Clear Sky Science · en
Distinct effects of different Bacteroides strains on depressive-like behavior via a gut-Th1/Th17 cells-brain axis
How Gut Bugs May Shape Our Moods
Depression is usually blamed on brain chemistry, stress, or life events. But a growing body of research suggests something more surprising is involved: the trillions of microbes living in our intestines. This study zooms in on three common gut bacteria and shows how they can nudge the immune system in ways that either worsen or ease depression-like behavior in mice, and how similar patterns show up in people with major depression.
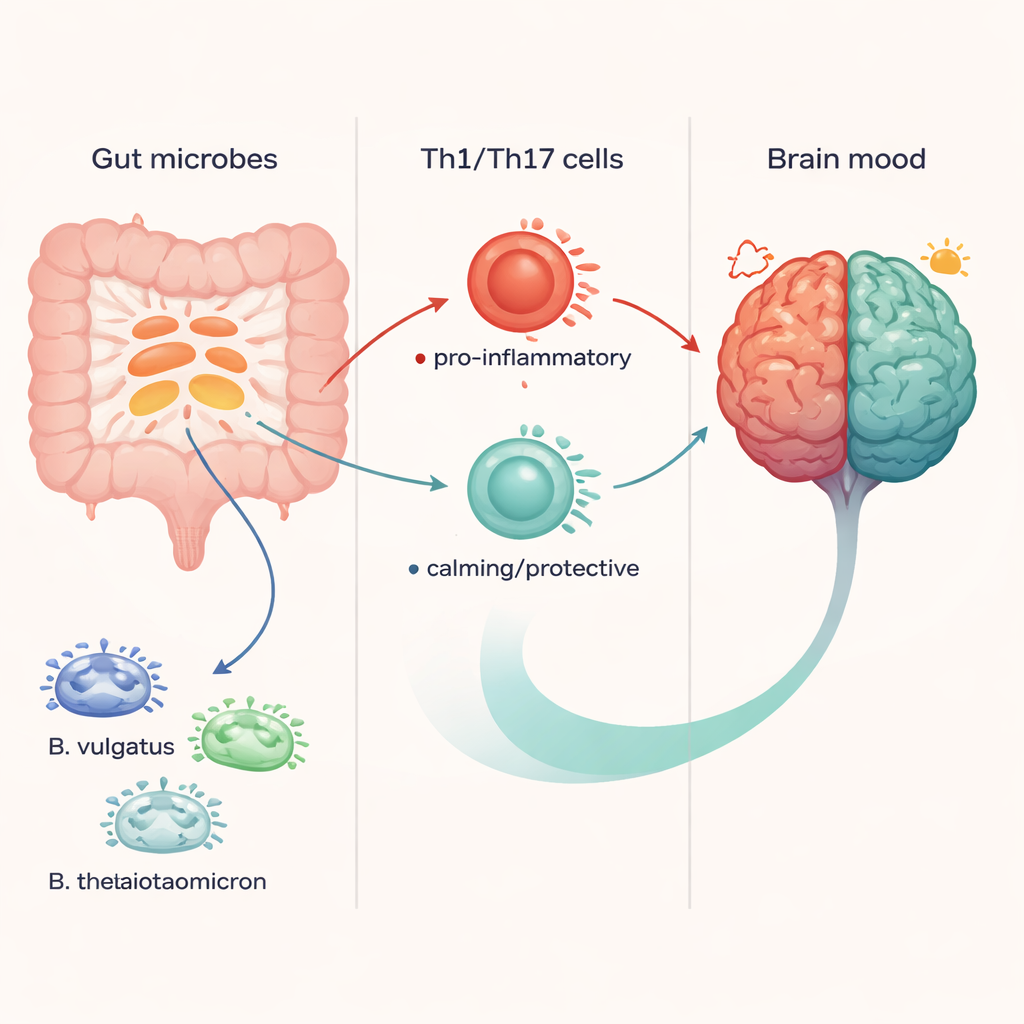
Different Gut Neighbors, Different Mood Outcomes
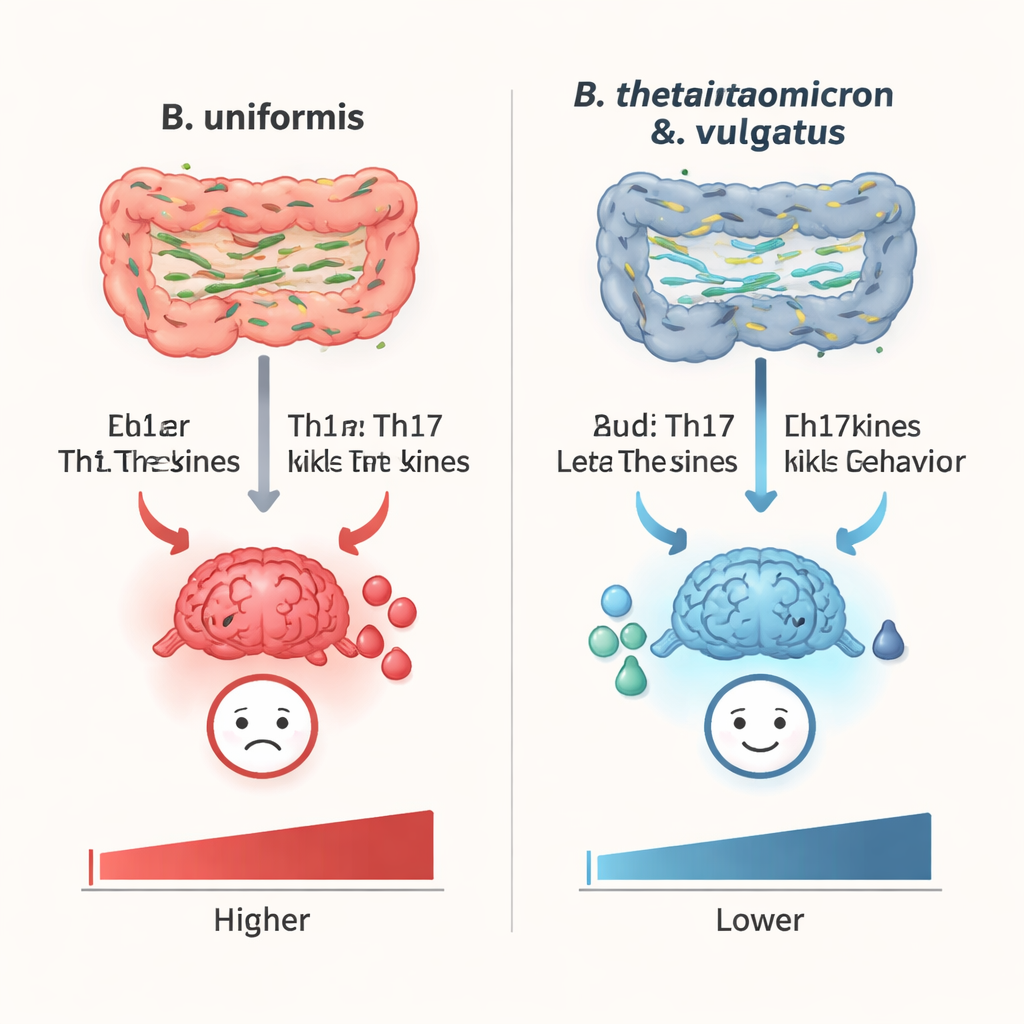
The researchers focused on three species from the Bacteroides family, which are abundant in the human gut and often altered in people with major depressive disorder. They tested Bacteroides uniformis, Bacteroides vulgatus, and Bacteroides thetaiotaomicron, asking a simple but powerful question: do these microbes push the body toward or away from depression? Using mouse models of chronic stress, they found that B. uniformis made animals more prone to depression-like behavior, while B. vulgatus and B. thetaiotaomicron had the opposite effect, helping stressed mice behave more like healthy controls.
Immune Cells as Middlemen Between Gut and Brain
To uncover how these bacteria influence mood, the team looked closely at key immune cells called T helper cells, especially two types named Th1 and Th17. These cells help coordinate inflammation and can send powerful signals throughout the body. In lab dishes, B. uniformis pushed naïve immune cells to become more Th1 and Th17 cells, while B. thetaiotaomicron did the reverse, dampening their formation; B. vulgatus had little effect in this simplified setting. In stressed mice, B. uniformis boosted Th17 cells in the spleen, blood, and gut-associated tissues and raised levels of inflammatory molecules in the hippocampus, a brain region linked to mood. In contrast, B. vulgatus and B. thetaiotaomicron lowered Th1 and Th17 levels and reduced brain inflammation.
Proving Cause and Effect in Depressive-like Behavior
Behavioral tests gave these immune shifts real-world meaning. Mice treated with B. uniformis after antibiotics and mild stress spent less time exploring open areas, lost interest in sweet solutions, floated longer in forced swim tests, and hesitated more before eating in a new environment—all standard hallmarks of depression-like behavior in animals. Blocking Th17 cell development with a drug called SR1001 largely reversed these changes, suggesting that B. uniformis drives vulnerability to depression mainly through Th17 cells and their signature inflammatory signal, IL-17A, in the brain. By contrast, in a stronger stress model, mice given B. vulgatus or B. thetaiotaomicron showed improved exploration, more sucrose drinking, and less immobility and anxiety, indicating these microbes can actively protect against stress-induced depressive-like behavior.
Human Clues From Blood and Stool
The team then looked for similar patterns in people. In blood samples from patients with major depressive disorder, they found more Th1 and Th17 cells and fewer regulatory T cells compared with healthy volunteers. Higher Th17 levels closely tracked with more severe depression scores. Stool DNA sequencing revealed that B. uniformis was more abundant in depressed patients and rose in step with both symptom severity and Th17 levels. B. thetaiotaomicron tended to be less abundant and showed the opposite pattern, being linked to milder symptoms and fewer Th17 cells. B. vulgatus did not show clear changes in this clinical sample, hinting that its protective effects might depend more on specific contexts or strains.
Microbial Chemistry and Future Therapies
Finally, the researchers examined chemical by-products in mouse feces to see how these microbes might be steering the immune system. B. uniformis lowered levels of butyric acid and cholesterol sulfate, compounds known to restrain Th17 and related inflammatory responses. B. thetaiotaomicron, in contrast, boosted butyric and propionic acids and biotin, all linked to calming Th1 and Th17 activity. Intriguingly, even heat-killed bacteria—dead cells that cannot colonize—still shifted immune cells, behavior, and metabolites, hinting that bacterial components themselves can send powerful signals. Together, the results support the idea of a “gut–Th1/Th17–brain” axis, where certain microbes foster inflammatory immune cells that disturb the brain, while others promote a more balanced immune state that protects mood.
What This Means for People Living With Depression
For non-specialists, the key message is that not all gut bacteria are equal when it comes to mental health. In this work, one species, B. uniformis, acted like a “pro-depression” microbe in mice and was more common in people with severe depression, likely by fueling immune cells that inflame the brain. Two others, B. vulgatus and B. thetaiotaomicron, did the opposite, calming those same immune pathways and easing depressive-like behavior. While much remains to be learned before doctors can prescribe specific microbes or microbial products as treatments, the study offers a concrete blueprint: by tuning the gut community and the immune messengers it controls, we may one day develop new, targeted ways to prevent or treat depression.
Citation: Li, Z., Qin, P., Sun, Z. et al. Distinct effects of different Bacteroides strains on depressive-like behavior via a gut-Th1/Th17 cells-brain axis. Commun Biol 9, 247 (2026). https://doi.org/10.1038/s42003-026-09525-x
Keywords: gut microbiome, depression, immune system, Bacteroides, Th17 cells