Clear Sky Science · en
WDR62 is required for proper proliferation and early differentiation of skeletal myoblasts
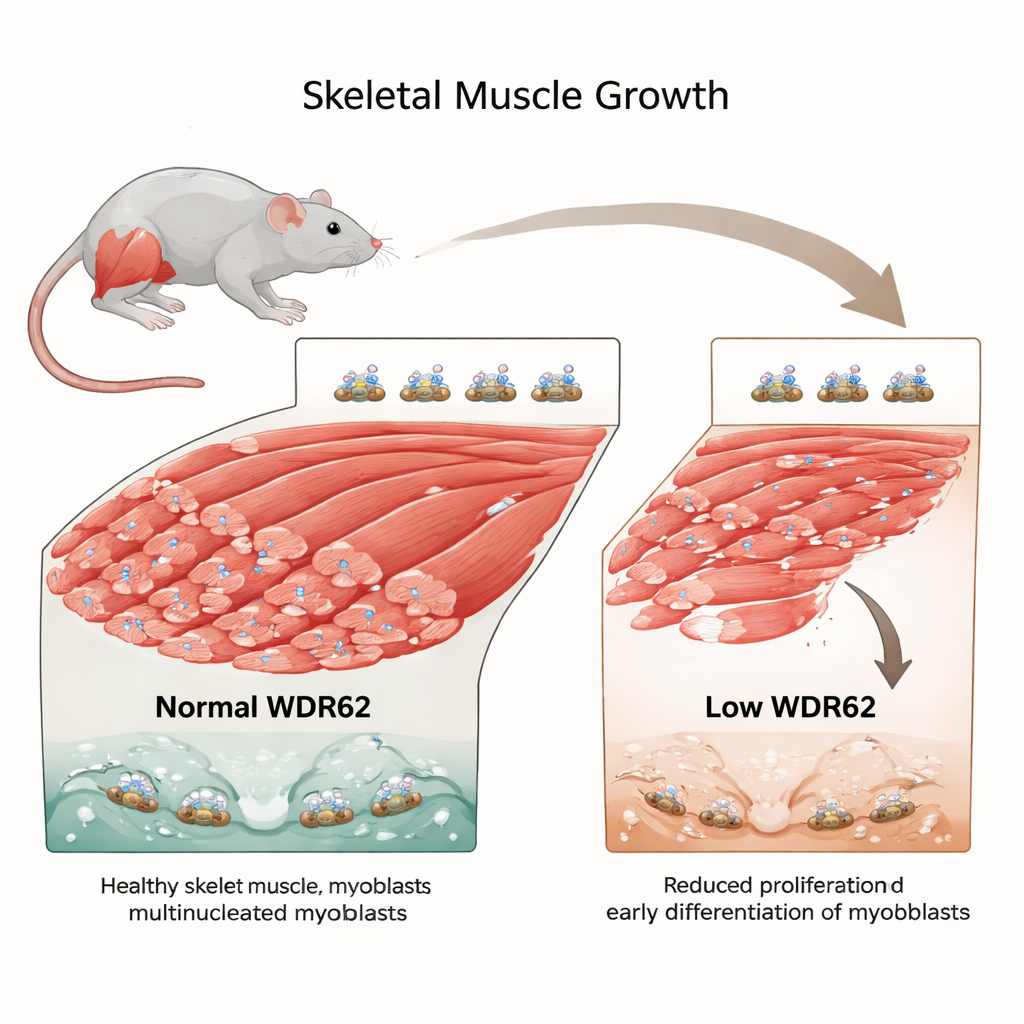
Why muscle growth needs careful timing
Our muscles don’t just get bigger because we exercise; they grow and repair themselves through a precise dance of stem cells that must divide, wait, and then finally turn into working muscle. This paper unpacks the role of an often-overlooked protein, WDR62, in keeping that dance on beat. When WDR62 is missing or faulty, muscles end up smaller, and the cells that should patiently multiply rush too quickly into becoming mature fibers, leaving too few cells in reserve for long-term strength and repair.
A hidden organizer inside muscle stem cells
WDR62 is a protein that sits at tiny structures in cells called centrosomes, which help arrange the scaffolding that pulls chromosomes apart when cells divide. WDR62 has already been linked to brain development, where its loss can cause a smaller brain because early stem cells stop dividing too soon. The authors wondered whether something similar might be happening in skeletal muscle, where specialized stem cells—called satellite cells—wake up after injury, divide several times as myoblasts, and then fuse together to build or repair muscle fibers. If WDR62 helps these cells divide properly in the brain, could it also be crucial for building healthy muscle?
Smaller muscles and rushed development
To test this, the team studied mice engineered to lack working WDR62. Even without any injury, two leg muscles important for movement—the tibialis anterior and extensor digitorum longus—were noticeably smaller than in normal mice. Under the microscope, the basic muscle architecture looked normal, but there were slightly fewer fibers overall, suggesting that not enough myoblasts had been available to build a full-sized muscle. When the researchers injured the leg muscle with a standard toxin that forces it to regenerate, the overall repair sequence looked broadly normal. However, around one week after injury, the WDR62-deficient mice had more immature, newly formed fibers than their healthy counterparts, hinting that their myoblasts were differentiating and fusing earlier than they should.
Conserved effects from flies to cultured cells
To see if this behavior was fundamental and not just a mouse oddity, the authors turned to fruit flies, which also have muscle precursor cells in their developing wing. Reducing Wdr62 in these fly myoblasts led to fewer precursor cells and more asymmetric divisions—cell splits that produce one stem-like cell and one more committed daughter. Too many of these asymmetric divisions drain the stem-cell pool, just as seen in the mouse brain. In parallel, the team used a classic mouse muscle cell line, C2C12, grown in dishes. When they knocked down WDR62 in these cells, fewer cells entered the cell cycle and divided, but those that did began to fuse into early muscle fibers slightly ahead of schedule, again pointing to a trade-off: reduced proliferation paired with premature differentiation.
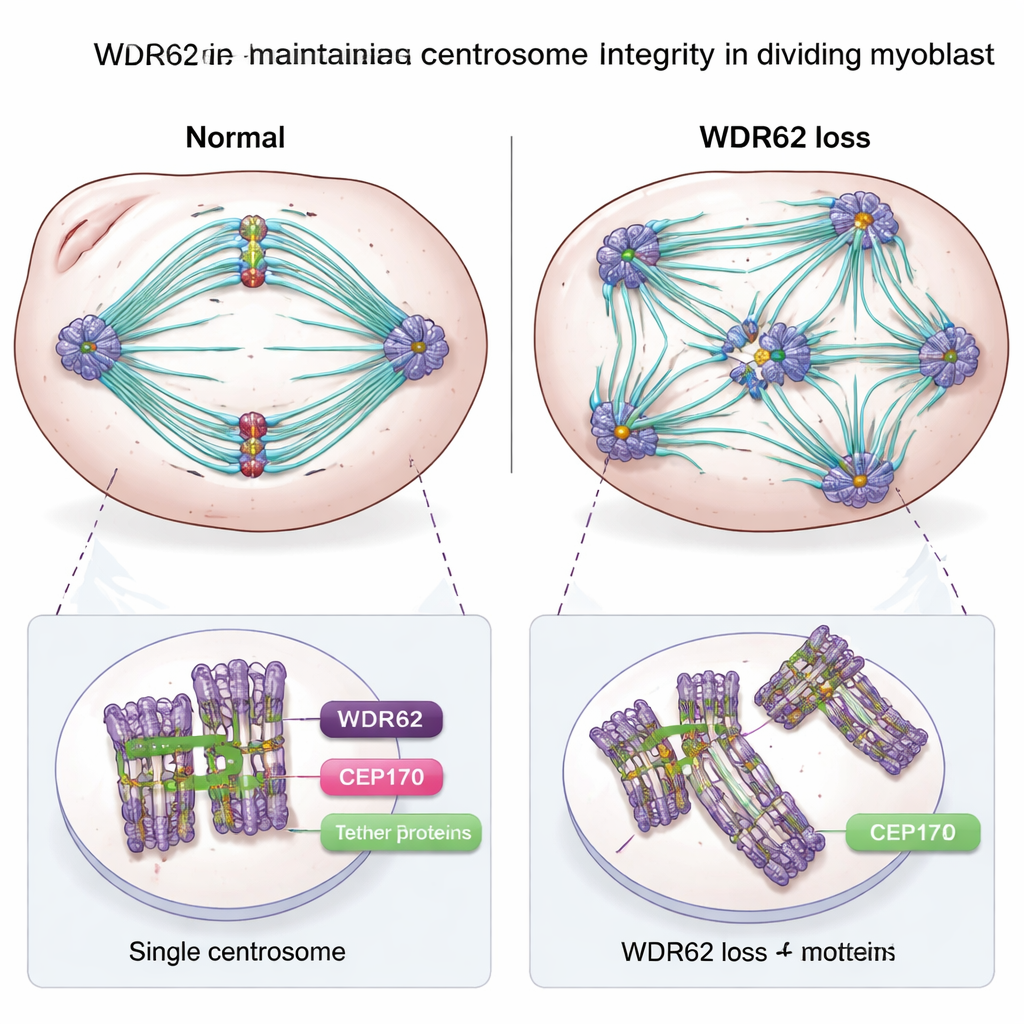
The cell’s “scaffold hubs” fall out of balance
Digging deeper, the researchers examined the centrosomes themselves. In healthy myoblasts, each dividing cell typically has two centrosomes, each with a pair of centrioles and carefully regulated links that hold them in the right configuration. In WDR62-deficient myoblasts, many cells instead carried extra centrioles and multiple centrosomes, along with abnormal patterns of cohesion between them. This kind of structural confusion is known to disrupt the orderly formation of the spindle that separates chromosomes, slowing or impairing cell division. The team also showed that WDR62 helps recruit another centrosome protein, CEP170, during the transition from dividing myoblast to early fused muscle cell. Together, these functions suggest that WDR62 is a key part of the machinery that both preserves centrosome integrity during proliferation and helps reorganize that machinery as cells commit to becoming muscle.
What this means for muscle health
For a non-specialist, the core message is that building and repairing muscle isn’t just about making cells grow; it is about preserving a healthy stock of precursor cells and deciding exactly when they should stop dividing and start maturing. WDR62 acts like a quality-control manager at the cell’s internal construction hubs, ensuring that myoblasts divide cleanly and in the right orientation before they fuse into fibers. When WDR62 is missing, myoblasts divide less reliably and commit to becoming muscle too soon, leading over time to smaller muscles and a thinner reserve of repair cells. Understanding this balance could eventually inform strategies for treating muscle-wasting conditions, where preserving or restoring the pool of muscle stem cells is just as important as encouraging their growth.
Citation: Ho, U.Y., Shohayeb, B., Kamei, H. et al. WDR62 is required for proper proliferation and early differentiation of skeletal myoblasts. Commun Biol 9, 259 (2026). https://doi.org/10.1038/s42003-026-09537-7
Keywords: muscle stem cells, myoblast differentiation, centrosome integrity, WDR62, skeletal muscle regeneration