Clear Sky Science · en
Long-term exposure to the ethanol-derived metabolite acetaldehyde elevates structural genomic alterations but not base substitutions
Why alcohol’s hidden by-product matters
Many people know that heavy drinking raises the risk of cancers of the mouth, throat, stomach and breast, but it has been unclear exactly how alcohol does this damage to our DNA. This study zooms in on acetaldehyde, a highly reactive chemical that our bodies make when we process alcohol, and that is also present in cigarette smoke and some foods. Using sensitive whole-genome sequencing and cancer data from patients, the authors show that acetaldehyde does not spray the genome with countless tiny typos, as many assumed. Instead, it quietly increases larger cuts and rearrangements in our chromosomes—changes that may help healthy cells turn cancerous.
A closer look at everyday exposure
Acetaldehyde is classified as a proven human carcinogen. After a drink, especially strong alcoholic beverages, its levels can briefly reach cancer‑relevant concentrations in the mouth and esophagus, and smaller amounts circulate in the blood. Some people, particularly in East Asia, carry variants of the ALDH2 gene that slow acetaldehyde breakdown, causing it to build up even more. Yet in laboratory tests it has been hard to pin down exactly how acetaldehyde alters DNA, partly because it evaporates so quickly from open cell cultures. To overcome this, the researchers built an enclosed system that keeps acetaldehyde at stable, realistic levels similar to those seen in human tissues after drinking.
Long-term exposure without a storm of small mutations
The team exposed four types of human cells—including blood cells, breast cells and two esophageal cancer cell lines—to 100 micromolar acetaldehyde for 30 days, a dose that stressed the cells but still allowed them to grow. They then sequenced the entire genomes of individual descendant cells and compared them with untreated controls. Across all cell types and under normal or reduced oxygen conditions, acetaldehyde did not increase the number of “single-letter” DNA changes or small insertions and deletions. Nor did it create the known alcohol-linked mutation pattern, called SBS16, that has been seen at low levels in some human tumors. In other words, chronic acetaldehyde exposure did not behave like a classic chemical mutagen that litters the genome with tiny spelling errors.
Big hits to chromosomes instead of tiny typos
Although small-scale mutations barely changed, the architecture of the genome told a different story. The researchers found more structural variations—large deletions and duplications of stretches of DNA up to around one million letters long—in most acetaldehyde-treated cell lines. Many of these deletions bore short matching DNA sequences at their junctions, a hallmark of repair by non-homologous end joining, a quick but error-prone way for cells to glue broken DNA ends together. In parallel experiments, the authors saw more sister chromatid exchanges, where matching chromosomes swap segments, and detected direct evidence of DNA breaks and activation of DNA damage signals inside the cell. Together, these results point to acetaldehyde as a trigger for chromosome breaks that sometimes get mis-repaired, reshaping the genome.
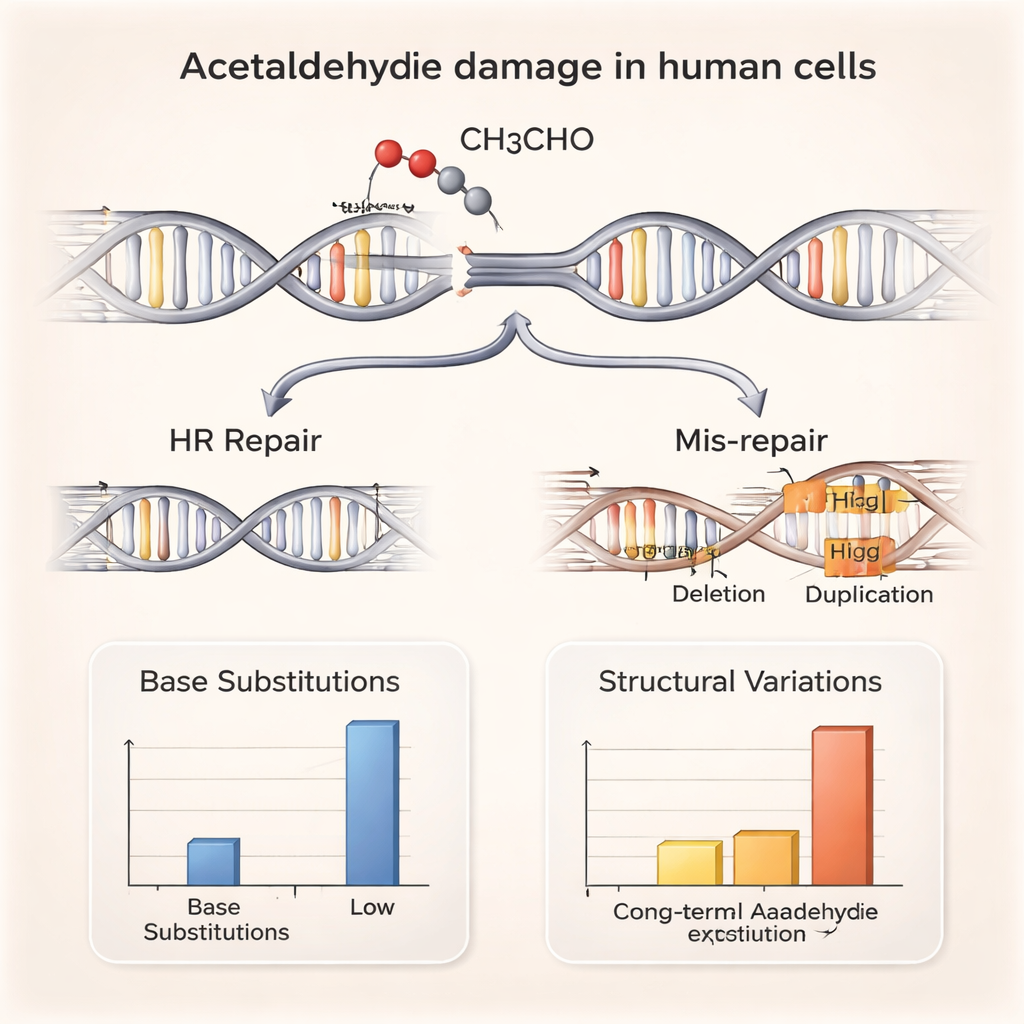
How cells cope with the damage—and when they cannot
To understand how cells survive this onslaught, the team tested mutant cell lines lacking specific DNA repair systems. Cells missing key proteins involved in homologous recombination, a high-fidelity pathway that uses an intact copy of DNA as a template to repair breaks, were especially sensitive to acetaldehyde. In contrast, cells defective in several other repair processes, including classical end-joining and nucleotide excision repair, were not particularly vulnerable. This pattern suggests that many acetaldehyde-induced breaks arise at stalled or collapsed DNA replication forks, structures that normally depend on homologous recombination for safe repair. When this system is weakened—as in people carrying harmful BRCA1 or BRCA2 variants—acetaldehyde damage may be harder to manage, potentially compounding cancer risk.
Evidence from real tumors
The researchers then turned to genome data from 170 gastric cancers in Japan, where detailed histories of alcohol use were available. They saw that tumors from drinkers carried significantly more medium-sized deletions and duplications, roughly between 32,000 and 1 million DNA letters—exactly the size range that increased in their acetaldehyde-treated cells. A similar pattern did not appear in an esophageal cancer type that is not strongly linked to alcohol. While smoking can also contribute to such structural variations, the close match between the laboratory and patient data supports the idea that alcohol-derived acetaldehyde fosters this particular form of genomic scarring during cancer development.
What this means for drinkers
For non-specialists, the key message is that alcohol’s danger may lie less in causing countless tiny DNA misprints and more in its ability to produce acetaldehyde, which occasionally snaps and rearranges large chunks of our chromosomes. These structural changes can disrupt or amplify important genes that control cell growth, nudging cells toward cancer over years of repeated exposure. The work does not argue that any single drink will trigger cancer, but it sharpens our understanding of how regular alcohol use—and factors like ALDH2 or BRCA mutations—can interact at the DNA level. By revealing a specific pattern of large-scale damage rather than a flood of small mutations, the study helps explain why alcohol is carcinogenic and may guide future efforts to identify and protect those at highest risk.
Citation: Lózsa, R., Szikriszt, B., Németh, E. et al. Long-term exposure to the ethanol-derived metabolite acetaldehyde elevates structural genomic alterations but not base substitutions. Commun Biol 9, 243 (2026). https://doi.org/10.1038/s42003-026-09521-1
Keywords: acetaldehyde, alcohol and cancer, DNA damage, genome rearrangements, homologous recombination