Clear Sky Science · en
Nimbolide ameliorates ARDS and ulcerative colitis by disrupting NLRP3 inflammasome activation
Why calming runaway inflammation matters
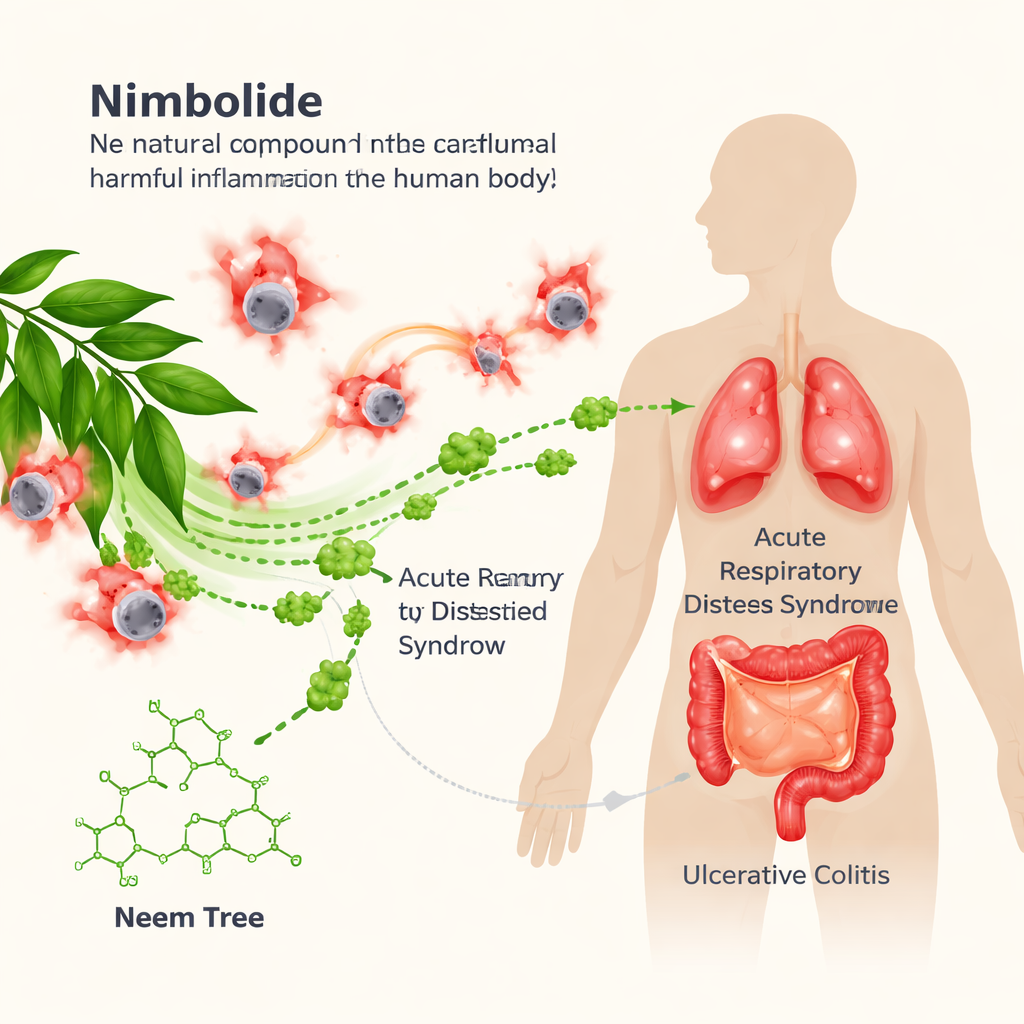
Many serious illnesses, from life‑threatening lung failure to painful bowel disease, are driven not just by germs or injuries, but by the body’s own immune system going into overdrive. This study explores whether a natural compound called nimbolide, derived from the neem tree, can act as a precise “brake” on one powerful inflammatory machine inside our immune cells, potentially opening a safer path to treating conditions such as acute respiratory distress syndrome (ARDS) and ulcerative colitis.
A troublemaking alarm inside immune cells
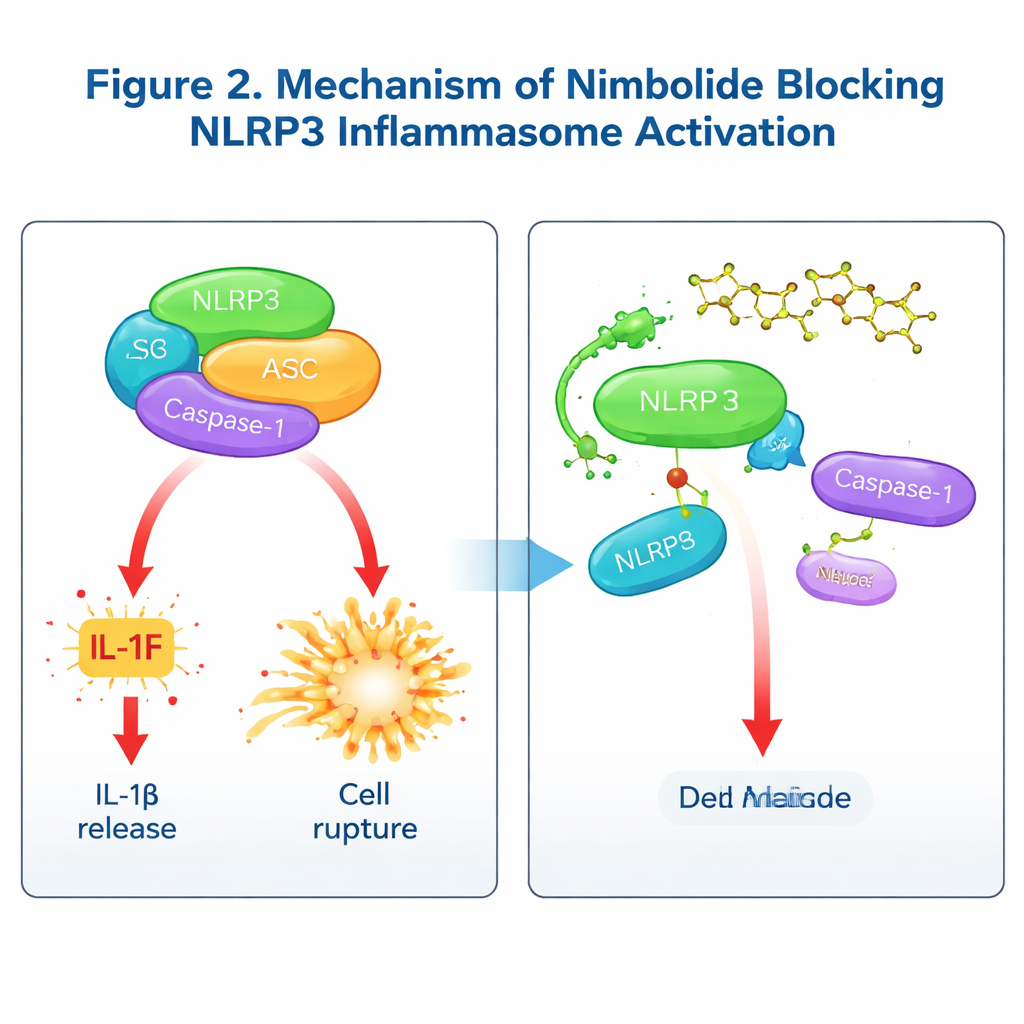
At the heart of this story is a molecular alarm system called the NLRP3 inflammasome. When immune cells sense danger—signals from infections, damaged tissues, or crystals—it can snap together like a Lego structure made of three parts: the sensor NLRP3, the adaptor ASC, and the enzyme Caspase‑1. Once assembled, this complex activates potent messenger molecules such as IL‑1β and IL‑18 and can trigger a fiery form of cell death called pyroptosis. While this response helps clear threats, when it is over‑activated it contributes to chronic and acute diseases, including arthritis, diabetes complications, Alzheimer’s disease, ARDS, and inflammatory bowel disease. Drug makers have tried to tame NLRP3 with synthetic compounds, but many candidates have stumbled due to toxicity, side effects, or poor behavior in the body, leaving a clear need for safer and more selective inhibitors.
Discovering a potent plant‑based inhibitor
The researchers screened 126 natural compounds to see which best blocked IL‑1β release from mouse immune cells. Nimbolide stood out, reducing IL‑1β secretion by more than 80% at test doses. Further experiments showed that nimbolide strongly reduced activation of Caspase‑1, release of IL‑1β, and pyroptotic cell death in mouse bone‑marrow‑derived macrophages, with potency similar to a benchmark drug called MCC950. Importantly, nimbolide did not blunt the release of other immune messengers like IL‑6, suggesting it acts in a focused way rather than simply shutting down immunity. When tested across different triggers that all activate NLRP3—such as ATP, nigericin, alum crystals, and imiquimod—nimbolide consistently reduced inflammasome activity, including in human THP‑1 cells. By contrast, it did not block two related inflammasomes, AIM2 and NLRC4, highlighting its specificity for NLRP3.
How nimbolide blocks the inflammatory machine
To understand how nimbolide works, the team asked whether it interferes with early “priming” steps controlled by the NF‑κB pathway, or with later stages when the inflammasome physically assembles. Short‑term treatment, given after priming, left key priming signals and gene expression intact but still reduced IL‑1β and Caspase‑1 activation, showing that nimbolide acts mainly on the assembly step. Detailed biochemical tests revealed that nimbolide does not alter potassium or calcium flux, mitochondrial damage, or the interaction of NLRP3 with its helper protein NEK7. Instead, it disrupts the crucial handshake between NLRP3 and ASC, reduces ASC clustering into specks, and prevents NLRP3 from forming higher‑order oligomers. Using binding assays and molecular modeling, the authors showed that nimbolide directly attaches to the NACHT region of NLRP3 at a specific amino acid, Lys565. When this site was mutated, nimbolide could no longer protect NLRP3 from degradation or break its link with ASC, confirming Lys565 as the key docking point. Notably, while other drugs often target NLRP3’s ATPase activity, nimbolide blocks assembly without affecting ATP use, offering a distinct mode of action.
Testing nimbolide in lung and gut disease
The team next asked whether this molecular effect translates into real protection in disease models. In mice given a bacterial toxin to induce ARDS, nimbolide treatment before the insult reduced lung tissue damage, fluid buildup, and leakage of protein into the air spaces. There were fewer invading immune cells in the lungs and lower levels of IL‑1β in both lung tissue and blood. Similar benefits were seen in a model where the toxin was delivered directly into the airways. In a separate model of ulcerative colitis, where mice drank a chemical that injures the colon, nimbolide limited weight loss, improved stool consistency and bleeding scores, preserved colon length, and reduced microscopic signs of inflammation and tissue erosion, along with lowering IL‑1β in the colon. Crucially, in genetically engineered mice lacking NLRP3, disease was already milder and nimbolide no longer provided added benefit, underscoring that its protective effects depend on blocking NLRP3.
What this could mean for future treatments
For non‑specialists, the key lesson is that nimbolide acts like a smart brake on one of the body’s most powerful inflammatory switches. By latching onto a precise site on NLRP3 and preventing the inflammasome from snapping together, it calms excessive inflammation in both the lungs and the gut without broadly shutting down immune defenses. While nimbolide itself will still need improvement in areas like stability and delivery before it can become a medicine, this work positions it as a promising starting point—and a blueprint—for designing next‑generation drugs against NLRP3‑driven diseases such as ARDS and ulcerative colitis.
Citation: Xu, H., Lin, Y., Luo, W. et al. Nimbolide ameliorates ARDS and ulcerative colitis by disrupting NLRP3 inflammasome activation. Commun Biol 9, 246 (2026). https://doi.org/10.1038/s42003-026-09524-y
Keywords: nimbolide, NLRP3 inflammasome, inflammation, acute respiratory distress syndrome, ulcerative colitis