Clear Sky Science · en
INSIG1 parallel substitution drives lipid/sterol metabolic plasticity mediating desert adaptation in ungulates
How Desert Giants Beat Heat and Hunger
Camels and desert antelopes live where rain is rare, food is patchy, and summer heat can be deadly. Yet they endure months of thirst and hunger that would kill most mammals. This study asks a simple question with far‑reaching implications: what in their DNA and metabolism lets these animals survive such extremes—and can those secrets help us prepare livestock and even human health for a hotter, drier future?
Reading the Camel’s Genetic Blueprint
To probe this mystery, researchers first built the most detailed genetic map to date of the domestic Bactrian camel, the familiar two‑humped work animal of Central Asian deserts. Using long‑read DNA sequencing and 3D “Hi‑C” mapping, they assembled its genome into full chromosomes, capturing about 2.4 billion DNA letters with high accuracy. This new reference genome outperforms previous camel genomes, with longer continuous stretches of DNA and more complete coverage of genes, especially around hard‑to‑assemble regions like chromosome ends and centers. 
Comparing Desert Dwellers and Their Relatives
The team then examined the genomes of 22 hoofed mammals, including desert specialists like camels and Hippotraginae antelopes (addax, scimitar‑horned oryx, gemsbok), as well as non‑desert species such as cattle and sheep. These lineages split from each other roughly 15–16 million years ago, yet desert forms independently evolved similar traits: the ability to let body temperature fluctuate, conserve water, and rely heavily on fat as a fuel and water source. By tracking changes in over 12,000 shared genes across the evolutionary tree, the scientists found that desert lineages show faster rates of molecular evolution and a higher share of genes under strong positive selection—signs of intense adaptation pressure from heat, drought, and limited food.
Energy and Salt: Core Survival Themes
Genes that changed in parallel across desert species clustered in a few key job categories. Many help manage energy balance, including how fat tissue develops, how the body maintains steady blood sugar, and how cholesterol is handled. Others govern the flow of ions like calcium and potassium, which are crucial for heart function, blood pressure, and kidney performance—systems that are pushed to their limits when water is scarce. Notably, the team found that specific genes carried matching amino‑acid changes in both camel and antelope lineages, even though these groups are only distantly related. Statistical tests suggest such repeated changes are unlikely to be random, pointing instead to shared solutions to life without reliable water.
Flexible Fat and Cholesterol as a Desert Tool
Digging deeper, the researchers uncovered a striking pattern: many of the convergent changes occurred in genes that control fats and sterols, especially cholesterol. Desert ungulates showed parallel tweaks in proteins involved in cholesterol production and handling, including a regulatory gene called INSIG1. Blood chemistry told a matching story. When the team compared blood from fasting camels and fasting mice, camels had a higher proportion of fatty‑acid‑related molecules and a lower proportion of steroid‑type lipids such as cholesterol. This suggests desert animals are wired to store fat efficiently when food is available, yet quickly tap those reserves—and their “metabolic water”—during prolonged fasting and dehydration. 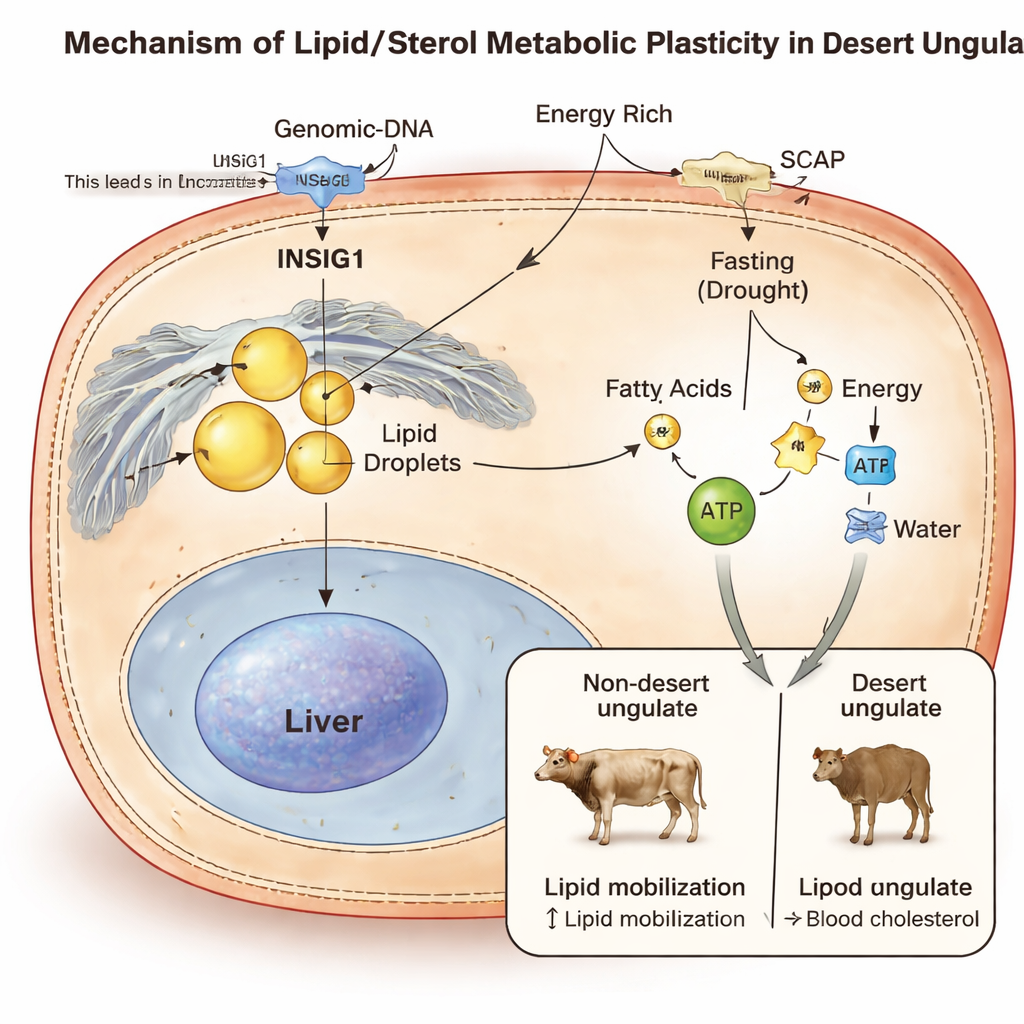
A Single Mutation with Big Consequences
INSIG1 emerged as a key player. This gene acts as a brake on cholesterol and fat synthesis by controlling a partner protein called SCAP. In both camels and desert antelopes, the team found the same amino‑acid change in a crucial part of INSIG1. Lab experiments in human liver‑like cells showed that this desert‑style version of INSIG1 binds SCAP less effectively, loosening the brake and allowing more fat to build up under rich‑energy conditions. At the same time, it altered how cells take up cholesterol from the bloodstream. To test the effect inside a living animal, the researchers engineered mice carrying the equivalent INSIG1 mutation. These mice showed shifts in the activity of many genes involved in fat breakdown and cholesterol handling, as well as measurable changes in liver metabolites, consistent with faster mobilization of stored lipids during fasting.
What This Means for Animals and People
Put together, the results highlight “lipid and sterol plasticity”—the ability to flexibly switch between storing and burning fat and managing cholesterol—as a cornerstone of desert survival in large mammals. Rather than a single “desert gene,” the study reveals a network of changes that let animals bank energy in good times and efficiently transform it into fuel and water when the environment turns hostile. Understanding these natural strategies could inform selective breeding of drought‑resistant livestock and inspire new treatments for human metabolic disorders, from obesity to fatty liver disease, as societies grapple with the health challenges of a warming, drying world.
Citation: Li, X., He, Z., Liu, A. et al. INSIG1 parallel substitution drives lipid/sterol metabolic plasticity mediating desert adaptation in ungulates. Commun Biol 9, 245 (2026). https://doi.org/10.1038/s42003-026-09523-z
Keywords: desert adaptation, camel genomics, lipid metabolism, cholesterol regulation, INSIG1