Clear Sky Science · en
IL-15 overexpression promotes memory program and anti-tumor activity of CD64 CAR T cells in a preclinical AML model
Why this research matters
For adults with a hard-to-treat blood cancer called acute myeloid leukemia (AML), especially after the disease comes back, existing therapies often fail and survival rates are low. This study explores an upgraded form of a cutting-edge treatment—CAR T cells—that is redesigned to better hunt down leukemia cells while lasting longer in the body. By adding a natural immune-boosting signal called IL-15 to AML-targeting CAR T cells, the researchers aim to build a smarter, more durable “living drug” for patients who currently have few options.
A stubborn form of blood cancer
AML is a cancer of the bone marrow in which immature white blood cells multiply uncontrollably, crowding out healthy blood production. The outlook is especially grim for older adults and for patients whose disease relapses after chemotherapy or a stem cell transplant. While CAR T cell therapy has transformed treatment for some B cell blood cancers, it has not yet worked as well in AML. One roadblock is finding targets that are present on leukemia cells but spare healthy stem cells; another is that CAR T cells tend to fizzle out too quickly in the harsh AML environment. The team previously identified a surface marker called CD64, abundant on certain AML subtypes (M4/M5), as a promising target with limited impact on blood-forming stem cells—but early CAR T versions using CD64 did not keep the disease under control for long.
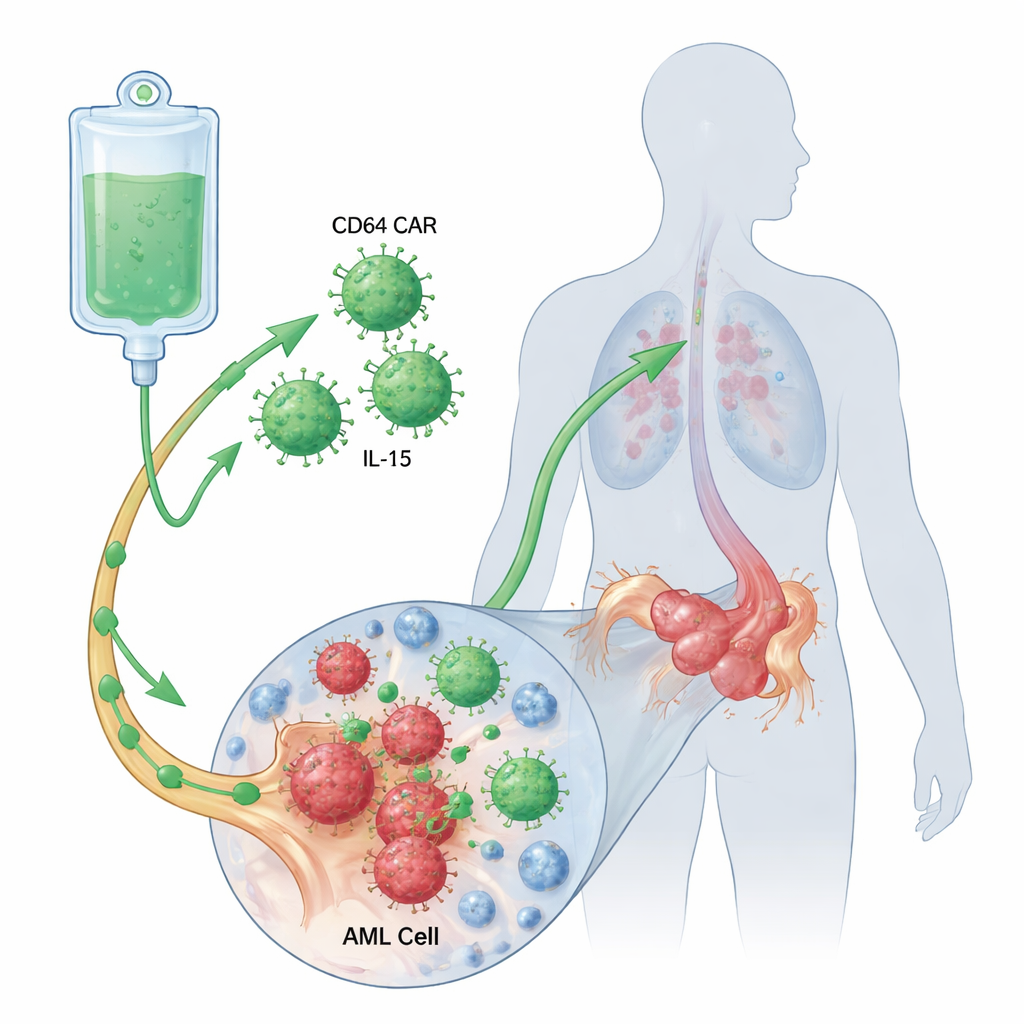
Designing a more persistent cancer hunter
To strengthen these CD64-targeting CAR T cells, the researchers engineered them to overproduce IL-15, a natural immune messenger that helps T cells survive, divide, and develop into long-lasting “memory” cells. They compared three setups in the lab: standard CD64 CAR T cells, the same cells grown with extra IL-15 added to the culture, and CAR T cells that continuously make their own IL-15. In short-term killing tests against several CD64-positive leukemia cell lines, the IL-15-armed CAR T cells were more efficient at destroying cancer cells, particularly at lower T cell–to–tumor ratios. These cells also released more key immune messengers such as IL-2 and interferon-gamma when engaging leukemia, without an across-the-board surge in inflammatory signals.
Building lasting immune memory
Next, the team tested how well the different CAR T cells could expand and endure when repeatedly challenged with leukemia cells in culture. IL-15-producing CAR T cells showed roughly double the expansion of standard CAR T cells and favored a CD8-positive, central memory–like state—cell types known to persist longer and respond more vigorously when they re-encounter cancer. Markers of cell death were reduced, and proteins linked to T cell “exhaustion,” such as LAG-3 and TIM-3, were lower on IL-15 CAR T cells. Gene expression analysis revealed that these cells turned on programs related to proliferation, survival, energy metabolism, and central memory formation, especially through pathways like PI3K–AKT and mitochondrial function. Importantly, simply adding IL-15 from the outside did not reproduce the same durable reprogramming, underscoring the advantage of built-in IL-15 production.
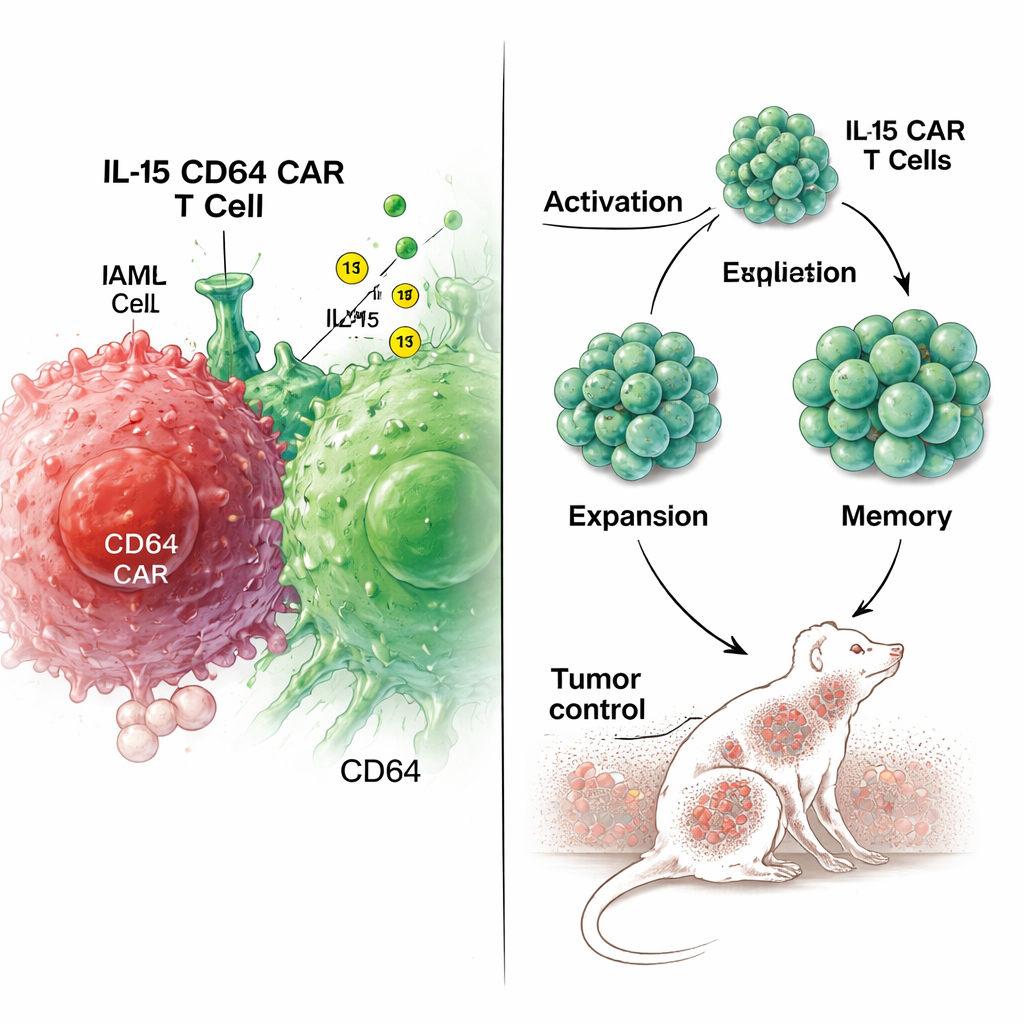
Stronger tumor control in mouse models
The real test came in mice transplanted with human AML cells. When treated with either standard CD64 CAR T cells or IL-15-overexpressing CD64 CAR T cells, both groups initially slowed tumor growth compared with control T cells. However, animals receiving IL-15 CAR T cells cleared leukemia faster, stayed in remission longer, and survived more than twice as long as those treated with conventional CAR T cells. Blood tests showed that IL-15 CAR T cells expanded better and persisted longer in circulation. They also released more of certain cell-killing proteins such as perforin, suggesting enhanced direct attack on tumor cells, while levels of many inflammatory cytokines remained similar or only modestly changed. Tissue examinations at the end of the experiments showed no major organ damage, and only mild lung changes in both CAR T–treated groups, indicating no obvious added toxicity from IL-15.
What this could mean for patients
This preclinical work suggests that equipping CD64-targeted CAR T cells with IL-15 can turn them into tougher, longer-lived cancer fighters that are better suited to the demanding setting of relapsed or refractory AML. By promoting a memory-like state, reducing exhaustion, and boosting expansion without clear additional organ damage in mice, IL-15 overexpression offers a promising strategy to improve future CAR T therapies. While more safety testing—including in models that better mimic human blood stem cells and immune reactions—is still needed, this research lays groundwork for clinical trials that could eventually provide a new treatment lifeline for patients with aggressive AML who currently have limited options.
Citation: Shan, L., Li, C., Li, T. et al. IL-15 overexpression promotes memory program and anti-tumor activity of CD64 CAR T cells in a preclinical AML model. Commun Biol 9, 251 (2026). https://doi.org/10.1038/s42003-026-09528-8
Keywords: acute myeloid leukemia, CAR T cells, CD64, interleukin-15, immunotherapy