Clear Sky Science · en
Differentially expressed miRNAs in the temporal cortex of Alzheimer’s disease patients and their association to tau pathology
Why tiny brain switches matter in Alzheimer’s
Alzheimer’s disease slowly robs people of memory and independence, yet we still lack treatments that stop or reverse it. This study looks at some of the brain’s tiniest control switches—short RNA molecules called microRNAs—that help decide which proteins cells make. By examining brain tissue from people who had Alzheimer’s, the researchers asked whether changes in these microRNAs are tied to a key disease feature: the buildup and spread of abnormal tau protein inside brain cells.
Looking at messages in a vulnerable brain region
The team focused on the temporal cortex, a region important for language and memory that is heavily affected in Alzheimer’s. After death, samples from 36 individuals—13 without Alzheimer’s and 23 with the disease—were collected from two European brain banks. The scientists extracted RNA from this tissue and used high-throughput sequencing to count hundreds of different microRNAs. Because brain tissue degrades after death, they carefully checked RNA quality and selected 19 samples with acceptable integrity for the most detailed analysis, then used all 36 samples to confirm their key findings.
Discovering a microRNA signature of Alzheimer’s
Among 449 detected microRNAs, 13 stood out as consistently altered in the Alzheimer’s temporal cortex. Eight were reduced and five were increased compared with control brains. Several of the strongest signals involved microRNAs already suspected to be important in Alzheimer’s: miR-129-5p, miR-132-3p, and miR-146b-5p were all lower, while miR-151a-5p was higher. These molecules are abundant in the brain and are known to help regulate networks of genes involved in neuronal health. Using a second, more targeted technique (RT-qPCR), the authors validated that these same microRNAs changed in the same direction, even when including lower-quality samples, strengthening confidence that the pattern is real.
Linking tiny switches to worsening tau tangles
Alzheimer’s severity in the brain is often graded by Braak stage, which reflects how far tau tangles have spread through different regions. The researchers found that levels of miR-129-5p, miR-132-3p, and miR-146b-5p declined steadily as Braak stage increased, while miR-151a-5p tended to rise. They also directly measured a highly phosphorylated, tangle-prone form of tau in the same samples. Brains with more of this abnormal tau showed the same pattern: less miR-129-5p, miR-132-3p, and miR-146b-5p, and more miR-151a-5p. In other words, the microRNA changes tracked with both microscopic staging and biochemical signs of tau pathology, suggesting they are closely tied to the disease process rather than random bystanders.
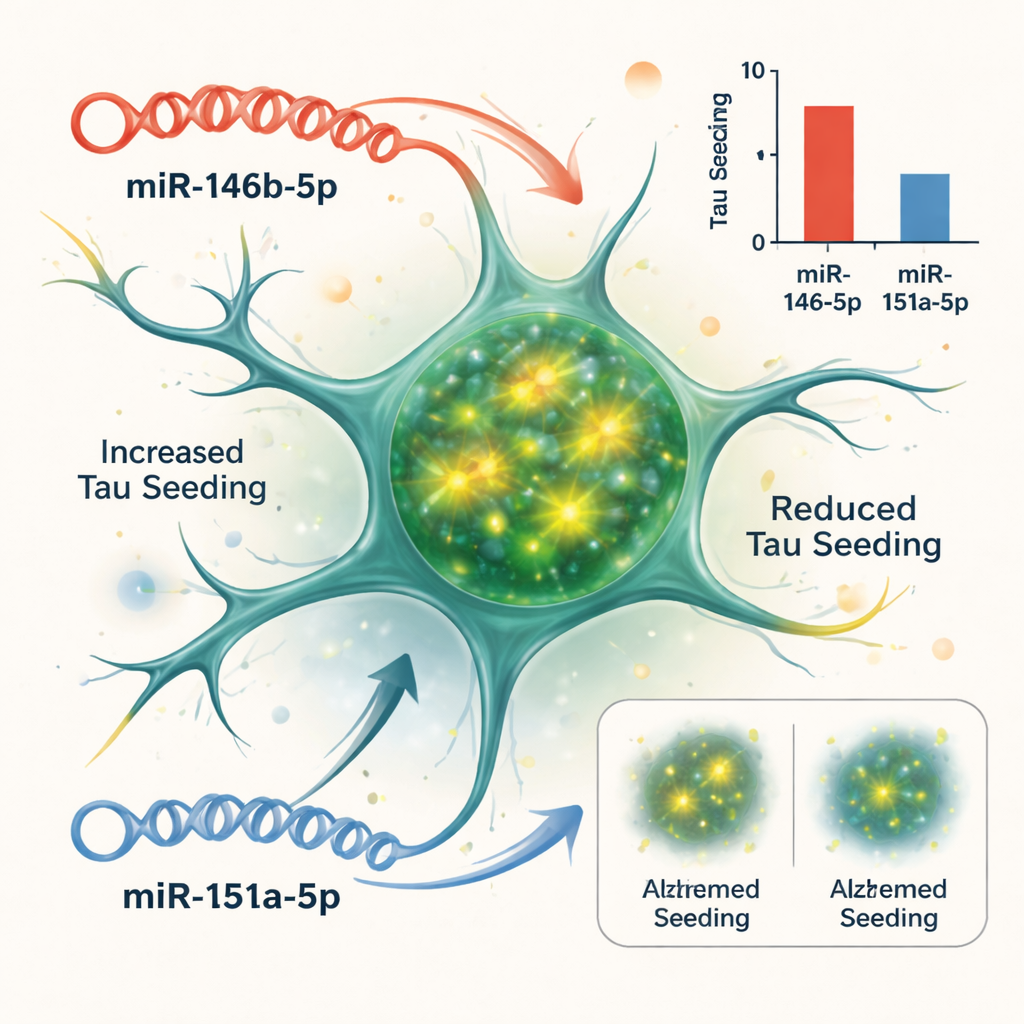
Testing cause and effect in a cellular model
Correlation does not prove that microRNAs drive disease, so the team turned to a cell-based system that reports how easily tau aggregates can “seed” new clumps. They used biosensor cells engineered to light up when tau fibers form inside them. When they added tau-rich material from Alzheimer’s brains, the cells produced a strong fluorescence signal. By boosting or blocking specific microRNAs in these cells, the scientists could see which ones altered this tau seeding. Two stood out: increasing miR-146b-5p made tau seeding worse, while blocking it reduced seeding; in contrast, increasing miR-151a-5p dampened tau seeding, while inhibiting it made seeding more robust. Other tested microRNAs had little effect in this assay.
What this could mean for future diagnosis and treatment
To understand what these microRNAs might be doing, the authors used computational tools to map their predicted gene targets onto known biological pathways. The downregulated microRNAs—especially miR-129-5p, miR-132-3p, and miR-146b-5p—were linked to networks involved in nerve cell survival, chemical signaling at synapses, and Alzheimer’s-related pathways. This suggests that losing their normal “braking” influence could disrupt multiple protective systems at once, while the rise in miR-151a-5p may represent a late, partial attempt by the brain to counteract tau aggregation. Although this work relies on post-mortem tissue and simplified cell models, it strengthens the idea that specific microRNAs both mirror and modulate tau pathology. In the long run, measuring these molecules in cerebrospinal fluid or blood could help track disease progression, and precisely adjusting their levels in the brain might offer a new route to slow or prevent the spread of toxic tau.
Citation: Nagaraj, S., Quintanilla-Sánchez, C., Ando, K. et al. Differentially expressed miRNAs in the temporal cortex of Alzheimer’s disease patients and their association to tau pathology. Commun Biol 9, 253 (2026). https://doi.org/10.1038/s42003-026-09530-0
Keywords: Alzheimer’s disease, microRNA, tau pathology, temporal cortex, neurodegeneration