Clear Sky Science · en
Linking molecular tension and cellular tractions: a multiscale approach to focal adhesion mechanics
How Cells Feel and Pull on Their Surroundings
Every time a cell moves, divides, or reshapes a tissue, it is quietly tugging on its surroundings. These tiny mechanical pulls are crucial in wound healing, cancer spread, and how organs develop, yet they are hard to see and even harder to measure. This study presents a new way to look at those forces from two angles at once: how hard an entire cell pulls on a soft material, and how much force individual “molecular springs” inside the cell’s anchoring sites carry. By linking these views, the work helps explain how cells sense stiffness and regulate their grip on the world around them.
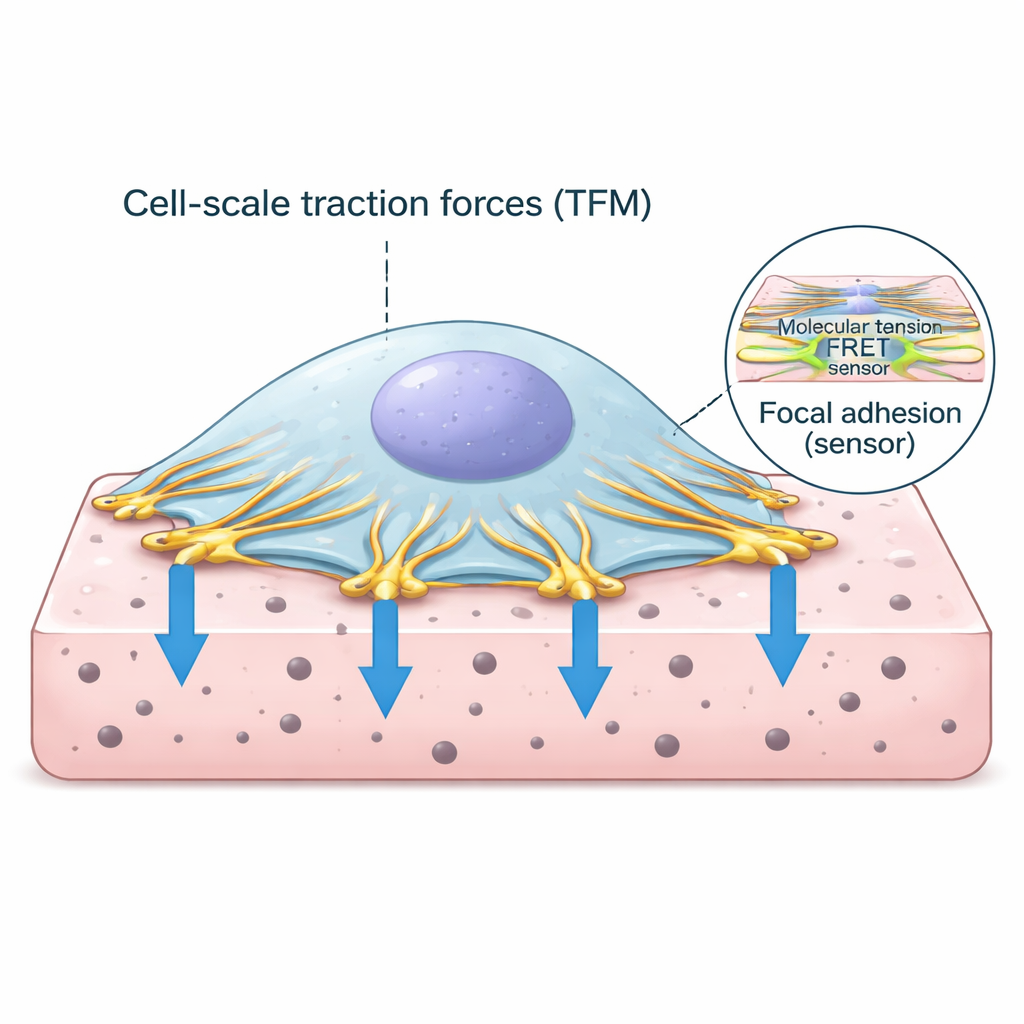
The Cell’s Grip Points on the Outside World
Cells do not simply float in tissues; they anchor themselves through specialized contact points called focal adhesions. At these spots, the cell’s internal scaffolding of actin fibers connects to proteins that span the cell membrane and latch onto the surrounding matrix. One of the key proteins in this anchor complex is vinculin, which behaves like a force-sensitive link. When the cell’s contractile machinery pulls on actin, vinculin feels the load and helps strengthen the connection. Understanding how much force passes through these links, and how that relates to the overall pulling of the cell, is central to decoding how tissues stay healthy or become diseased.
Two Windows Into Cellular Force
The researchers combined two powerful techniques into a single workflow. First, traction force microscopy measures how much a cell deforms a soft, gel-like substrate by tracking the motion of tiny fluorescent beads in the gel. From these bead movements, one can calculate the distribution of push-and-pull forces under the cell. Second, a specially engineered vinculin protein carries a fluorescent tension sensor that changes its light signal when it is stretched. Using advanced lifetime imaging, the team converted this light signal into a readout of molecular tension on vinculin. They designed thin, flat hydrogels that could be imaged at high resolution and wrote custom software to align, segment, and analyze both datasets down to individual adhesion sites.
How Stiffness Changes the Cell’s Effort
When cells were grown on soft versus stiffer gels, their overall behavior changed in a clear way. On stiffer substrates, cells spread out more and exerted stronger traction forces on the gel surface. At the same time, the fluorescent readout from the vinculin sensor indicated higher molecular tension inside focal adhesions. Interestingly, the basic structural features of these adhesions—such as how many there were or their average size—did not change much between soft and stiff gels. Instead, the way forces were organized did. Large, radially oriented adhesions with more vinculin tended to carry higher tractions, suggesting that both the geometry and molecular makeup of these sites help set how hard the cell pulls.
A Complex Relationship Between Local and Molecular Forces
Looking more closely at individual adhesions revealed that the link between local traction and vinculin tension is not one-size-fits-all. In some cells, adhesions that generated higher tractions also showed higher vinculin tension, implying that engaged, load-bearing contacts share force more directly across their molecular links. In other cells, the opposite pattern appeared: regions of strong traction were associated with lower vinculin tension, while other adhesions bore more molecular load without producing large outward pulls. Many cells showed no clear pattern at all. These distinct behaviors likely reflect different cell states—such as active spreading, stable adhesion, or retraction—and hint that cells can redistribute forces across their adhesion network in multiple ways.
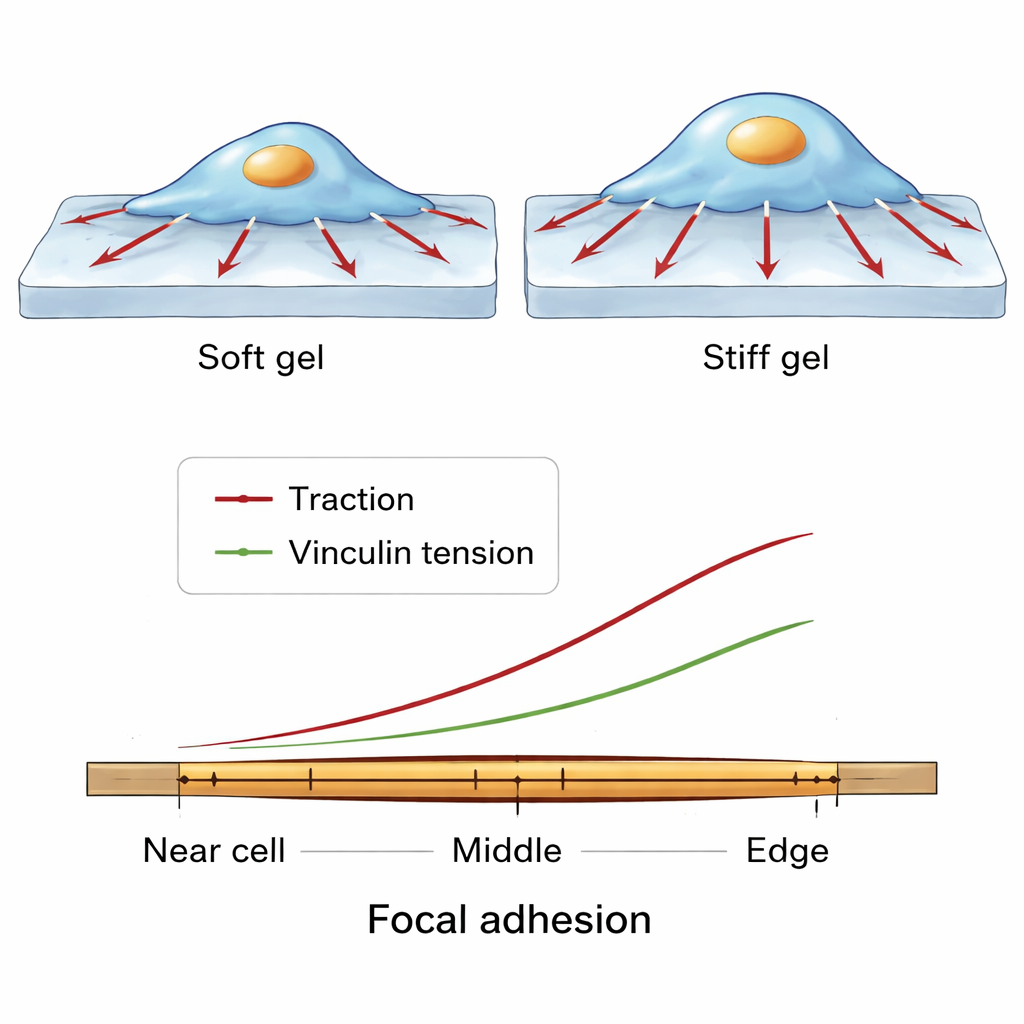
Fine-Scale Force Patterns Along a Single Grip Point
The team then zoomed in even further, examining how forces change along the length of a single focal adhesion, from the side closer to the cell center to the edge near the cell perimeter. Across many cells and conditions, a consistent pattern emerged. Vinculin molecules were most densely packed toward the middle of the adhesion. However, both the traction exerted on the substrate and the molecular tension on vinculin rose toward the outer, more peripheral end. This suggests a balancing act: in central regions, many vinculin molecules can share the load, so each one feels less tension, whereas at the outer edge fewer molecules carry relatively more force, supporting strong local pulling while keeping the adhesion intact.
What This Means for Health and Disease
By merging whole-cell traction maps with protein-level tension measurements, this study offers a multiscale picture of how cells manage their mechanical grip. The work shows that as the environment stiffens, cells both pull harder and load their vinculin links more, but that the detailed relationship between outward forces and molecular tension varies from adhesion to adhesion and from cell to cell. At the same time, a robust spatial pattern of force distribution within individual adhesions appears to be conserved. For a non-specialist, the key message is that cells finely tune both where and how they pull, redistributing force across many small molecular “springs” to adapt to different mechanical environments—a principle that may underlie processes as diverse as tissue development, fibrosis, and cancer invasion.
Citation: Aytekin, S., Kimps, L., Coucke, Q. et al. Linking molecular tension and cellular tractions: a multiscale approach to focal adhesion mechanics. Commun Biol 9, 236 (2026). https://doi.org/10.1038/s42003-026-09514-0
Keywords: cell mechanics, focal adhesions, vinculin, traction force microscopy, mechanotransduction