Clear Sky Science · en
Fetal reversion from diverse lineages sustains the intestinal stem cell pool and confers stress resilience
How the Gut Heals Itself Under Fire
The lining of your intestine is one of the busiest tissues in your body. It digests food, absorbs nutrients, and protects against germs, all while renewing itself every few days. This paper explores how that fragile surface survives harsh assaults—from inflammation to chemotherapy—by briefly rewinding cells to a more fetal-like, flexible state that helps the gut repair and resist damage.
The Everyday Assembly Line in the Gut
In healthy intestines, a specialized group of cells called crypt base columnar cells (CBCs) sit at the bottom of tiny pits known as crypts. These stem-like cells constantly divide and send their offspring upward toward the fingerlike villi that project into the gut tube. As they travel, the cells mature into different roles, such as absorptive enterocytes that take up nutrients, or secretory cells that make mucus and antimicrobial compounds. Under normal conditions, this top-to-bottom flow keeps the gut lining renewed and orderly.
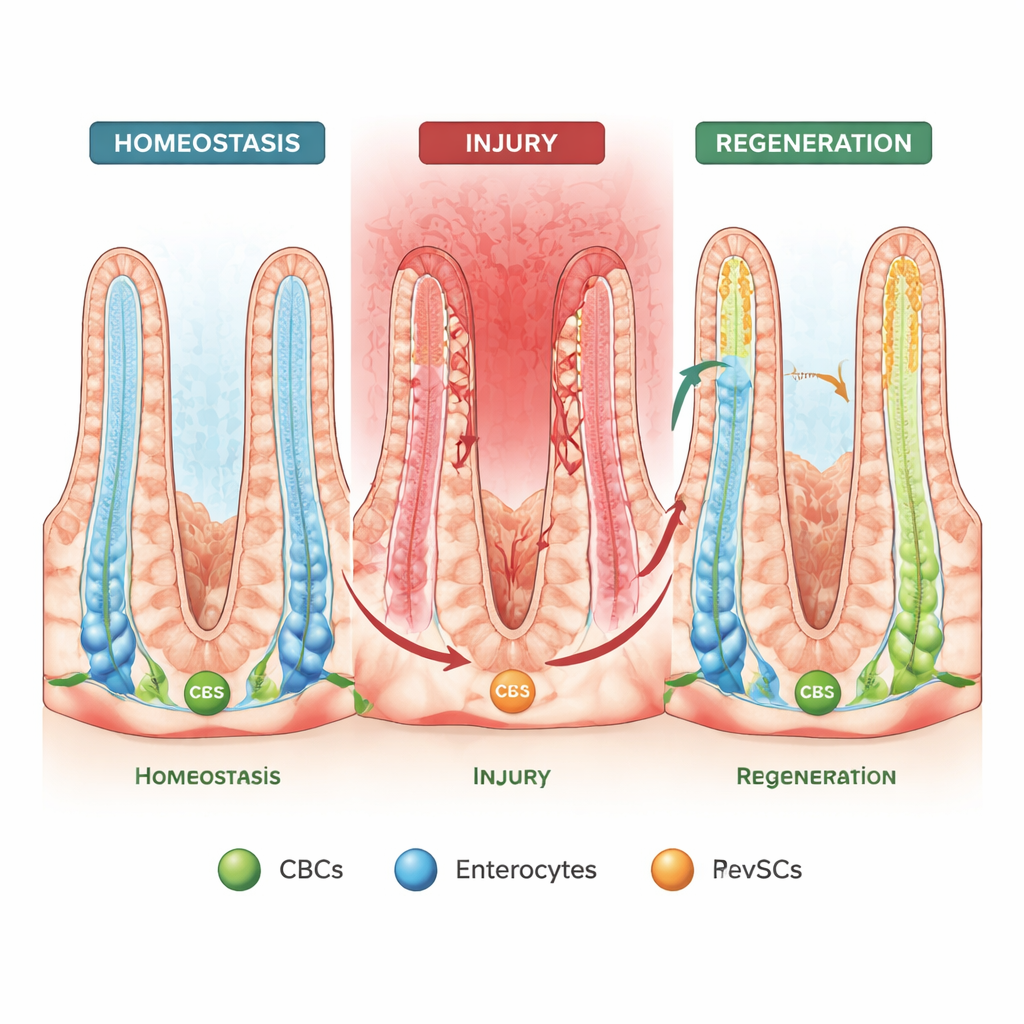
Cells That Can Run the Assembly Line Backwards
When the gut lining is injured and CBCs are lost, earlier work showed that some mature cells can “walk back” down the crypt and regain stem-like abilities, a behavior called spatial plasticity. A second, newer phenomenon is “fetal reversion,” in which cells adopt a gene activity pattern that resembles the developing intestine. In this study, the authors connect these two ideas using sophisticated organoid (mini-gut) cultures, single-cell RNA sequencing, and lineage tracing in mice. They identify a special fetal-like population called revival stem cells (revSCs) that can arise from both CBCs and ordinary absorptive enterocytes, and then regenerate a full set of gut cell types.
Mini-Guts Reveal Hidden Flexibility
To watch these changes in action, the team grew mouse and human intestinal organoids in two different gels. In Matrigel, organoids behaved like normal adult tissue, rich in CBCs and mature cell types. In a collagen gel, however, organoids became dominated by revSC-like cells with a fetal gene signature. Single-cell analyses showed that revSCs could be traced back along two main paths: from classic CBCs and from absorptive enterocytes. Sorting experiments confirmed that CBCs have the highest capacity to convert into revSCs, but even more mature cells could do so when placed in the right environment. Crucially, purified revSCs could be moved back into Matrigel and would then regenerate CBCs and all major intestinal lineages, proving that the fetal-like state is both reversible and fully functional.
Villus Cells and Real-World Injury
The study goes a step further by asking whether this flexibility appears outside the lab. The researchers labeled enterocytes in the villi of mice and then either cultured villus fragments or exposed animals to a gut-damaging chemotherapy drug, 5-fluorouracil (5-FU). In collagen, villus fragments—normally considered stem-cell-poor—formed new organoids that adopted revSC features and later regained CBC markers when shifted back to standard conditions. When transplanted into colitis models, these "villus-derived" organoids rebuilt healthy intestinal lining. In live mice treated with 5-FU, labeled enterocytes began expressing revSC markers, then later reappeared as long clonal stripes that included new CBCs at the crypt base, confirming that mature cells had stepped in to refill the stem cell pool.
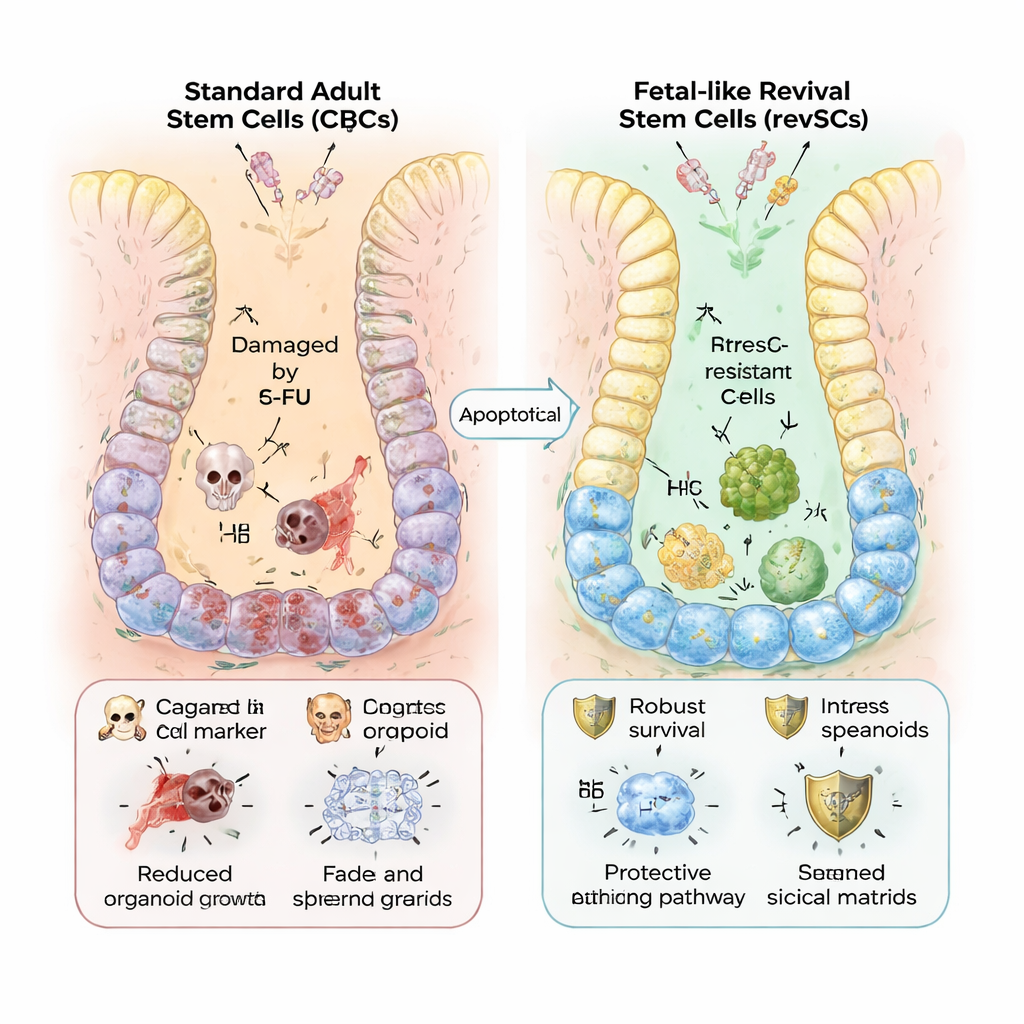
Stress-Resistant Repair Crews
Why bother with this fetal-like detour? The authors show that revSCs are far better at withstanding stress. When organoids were challenged with 5-FU, those in standard Matrigel shrank and showed broad gene programs linked to DNA damage and frantic proliferation. Collagen-grown, revSC-rich organoids, by contrast, kept growing and displayed a calmer, more targeted response. Gene analyses revealed higher activity of antioxidant systems, anti-death proteins, and heat shock chaperones—molecular shields that help cells survive inflammation and toxic insults. Similar patterns were seen in both mouse and human cells, and the same revSC genes are elevated in inflamed human intestines.
What This Means for Gut Disease and Therapy
To a non-specialist, the takeaway is that the gut lining has a built-in emergency escape plan. When inflammation or drugs threaten its usual stem cells, both stem cells and ordinary absorptive cells can temporarily revert to a fetal-like, stress-resistant revSC state. From there, they can recreate the normal stem cell compartment and rebuild the tissue. This work unifies two regeneration concepts—spatial plasticity and fetal reversion—into a single repair hierarchy, and suggests that carefully harnessing this flexible, fetal-like program could improve treatments for conditions such as inflammatory bowel disease and colorectal cancer, while also warning that mismanaged reversion may fuel tumor growth.
Citation: Kirino, S., Uefune, F., Miyake, K. et al. Fetal reversion from diverse lineages sustains the intestinal stem cell pool and confers stress resilience. Commun Biol 9, 255 (2026). https://doi.org/10.1038/s42003-026-09533-x
Keywords: intestinal stem cells, tissue regeneration, cell plasticity, inflammatory bowel disease, organoids