Clear Sky Science · en
MAT2A enhances PARN transcription via SRF to accelerate glycolysis and drive malignant progression in osteosarcoma
Why this bone cancer study matters
Osteosarcoma is the most common bone cancer in children and teenagers, and treatments have barely changed in decades. Many young patients still face poor outcomes, especially if the cancer spreads or resists chemotherapy. This study uncovers a hidden "master switch" protein called MAT2A that helps osteosarcoma cells grow, rewire their energy use, and form aggressive tumors—and shows that a drug aimed at MAT2A can slow or shrink these tumors in lab models.
A hidden engine inside tumor cells
The researchers began by comparing bone cancer samples with normal bone tissue at the single-cell level. They found that osteosarcoma tissue contains more tumor-building cells and fewer immune cells, and that one gene in particular—MAT2A—was unusually active in cancer cells. Analyses of public cancer databases and lab-grown cell lines confirmed that MAT2A levels were much higher in osteosarcoma than in healthy bone cells, and that patients with more MAT2A tended to have more advanced disease and lymph node spread. This placed MAT2A at the center of attention as a potential driver of the disease rather than a passive bystander.
Turning down MAT2A slows cancer growth
To test how important MAT2A really is for tumor behavior, the team used genetic tools to lower MAT2A levels in osteosarcoma cell lines. When MAT2A was silenced, cancer cells divided more slowly and were more likely to undergo programmed cell death. In mice implanted with these modified cells, tumors grew much more slowly and ended up significantly smaller, even though the animals’ body weight remained stable. These experiments showed that osteosarcoma cells are heavily reliant on MAT2A for both survival and their ability to form tumors.
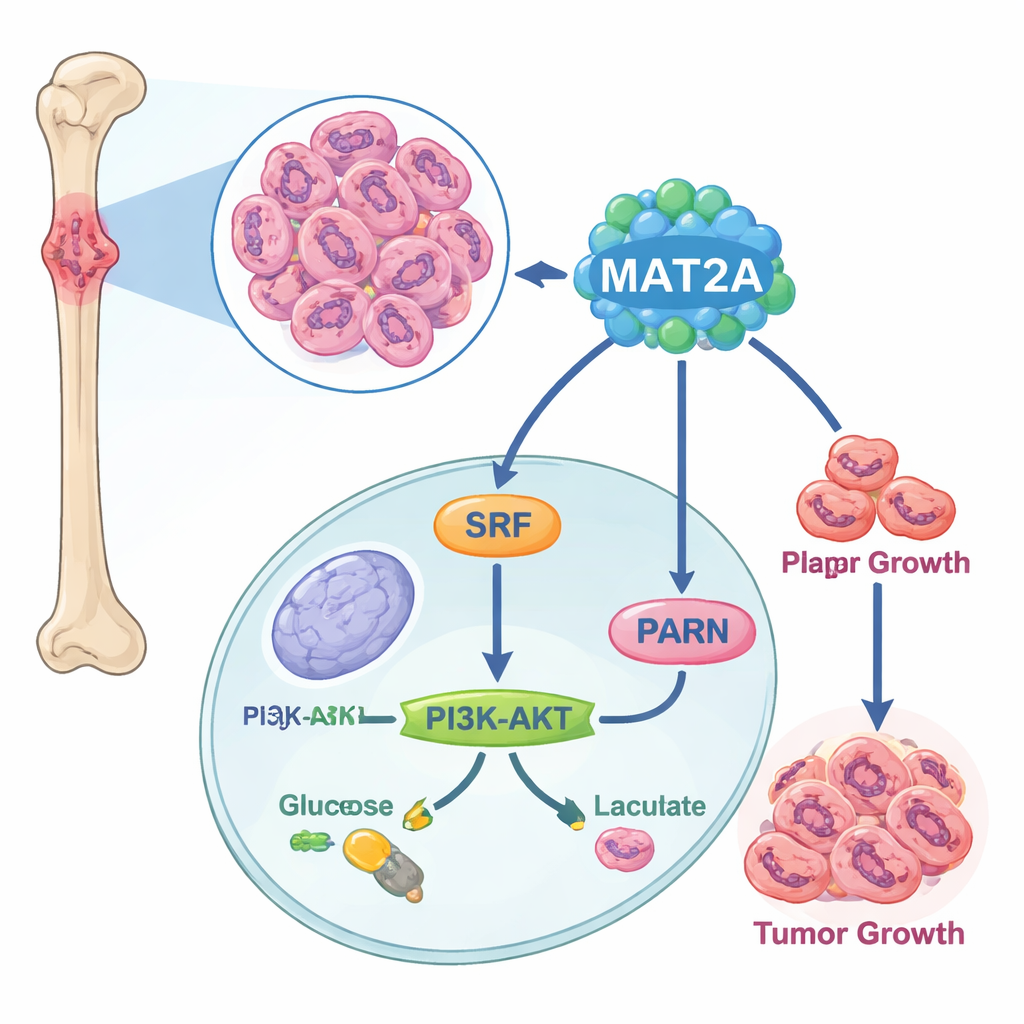
A signal relay that boosts cell fuel use
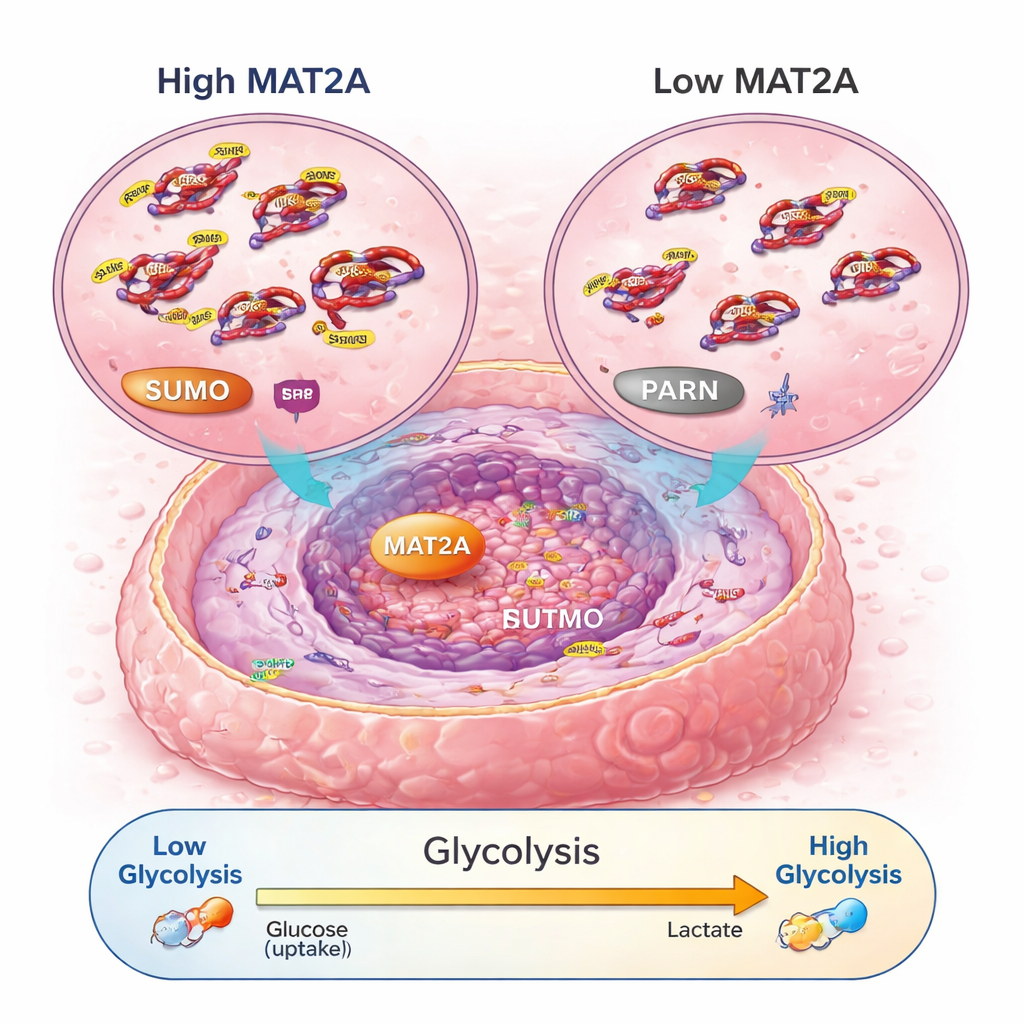
Digging deeper, the scientists discovered that MAT2A does more than perform its usual metabolic job. Inside the nucleus, it binds to a transcription factor called SRF, helping keep SRF stable and active by promoting a chemical tag known as SUMO. In turn, SRF switches on a gene called PARN, which then activates the well-known PI3K–AKT pathway and turns up the machinery of aerobic glycolysis—a fast but inefficient way for cells to make energy that cancer cells favor. When MAT2A was reduced, cancer cells made less ATP (their energy currency), took up less glucose, produced less lactic acid, and shifted back toward calmer, oxygen-based respiration. Disrupting PARN or AKT could undo the pro-growth and pro-glycolysis effects of MAT2A, confirming that this relay—MAT2A to SRF to PARN to PI3K–AKT—acts as a key energy and growth circuit in osteosarcoma.
A nontraditional role and a druggable target
Strikingly, a version of MAT2A that lacked its normal enzyme activity still supported SRF stability and PARN activation, meaning this cancer-promoting role does not depend on its usual chemical reaction. Instead, MAT2A acts more like a scaffold that organizes other proteins. This has practical implications: drugs do not necessarily need to block its catalytic center to weaken tumors. The researchers tested a small-molecule inhibitor called FIDAS-5, designed to hit MAT2A, in both cell cultures and mouse models. Treatment reduced nuclear SRF, lowered PARN and PI3K–AKT signaling, dampened glycolysis, slowed cell growth, and triggered cancer cell death. In mice, FIDAS-5 shrank or slowed tumors without noticeable weight loss, suggesting tolerable side effects in this setting.
What this means for patients and future therapies
For non-specialists, the core message is that osteosarcoma cells depend on MAT2A to keep their growth programs and sugar-burning engines running at full speed. By stabilizing key control proteins and boosting a chain of signals, MAT2A helps tumors grow faster and become more aggressive. Blocking this protein—either by gene-based methods or a drug like FIDAS-5—cuts off that support, forcing cancer cells to slow down, burn energy less frantically, and ultimately die. While much work remains before such treatments reach the clinic, this study positions MAT2A as a promising new target in the fight against bone cancer, opening a potential path to more precise and less toxic therapies for young patients.
Citation: Ren, Z., Chen, H., Qiao, Q. et al. MAT2A enhances PARN transcription via SRF to accelerate glycolysis and drive malignant progression in osteosarcoma. Commun Biol 9, 241 (2026). https://doi.org/10.1038/s42003-026-09518-w
Keywords: osteosarcoma, MAT2A, cancer metabolism, PI3K AKT pathway, targeted therapy