Clear Sky Science · en
Temporal dynamics of collateral RNA cleavage by LbuCas13a in human cells
Turning a Bacterial Defense into a Precision Cell Killer
Researchers have discovered a way to turn a bacterial immune protein into a highly selective “self-destruct” switch for human cells. By harnessing a CRISPR enzyme called LbuCas13a, they can recognize a chosen RNA message inside a cell and then shred almost all of that cell’s other RNA. For lay readers, this is intriguing because RNA messages control which proteins a cell makes; learning to erase them on command opens doors to new treatments for cancer, antiviral strategies, and powerful research tools.
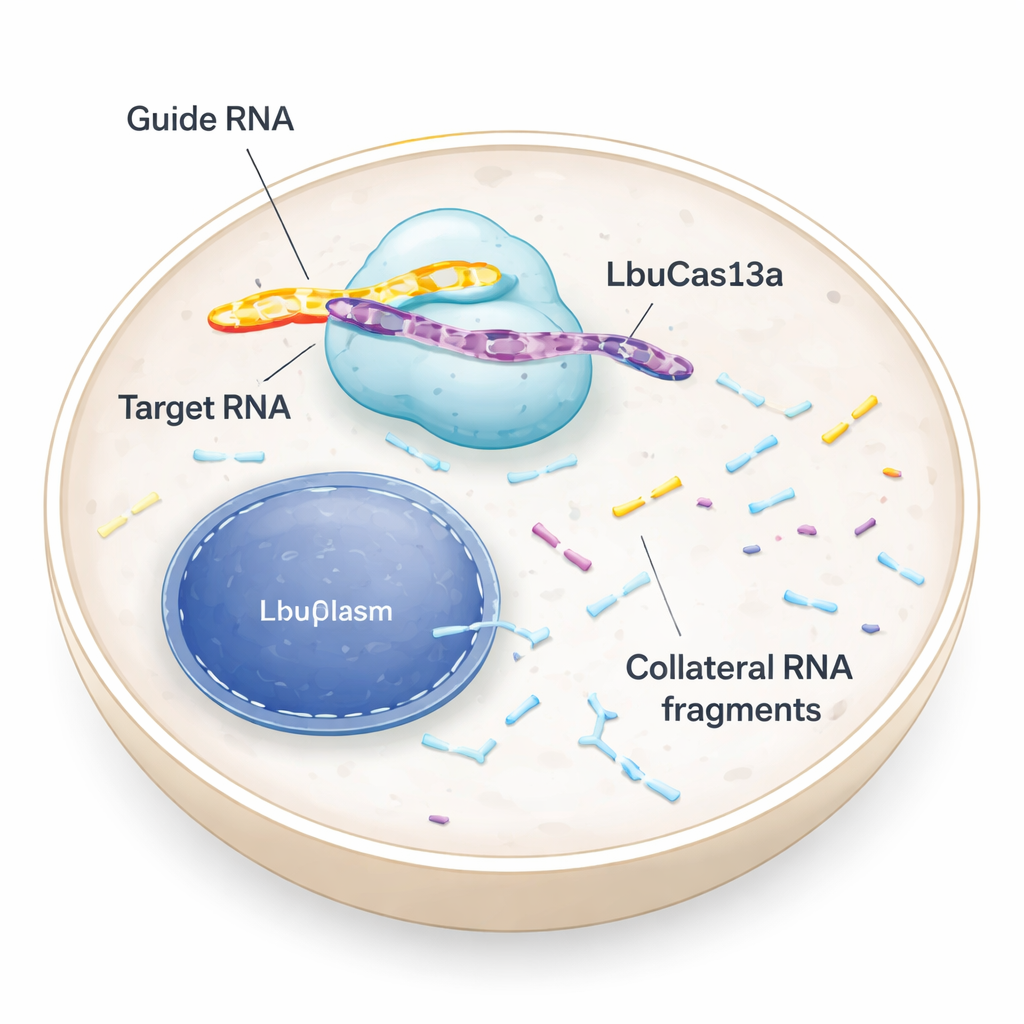
A Molecular Scissor That Targets RNA, Not DNA
Most people have heard of CRISPR tools that cut DNA, rewriting the genetic code. LbuCas13a is different: it recognizes and cuts RNA, the temporary messages that carry instructions from DNA to the cell’s protein factories. In bacteria, Cas13 enzymes are part of an antiviral defense. Once they spot a viral RNA, they not only slice up that invader but also start cutting many other RNAs nearby. This so‑called “collateral” activity can push infected cells into dormancy or death, helping protect the bacterial community. Early tests in animal and human cells suggested that Cas13’s collateral cutting was weak or absent, so the enzyme was mainly used as a precise RNA “off switch.” The new study revisits that assumption and shows that, under the right conditions, collateral activity in human cells can be both strong and useful.
Unleashing Collateral Cutting in Human Cells
The team compared several Cas13 variants and found that LbuCas13a was especially potent. They delivered purified LbuCas13a protein pre‑loaded with a short guide RNA directly into human cells, a form of molecular “protein injection” called ribonucleoprotein (RNP) delivery. When the guide matched a target RNA—such as a fluorescent marker gene or abundant natural messages like GAPDH and 18S ribosomal RNA—the enzyme first sliced that target and then began chewing up many other RNAs. Within about 50 minutes, the overall RNA profile of the cell changed dramatically, with distinctive cleavage fragments appearing. This collateral effect was seen with different delivery methods and in a range of cell types, showing that it was not a quirk of one cell line or one artificial target.
From RNA Shredding to Cell Death and Selection
What happens to a cell whose RNA messages suddenly disappear? Using live‑cell imaging, the researchers observed that cells expressing the target RNA gradually entered apoptosis, a tidy form of programmed cell death marked by characteristic “early warning” signals before the cell breaks apart. Importantly, neighboring cells that did not express the target RNA remained mostly unaffected, demonstrating that the self‑destruct is specific. The group then exploited this property as a selection tool. When they mixed target‑bearing cells with normal cells and activated LbuCas13a, the target‑bearing cells were selectively depleted over several days. Repeating the treatment multiple times pushed their fraction even lower. They showed this could enrich for successfully gene‑edited cells and could also be tuned to attack cancer cells that overproduce an oncogene, here CDK4, while sparing related cells that make much less of that RNA.
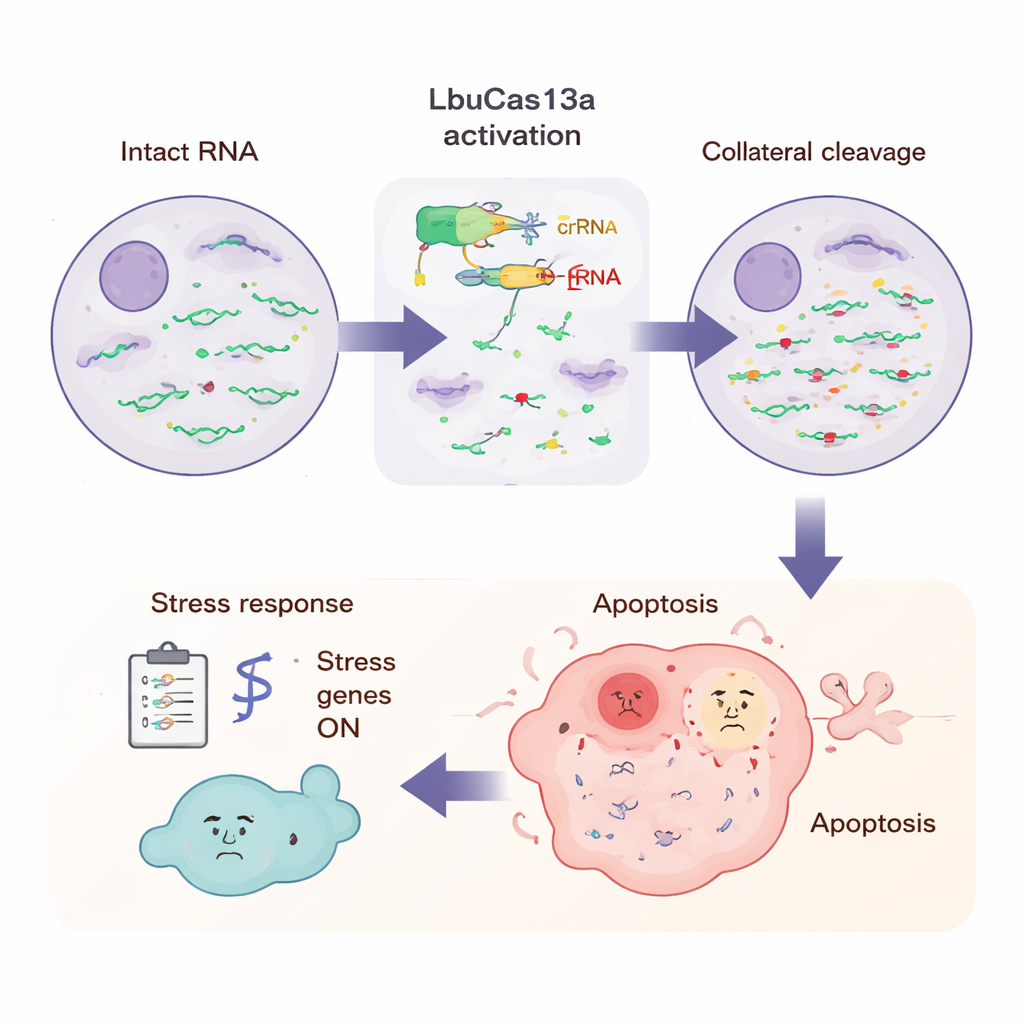
What the Cell Experiences During the Attack
To see the bigger picture, the scientists measured all RNAs in the cell at several time points after LbuCas13a activation. By spiking in known RNA standards, they could tell that most protein‑coding RNAs in the cytoplasm dropped by more than half within a few hours, whereas certain RNAs—such as mitochondrial messages and some nuclear non‑coding RNAs—were largely spared. Long‑read sequencing revealed that cutting happened at repeated, specific nucleotide positions, often in flexible loop regions of RNA rich in the base uracil, matching patterns seen in test‑tube experiments. At later time points, many stress and innate immune genes switched on, including those linked to inflammatory signals and antiviral defenses. This pattern suggests that the cell senses the sudden flood of broken RNA ends much like it would detect a viral infection, triggering an alarm program that culminates in apoptosis.
Why This Matters and Where It May Lead
In simple terms, the study shows that LbuCas13a can be turned into an RNA‑guided “kill switch” for cells: if a cell makes too much of a particular RNA, activating LbuCas13a against that RNA causes near‑global RNA loss, an immune‑like alarm, and then controlled cell death. Because the process depends strongly on how abundant the target RNA is, it could be used to eliminate cells that overexpress harmful genes—such as certain cancer cells—or to clean up unwanted cells in mixed cultures during research or cell therapy manufacturing. At the same time, the work is a cautionary tale: CRISPR tools aimed at RNA can have powerful side effects that must be carefully managed. Understanding when and how collateral RNA cleavage occurs is essential for safely using Cas13 technologies in medicine and biotechnology.
Citation: Bot, J.F., Zhao, Z., Li, M. et al. Temporal dynamics of collateral RNA cleavage by LbuCas13a in human cells. Commun Biol 9, 233 (2026). https://doi.org/10.1038/s42003-026-09511-3
Keywords: CRISPR-Cas13, RNA cleavage, cell death, cancer targeting, gene editing tools