Clear Sky Science · en
Single-cell analysis reveals multi-faceted features of B cell development, together with age-associated B cell subpopulations
How Our Body’s Antibody Factories Change with Age
B cells are the body’s antibody factories, defending us from infections and helping vaccines work. But B cells are not all the same: they develop through many stages and live in different tissues, and they also change as we age. This study uses powerful single-cell techniques to map B cells one by one in human bone marrow, blood, and tonsils, revealing how they grow up, talk to neighboring cells, and how special age-linked B cells may quietly fuel inflammation in older adults.
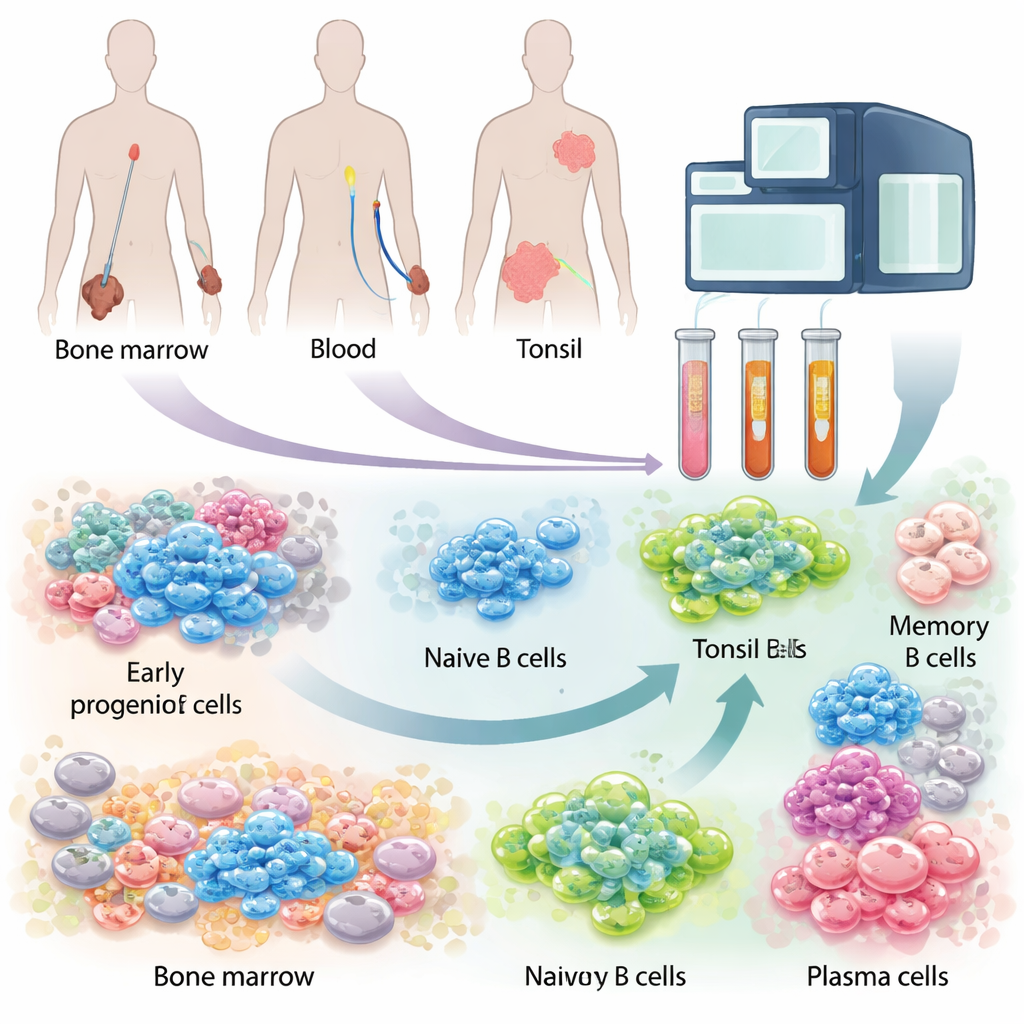
Following B Cells from Birth to Battle
The researchers combined several large datasets and new experiments to analyze nearly 200,000 individual cells. They focused on 18 distinct kinds of B cells, from the earliest precursors in bone marrow to fully armed antibody-secreting plasma cells. By reading which genes were switched on in each single cell, and which antibodies each B cell could make, they traced how cells move along the developmental path. They also examined the “neighbors” of B cells—such as myeloid cells and bone marrow stromal cells—to understand the support network that shapes B cell fates.
Quiet Middle Childhood for B Cells
One surprise was that immature and naïve B cells—cells that have not yet met their target germs—are unusually quiet. Using a method that looks at how fast genes turn on and off over time, the team found that early progenitor cells and late-stage plasma cells have very active gene programs, while immature and naïve B cells show low activity and few genes in motion. This suggests that B cells pass through a calm, low-metabolism “holding pattern” after completing key DNA rearrangements, before they leave the bone marrow and join the circulating pool that can respond to future infections.
Hidden Diversity in “Simple” B Cells
Though naïve B cells are often treated as one uniform group, the study uncovered specialized subsets. Some naïve cells showed signs of low-level homeostatic proliferation—gentle, self-renewing division that helps maintain numbers without obvious infection. Others showed stress-response signatures and early hints of antibody class switching, especially in the tonsil environment. For memory B cells, which remember past encounters, the authors found evidence for two major developmental routes: one that depends on help from T cells in classic germinal centers, and another more independent path. These two models, long debated, appear to coexist and may explain why memory B cells vary so widely in how they respond to new challenges.
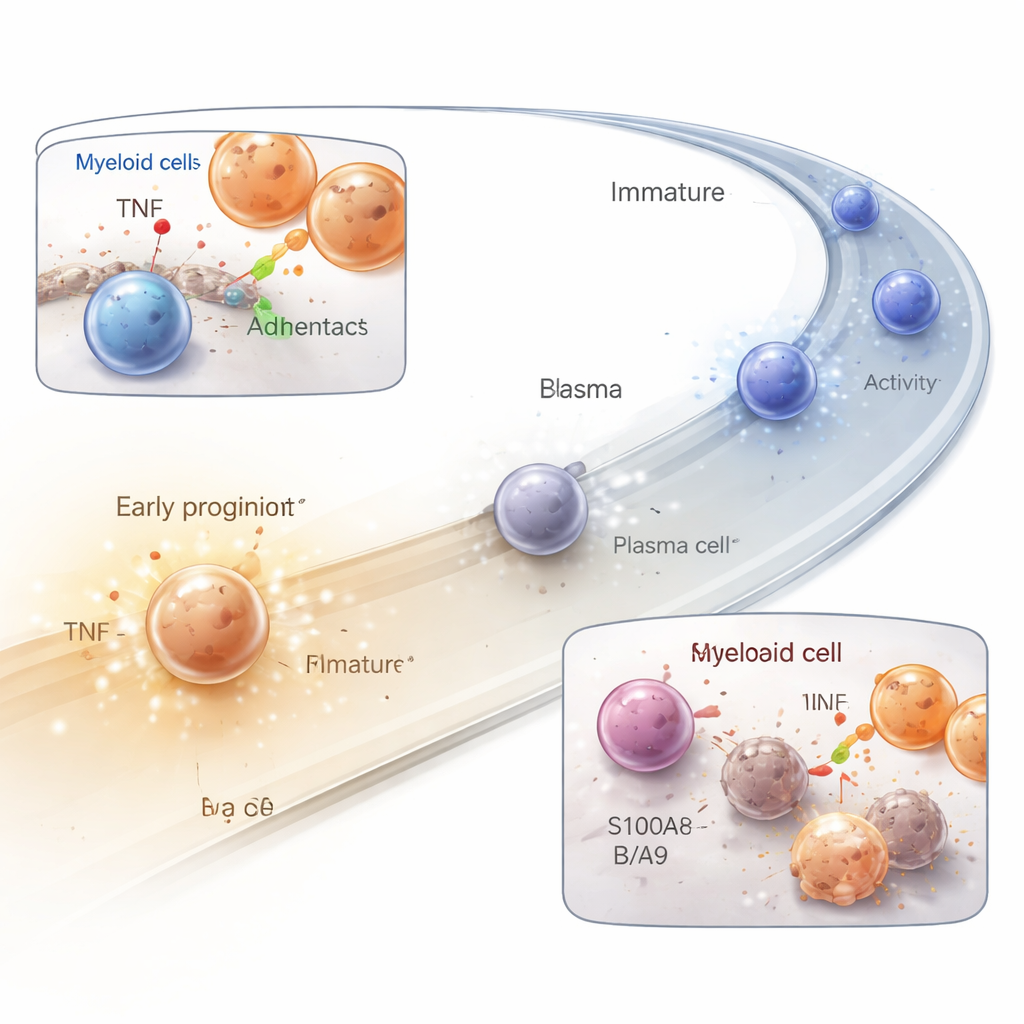
Cross-Talk with Helper Cells in the Neighborhood
B cells do not act alone. By systematically scanning for matching pairs of signaling molecules and receptors, the authors built interaction maps between B cells and surrounding cell types. In bone marrow, stromal cells formed a dense web of contacts that help nurture early B cells, while myeloid cells—such as dendritic cells and monocytes—emerged as key partners across bone marrow, tonsil germinal centers, and blood. Signals from the TNF family and various adhesion molecules dominated this cross-talk, changing in a stage-specific way as B cells matured. This pattern highlights how physical contact and inflammatory cues together steer whether B cells survive, divide, or die.
Age-Linked B Cells That May Drive “Inflammaging”
The team also discovered two unusual B cell subsets that appear mainly in older adults. One group of immature-like B cells produced S100A8 and S100A9, proteins normally made by myeloid cells and associated with chronic inflammation and aging. Another expressed C1q, a component of the complement system that rises with age. Follow-up experiments showed that human B cells can secrete S100A8/A9 into their surroundings, and mouse studies linked higher levels of these molecules in B cells to older age, especially in females. These findings suggest that certain B cells themselves may become sources of inflammatory factors, contributing to the low-grade, long-term inflammation often seen in aging.
What This Means for Health and Aging
By charting B cells across tissues and life stages at single-cell resolution, this work provides a detailed atlas of how our antibody system is built, maintained, and altered with age. For non-specialists, the main takeaway is that B cells are far more diverse and dynamic than once thought, and that specific B cell subsets in older individuals may help drive age-related inflammation. Understanding these pathways and cell-to-cell conversations could aid in designing better vaccines for the elderly, diagnosing immune aging earlier, and developing therapies that tame harmful inflammatory B cell states while preserving essential protection against infection.
Citation: Yang, X., Tang, H., Lan, C. et al. Single-cell analysis reveals multi-faceted features of B cell development, together with age-associated B cell subpopulations. Commun Biol 9, 240 (2026). https://doi.org/10.1038/s42003-026-09515-z
Keywords: B cell development, single-cell sequencing, immune aging, memory B cells, bone marrow microenvironment