Clear Sky Science · en
Multimodal single-cell protein and RNA profiling unveils dysregulated immature neutrophil dynamics in gestational diabetes mellitus
Why white blood cells matter in pregnancy
During pregnancy, a woman’s immune system must walk a tightrope: it has to defend against infection while also tolerating the growing baby, which is partly foreign to her body. This study looks at one key group of immune cells—neutrophils—to understand how their balance changes in healthy pregnancy and what goes wrong in gestational diabetes mellitus (GDM), a common form of high blood sugar that develops during pregnancy.
A closer look at pregnancy’s most common immune cells
Neutrophils are the most abundant white blood cells in our blood and are best known as rapid responders to microbes and tissue damage. For many years they were thought to be all alike, but newer tools have revealed that neutrophils actually come in multiple flavors and stages of maturity. Among these are so-called low-density neutrophils, which float with other immune cells when blood is separated in the lab. These low-density cells have been linked to autoimmune diseases, cancer, obesity, and infections, yet their roles in pregnancy—especially in women who develop GDM—have remained unclear.
Finding the “young” neutrophils
To map neutrophil types in pregnancy in fine detail, the researchers combined high-throughput antibody profiling (a technology called InfinityFlow) with single-cell RNA sequencing, which reads the activity of thousands of genes in individual cells. They compared blood from healthy pregnant women, women with GDM, and non-pregnant controls. By screening hundreds of surface proteins, they homed in on a trio of markers—CD10, CD49d, and Ig κ (a type of antibody fragment bound to the cell surface)—that cleanly separates immature from fully mature neutrophils. Cells that were CD10-negative but CD49d- and Ig κ–positive turned out to be “young” neutrophils with unsegmented nuclei and gene activity patterns typical of early development. These immature cells were found not only in the bloodstream but also in the maternal side of the placenta, hinting that they may help shape the local environment where mother and fetus meet. 
How neutrophil balance shifts in gestational diabetes
Using these markers, the team tracked how immature neutrophils change over pregnancy. In healthy pregnancies, the proportion of these young cells gradually increased from the end of the first trimester through mid-pregnancy, raising the ratio of immature to mature neutrophils in the blood. In women who developed GDM, this normal rise was sharply blunted: they had fewer immature neutrophils and a lower immature-to-mature ratio at multiple time points. The authors also found fewer circulating bone-marrow–derived precursor cells in GDM, suggesting that the bone marrow’s normal adjustment to pregnancy—the extra production and release of young neutrophils—was impaired.
From calm guardians to inflammatory responders
Gene activity profiles painted a functional picture of these cell types. Immature neutrophils in healthy pregnancy expressed genes linked to cell growth, energy production, and molecules that can suppress or fine-tune immune responses, while showing weaker signatures of inflammation. In contrast, mature neutrophils were primed for strong inflammatory action, with higher expression of alarm-signal genes and pathways responding to cytokines and interferons. In GDM, the remaining immature cells looked less “calm” and more activated: they dialed down cell-cycle programs and increased genes tied to inflammation and migration. A transitional “bridge” population, halfway between immature and mature, was more abundant in GDM, pointing to a skewed maturation process. 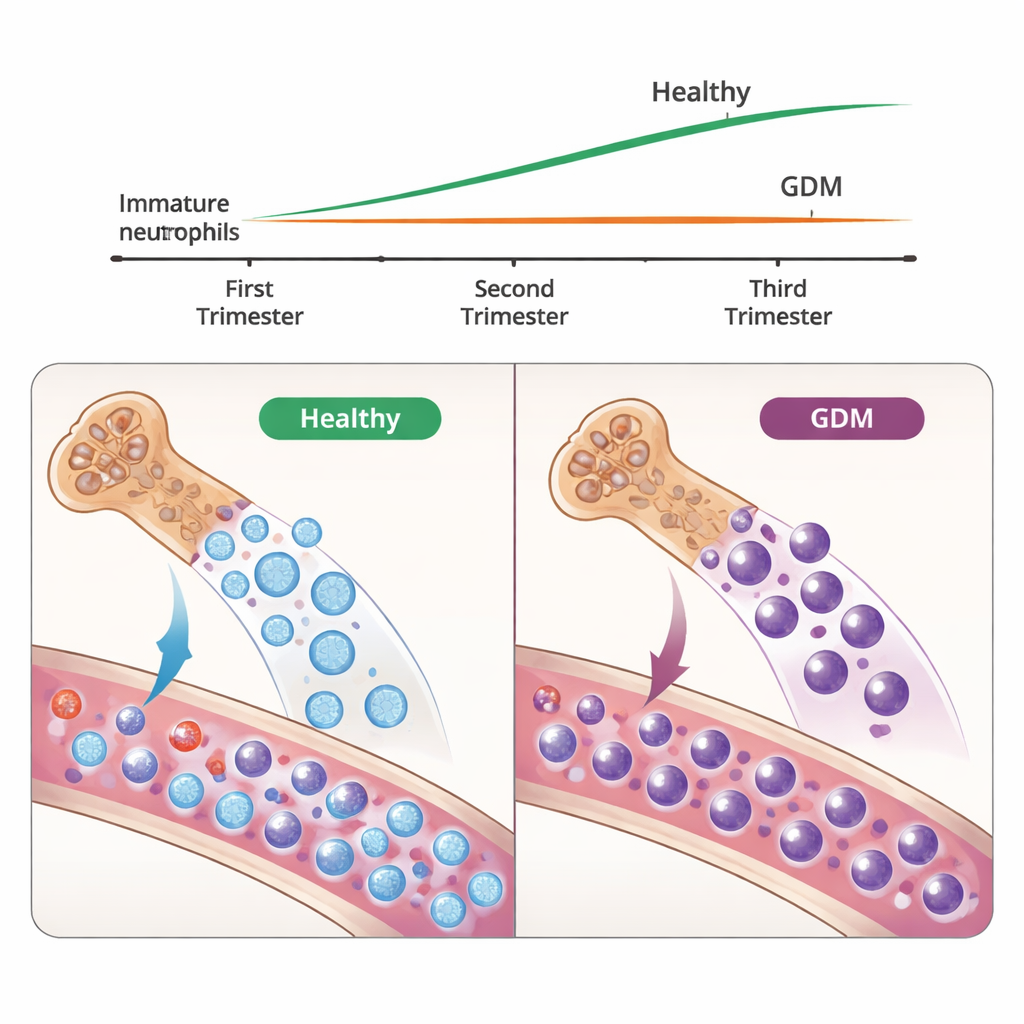
Linking immune imbalance to blood sugar control
When the researchers correlated neutrophil patterns with metabolic measurements, they found that women with lower immature-to-mature neutrophil ratios tended to have poorer blood sugar control and greater insulin resistance. This connection suggests that the immune system’s developmental balance is intertwined with metabolism during pregnancy. Rather than simply having “more inflammation,” GDM appears to involve a specific shortage of immunoregulatory, low-effector immature neutrophils and a tilt toward more inflammatory cell states. This distinct pattern sets GDM apart from other pregnancy complications, where neutrophils may be overly abundant or overly activated rather than developmentally delayed.
What this means for pregnant women
To a layperson, the take-home message is that not all white blood cells are equal, and their developmental stage matters. In a healthy pregnancy, the bone marrow releases extra young neutrophils that seem geared toward keeping the peace between mother and baby. In gestational diabetes, this “wave” of young cells is weaker, and the cells that do appear are more inflammatory. Because these shifts show up early and track with blood sugar and insulin resistance, carefully measuring neutrophil subsets using the CD10–CD49d–Ig κ combination could one day help flag women at higher risk of GDM or related complications, and may open the door to new strategies that support both immune balance and metabolic health.
Citation: Xu, J., Zhu, C., Xie, L. et al. Multimodal single-cell protein and RNA profiling unveils dysregulated immature neutrophil dynamics in gestational diabetes mellitus. Commun Biol 9, 316 (2026). https://doi.org/10.1038/s42003-025-09468-9
Keywords: gestational diabetes, pregnancy immunity, neutrophils, single-cell analysis, maternal metabolism