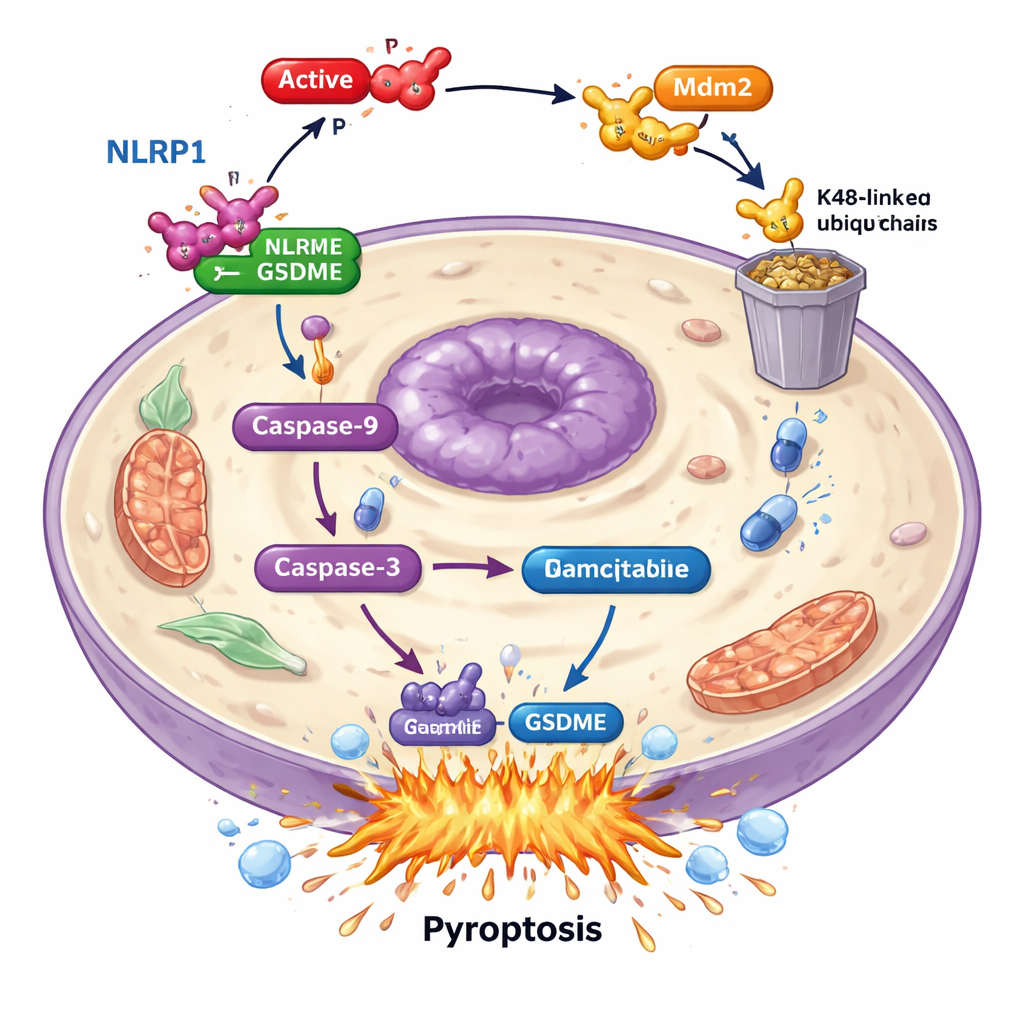
Turning off PAK4 to supercharge chemotherapy
Because PAK4 acts as a brake on pyroptosis, the team tested whether blocking its activity could reawaken gemcitabine’s killing power. They used a small-molecule PAK4 inhibitor called PF-3758309. On its own, this inhibitor already pushed pancreatic cancer cells toward pyroptosis, increasing caspase-3 activation, GSDME cleavage, and cell membrane rupture. When combined with gemcitabine, the effect was even stronger: more cells died through pyroptosis, and molecular signs of the death pathway were amplified. In mouse models carrying human pancreatic tumors, either reducing PAK4 or treating with the PAK4 inhibitor made gemcitabine shrink tumors more effectively and boosted pyroptotic markers inside the cancers.
What this means for patients and future treatments
In plain terms, this work reveals a molecular lock—PAK4—that keeps pancreatic cancer cells from dying in a particularly destructive way under chemotherapy. By disabling PAK4, doctors might be able to push gemcitabine-treated tumors into pyroptosis, making the drug more effective and potentially stirring up the immune system against the cancer. While more work in patient-derived models and clinical trials is needed, the study proposes a clear roadmap: target the PAK4–Mdm2–NLRP1–GSDME pathway to convert resistant tumors into ones that respond, using drugs we already have in smarter combinations.
Citation: Lu, T., Song, Y., Liang, K. et al. Targeting PAK4 promotes Gemcitabine-induced pyroptosis in pancreatic cancer via NLRP1/caspase-3/GSDME axis. Commun Biol 9, 260 (2026). https://doi.org/10.1038/s42003-026-09538-6
Keywords: pancreatic cancer, gemcitabine resistance, pyroptosis, PAK4, targeted therapy