Clear Sky Science · en
CHK1 inhibition rescues abnormal glycogen buildup in a Caenorhabditis elegans model for glycogen storage disease III
Why a tiny worm matters for a rare disease
Glycogen storage disease type III is a rare inherited condition in which the body cannot properly break down glycogen, the storage form of sugar. This leads to abnormal glycogen piling up in organs such as the liver and muscles, causing low blood sugar, enlarged livers, weakness, and other serious problems. Current treatments are limited, and existing animal models do not fully mirror what happens in people. In this study, researchers turned to an unlikely ally—a microscopic roundworm called Caenorhabditis elegans—to build a more accurate model of the disease and to search for new therapeutic targets.
Building a worm version of the human disorder
The team focused on the AGL gene, which makes the enzyme that trims branches off glycogen so it can be broken down. Mutations in AGL cause glycogen storage disease type III. Worms have a closely related gene called agl-1, so the researchers used CRISPR gene editing to introduce worm versions of two patient-linked mutations and to create a complete gene deletion. These engineered worms all showed clear signs of abnormal glycogen buildup, visible as heavy staining in their intestines, skin-like tissues, and muscles. They also became more sensitive to glucose: when exposed to sugar, more of their embryos failed to survive, closely echoing how patients’ cells struggle to handle energy properly. 
Health problems beyond sugar buildup
Although the mutant worms looked normal to the naked eye, closer tests revealed broad health issues. All three agl-1 mutants lived shorter lives and produced fewer offspring than normal worms. They developed more slowly from larvae into adults, and their movement was impaired. One variant in particular, called S1444R, showed striking problems with swimming and developed age-related paralysis on solid surfaces. These observations suggest that the worm model does not just store glycogen incorrectly; it also captures the broader decline in vitality seen in people with the disease, making it a powerful tool for studying how specific mutations affect the body.
Reading the worms’ genes for hidden clues
To understand what was going wrong inside the cells, the researchers examined gene activity across the entire worm genome. They found more than a thousand genes that were more active in the mutants than in normal worms, and a smaller set that were less active. Genes involved in sperm function, growth, and muscle development tended to be turned down, matching the observed fertility and movement problems. Meanwhile, pathways related to protein modification and phosphate metabolism were turned up, hinting that the worms’ cells were trying to compensate through changes in how proteins are switched on and off. This broad shift in gene activity painted a picture of a body under chronic metabolic stress.
From thousands of drugs to a few promising hits
The team next asked whether any existing drugs could improve the worms’ health. They screened nearly 4,000 small molecules and identified 25 that boosted swimming in the S1444R mutant. When they checked which of these also reduced glycogen buildup, two compounds stood out: pimozide, an antipsychotic drug, and pramoxine, a local anesthetic. Both are known to affect cell signaling pathways, and their impact on glycogen suggested deeper links between nerve-related signaling and energy handling. In parallel, the researchers grouped drug hits by their known targets and built a computational pipeline to highlight genes whose inhibition might mimic the beneficial effects of the compounds. This analysis pointed to several candidate genes that, when shut down by RNA interference, changed glycogen levels in the worms.
CHK1 inhibition as a new therapeutic angle
Among the candidate genes, one stood out: chk-1, which encodes a protein called CHK1 best known for controlling the cell cycle and helping cells respond to DNA damage. Turning down chk-1 in the S1444R worms reduced their glycogen buildup and improved their paralysis, without having the same effect in other mutants—indicating that the benefit depends on the specific disease variant. A drug that selectively blocks CHK1, called rabusertib, produced similar improvements in glycogen levels and movement. The team tested whether this effect worked through AMPK, a master energy sensor, but found that blocking AMPK did not change glycogen or chk-1 activity. This suggests that CHK1 influences glycogen storage through a separate, previously unrecognized route. 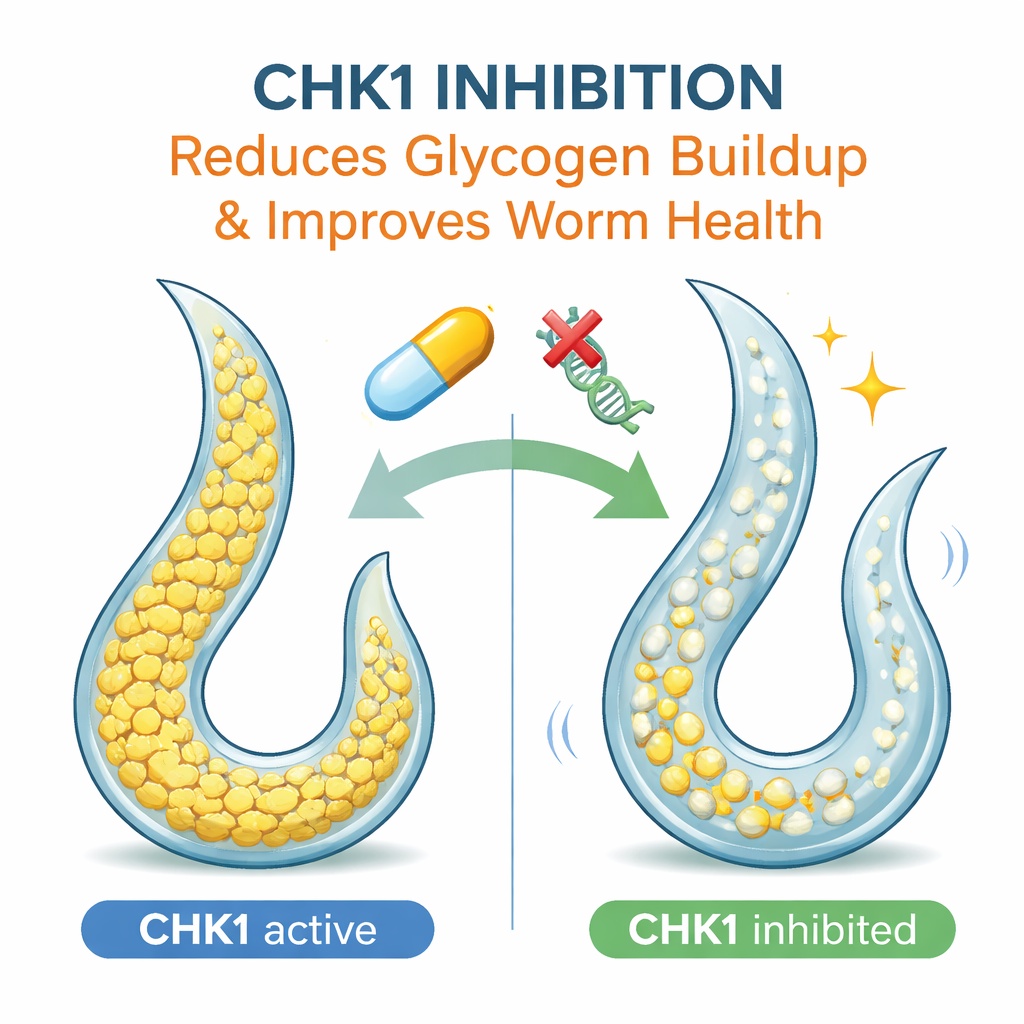
What this means for patients and future research
The study shows that a simple worm can faithfully reproduce key features of a complex human disease, including variant-specific differences in severity. Using this model, the researchers uncovered evidence that dialing down CHK1, either genetically or with a drug, can reduce harmful glycogen buildup and improve health measures in the worms. While these results are still limited to C. elegans and to one particular mutation, they point to CHK1 as a promising therapeutic target and illustrate how combining animal models, large-scale drug screens, and computational analysis can reveal unexpected strategies for treating rare metabolic disorders.
Citation: Daghar, H., Pyman, B., Maios, C. et al. CHK1 inhibition rescues abnormal glycogen buildup in a Caenorhabditis elegans model for glycogen storage disease III. Commun Biol 9, 257 (2026). https://doi.org/10.1038/s42003-026-09535-9
Keywords: glycogen storage disease, Caenorhabditis elegans, CHK1 inhibitor, rare metabolic disorder, drug repurposing