Clear Sky Science · en
Human class B1 GPCR modulation by plasma membrane lipids
Fats that fine-tune our cell's message receivers
Our cells rely on tiny protein antennas, called receptors, to sense hormones and brain chemicals. Many blockbuster drugs work by targeting these receptors. This study reveals that common fats in the outer layer of our cells do much more than form a passive backdrop: they directly grip and shape a whole family of key receptors, subtly steering how signals are turned on or off. Understanding this hidden layer of control could help design smarter medicines with fewer side effects.
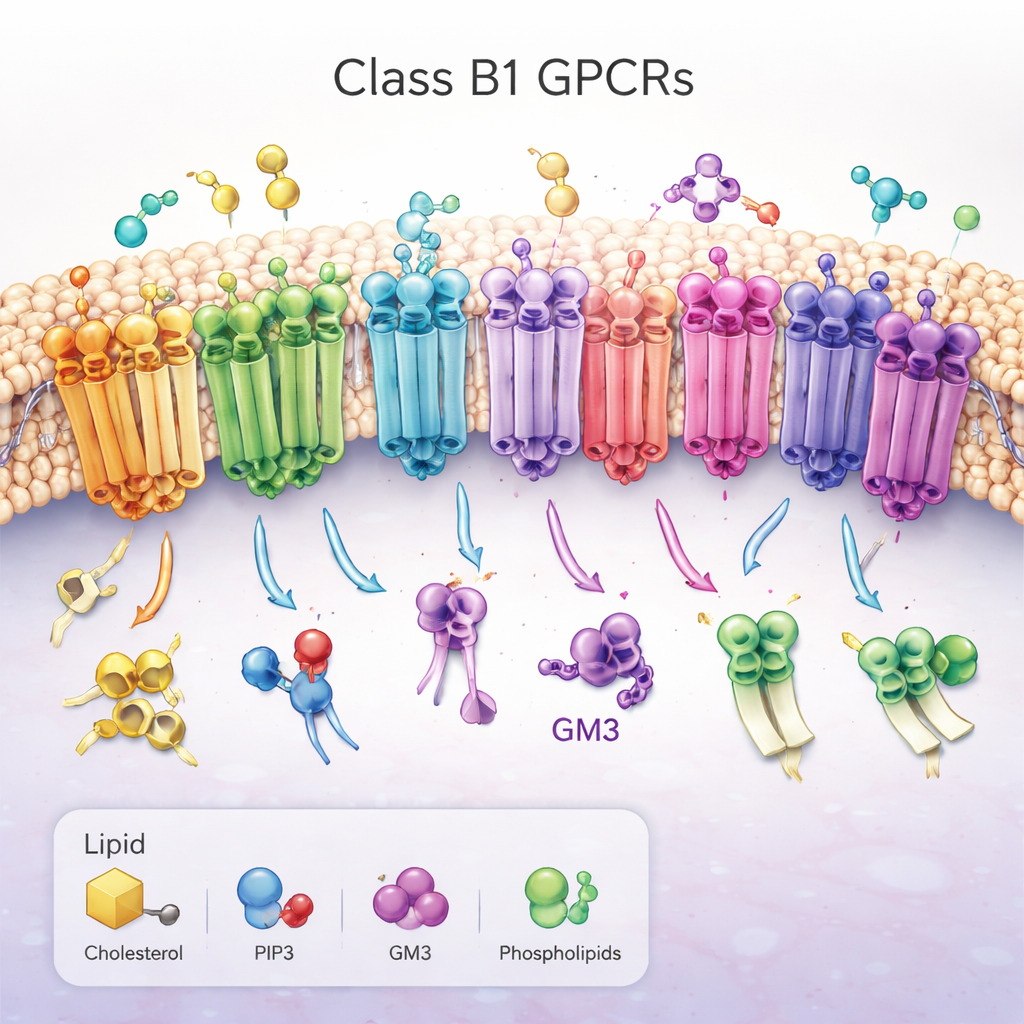
A family of important molecular antennas
The work focuses on class B1 G protein–coupled receptors (GPCRs), a set of 15 closely related receptors that help control blood sugar, metabolism, growth, and stress responses. They sit across the cell’s outer membrane, with a large “head” outside the cell that grabs hormone-like molecules and a bundle of seven helices threading through the fatty membrane. When activated, these receptors change shape on the inside of the cell, opening a groove that recruits partner proteins and triggers signaling cascades. Because of their central role in diseases such as diabetes and obesity, class B1 GPCRs are prime drug targets, yet how the surrounding membrane lipids regulate them has remained largely mysterious.
Simulating receptors in their natural neighborhood
Rather than studying receptors in artificial detergent bubbles, the researchers placed all 15 human class B1 GPCRs into computer-built membranes that mimic the complex mixture of fats in real cells. Using a technique called coarse-grained molecular dynamics, they ran multiple long simulations of each receptor in both its active and inactive state, adding up to about a millisecond of simulated time. They tracked how three “regulatory” lipids—cholesterol, the signaling lipid PIP2, and the sugar-decorated fat GM3—approached, bound, and let go of different regions of each receptor. To ensure that others can reproduce and build on this enormous dataset, they captured every step of the setup and analysis using an open workflow tool called aiida-gromacs, and they compared their physics-based results with predictions from a new AI model (Chai‑1) that guesses how proteins bind small molecules.
Hidden pockets and a conserved lipid grip
The simulations uncovered recurring binding patterns across the entire receptor family. Cholesterol, best known from diet discussions, was seen lodging not only in a previously known site on one of the helices but also in “deep membrane” pockets between helices, with its polar head buried unusually far into the membrane. One receptor, the secretin receptor, showed especially long-lived cholesterol binding in distinct pockets in its active and inactive forms, hinting that cholesterol may strongly bias how it signals. PIP2 showed a strikingly conserved behavior: it clustered around the inner face of the receptors at the junction of two helices and a short tail segment (TM6, TM7 and H8), particularly when the receptors were in the active state. At this site, networks of positively charged amino acids gripped the negatively charged PIP2 headgroup, suggesting a way this lipid could stabilize the active shape or help recruit signaling partners on the inside of the cell.
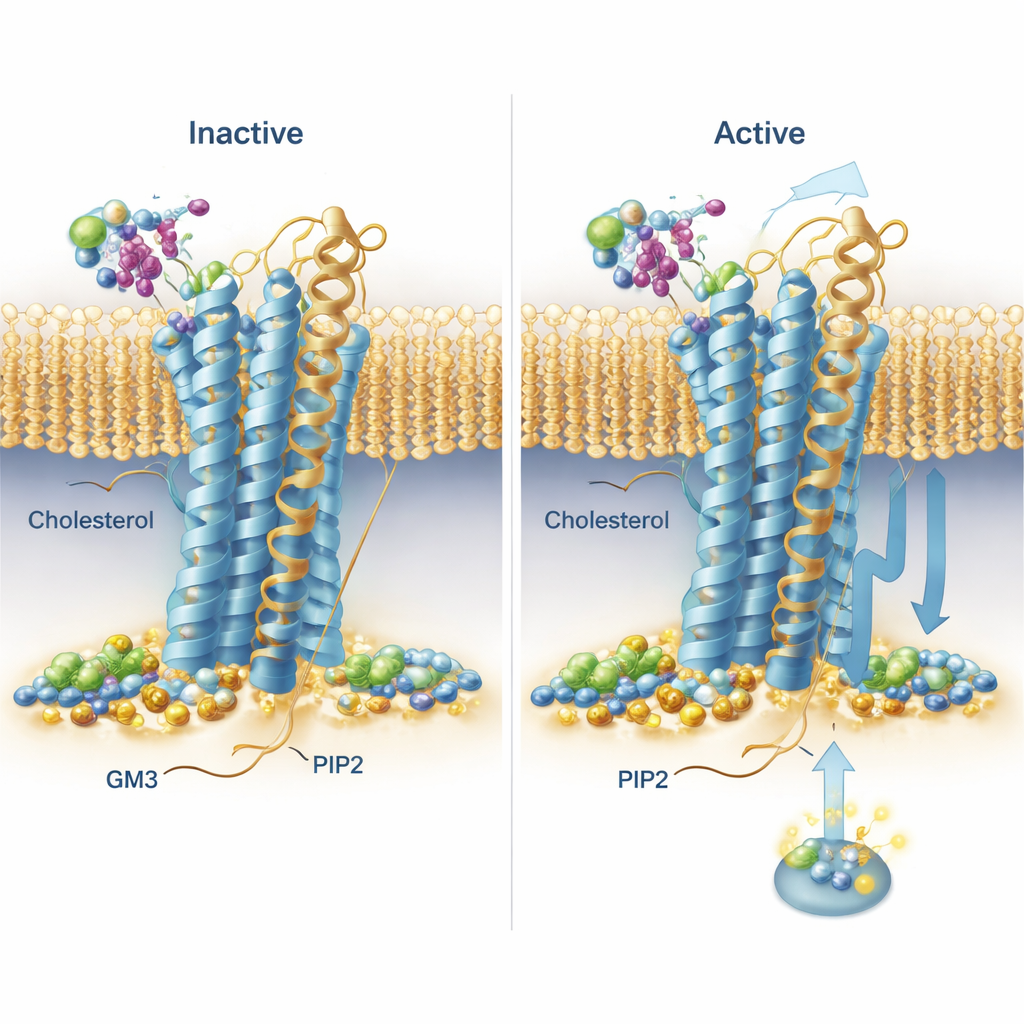
A sugar-coated lipid that nudges receptor motion
GM3, a complex lipid with a sugar head, mostly interacted with the large outer heads of the receptors. The team noticed that some receptors’ outer domains swung and bent a lot relative to the membrane, while others were more restrained. For two drug-relevant receptors—GLP‑1R and GIPR—they examined GM3’s role in detail. In simulations with GM3 present, the lipid either nestled against the base of the outer domain and ligand-binding pocket (GLP‑1R) or latched onto the far tip of the outer domain (GIPR), in both cases influencing how freely these domains could move. To test these predictions in living cells, the researchers used a light-based technique (TR‑FRET) that reports how close a labeled receptor head is to the membrane. When they reduced GM3 levels with a small-molecule inhibitor, they saw measurable changes in how the receptor heads shifted upon stimulation, broadly consistent with the idea that GM3 tunes receptor flexibility in a receptor- and state-specific way.
Why these lipid–receptor partnerships matter
Together, the study paints a picture of class B1 GPCRs as not just hormone switches but as parts of a tightly choreographed dance with surrounding lipids. Cholesterol can occupy hidden pockets and subtly favor certain signaling outcomes, PIP2 forms a conserved inner “handle” that may lock receptors into their active forms and guide partner proteins, and GM3 tweaks how the outer regions of receptors move and meet their ligands. For non-specialists, the key message is that the membrane is an active regulatory layer: small differences in lipid composition could change how the same receptor behaves in different tissues or disease states. By mapping these interactions in detail and sharing the underlying simulation data, this work lays a foundation for designing future drugs that exploit or avoid specific lipid contacts, potentially making treatments more selective and effective.
Citation: Chao, K.W., Wong, L., Oqua, A.I. et al. Human class B1 GPCR modulation by plasma membrane lipids. Commun Biol 9, 317 (2026). https://doi.org/10.1038/s42003-025-09445-2
Keywords: G protein-coupled receptors, membrane lipids, cholesterol, PIP2, GM3