Clear Sky Science · en
Fluvastatin suppresses breast cancer initiation and progression via targeting CYP4Z1
Why a cholesterol drug matters for breast cancer
Many women treated for breast cancer develop abnormal blood fat levels and face the ongoing threat of the disease coming back or spreading. This study explores an intriguing idea: could a common cholesterol-lowering pill, fluvastatin, be repurposed to slow or prevent breast cancer by disarming a protein that fuels particularly dangerous, stem cell–like tumor cells?
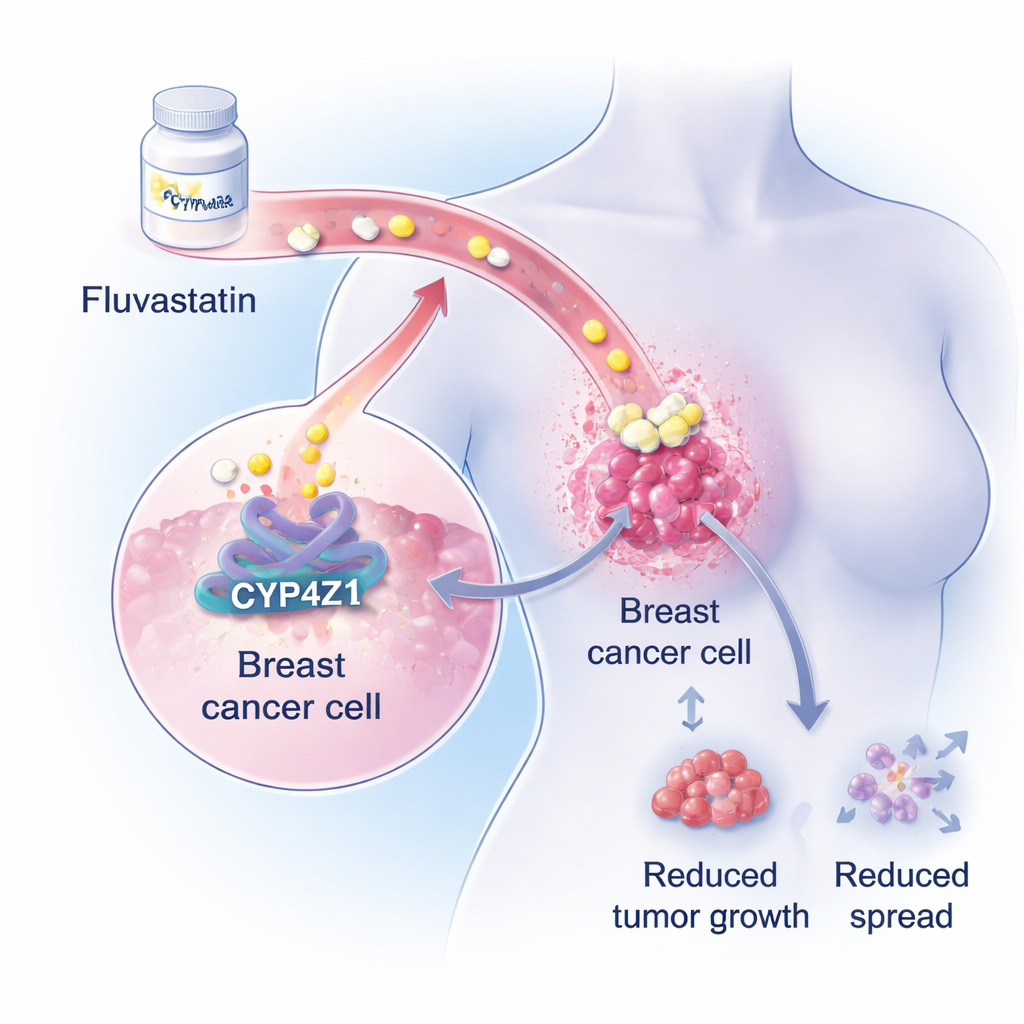
A hidden driver inside breast tumors
The researchers focus on a little-known protein called CYP4Z1, found at high levels in breast cancer cells but not in healthy breast tissue. Earlier work showed that CYP4Z1 helps cancer cells behave more like stem cells—cells that can endlessly self-renew, resist chemotherapy, and restart tumors after treatment. In large genetic and biochemical analyses, the team now shows that overactive CYP4Z1 strongly boosts fat-building pathways inside breast cancer cells, especially the production of triglycerides, a major storage fat. Because many breast cancer patients already develop disordered blood fats during chemotherapy, this fat-promoting role of CYP4Z1 suggested an opening for existing lipid-lowering drugs.
Repurposing fluvastatin from heart health to cancer care
To test this idea, the scientists screened a small library of 23 FDA-approved drugs used to lower blood lipids. Fluvastatin, a widely prescribed statin pill for high cholesterol, emerged as a standout: it directly blocked CYP4Z1’s enzyme activity in a dose-dependent way. At doses that were much less harmful to normal mammary cells than to cancer cells, fluvastatin sharply reduced key stemness markers, the ability of breast cancer cells to form free-floating “tumor spheres,” and their capacity to migrate and invade through tissue-like barriers in lab tests. It also made tumor cells more sensitive to the chemotherapy drug adriamycin, hinting that it could enhance existing treatments.
Putting the drug to the test in living animals
The team then evaluated fluvastatin in mouse models. In mice implanted with human breast cancer cells, periodic fluvastatin treatment slowed tumor growth, lowered levels of a stemness marker (ALDH1A1) and the cell division marker Ki67 in tumors, and reduced the number of metastatic nodules that appeared in the lungs. Importantly, mice receiving fluvastatin maintained their body weight and showed no major blood or bone marrow abnormalities, supporting a favorable safety profile at the tested dose. In a more realistic genetic model—mice engineered to develop breast tumors driven by a cancer-causing gene—the researchers further introduced human CYP4Z1 into the mammary tissue. These mice developed more pre-cancerous and cancerous lesions, and more liver and lung metastases, than control animals. Fluvastatin treatment substantially reduced both the number of mammary lesions and distant metastases, especially in mice carrying the extra CYP4Z1 gene.
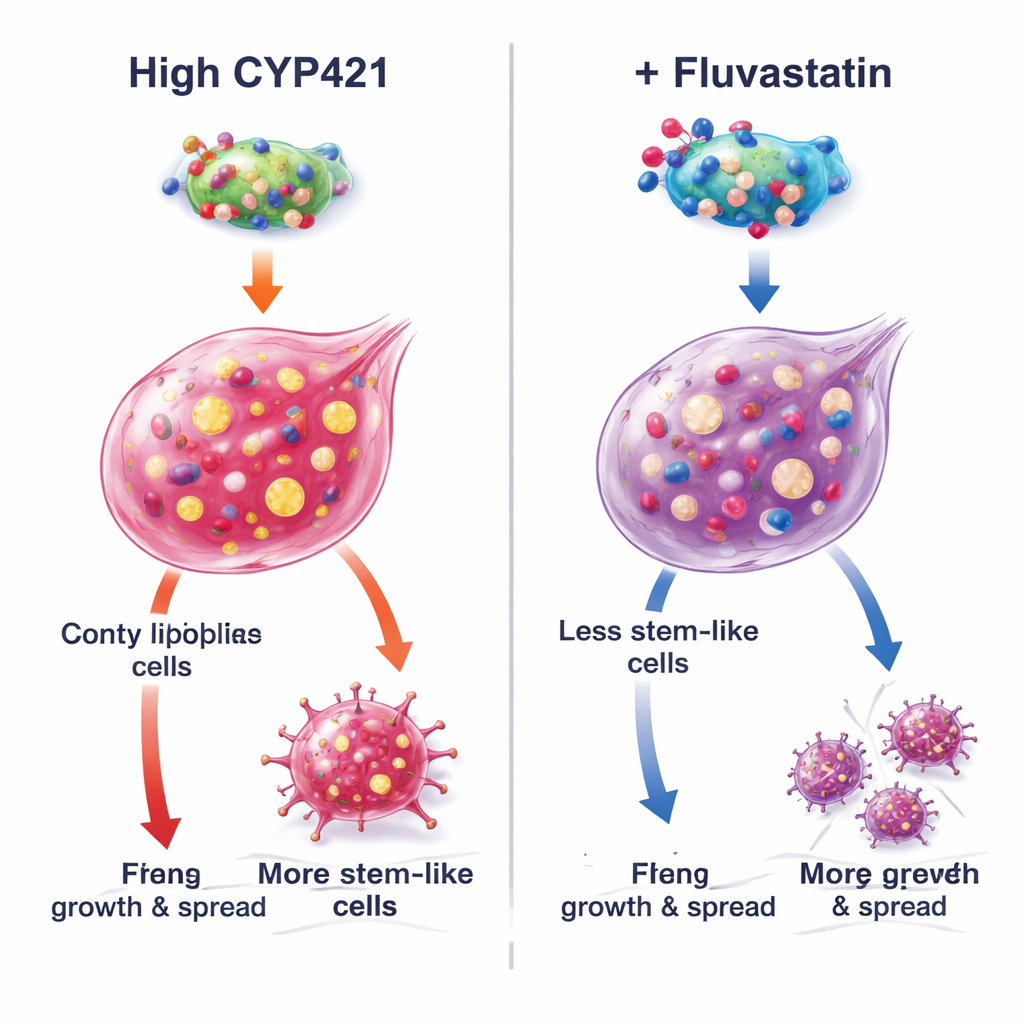
How fluvastatin disarms a cancer-promoting protein
To understand the mechanism, the authors combined computer modeling with precise protein engineering. They predicted that fluvastatin wedges into CYP4Z1’s active site at three critical amino acids (Lys109, Pro444, and Arg450). When those sites were mutated, CYP4Z1’s activity fell and fluvastatin could no longer further inhibit it, confirming these positions as docking points. In cells that naturally lack CYP4Z1, adding the normal protein increased stemness and activated growth-promoting PI3K/AKT signaling, while fluvastatin reversed these effects. Mutant versions of CYP4Z1 were less potent and less responsive to the drug. Additional experiments showed that fluvastatin’s impact weakened when triglyceride production was artificially increased, or when CYP4Z1 was knocked down, underscoring that the drug’s strongest effects come from blocking this specific protein and its fat-building consequences rather than from triggering cell death through iron-driven "ferroptosis."
What this means for patients
Overall, the study suggests that fluvastatin, beyond lowering cholesterol, can curb the earliest stages and later spread of breast cancer in experimental models by targeting CYP4Z1, a protein that drives fatty metabolism and stem cell–like behavior in tumors. Because fluvastatin is already approved and its safety is well characterized, these findings offer a promising path toward faster clinical testing as an add-on therapy, particularly in patients whose tumors strongly express CYP4Z1. While more work is needed to test this approach in people and to identify any additional targets of the drug, the research opens the door to using a familiar heart medicine as part of a new strategy against aggressive, relapse-prone breast cancers.
Citation: Li, H., Chen, Y., Shi, W. et al. Fluvastatin suppresses breast cancer initiation and progression via targeting CYP4Z1. Commun Biol 9, 254 (2026). https://doi.org/10.1038/s42003-026-09532-y
Keywords: breast cancer, fluvastatin, cancer stem cells, lipid metabolism, drug repurposing