Clear Sky Science · en
Structural insights into metallocluster trafficking in the nitrogenase assembly scaffold NifEN
How Nature Builds a Powerful Chemical Tool
Nitrogen is all around us in the air, but most living things can’t use it in that form. A special enzyme called nitrogenase solves this problem by turning atmospheric nitrogen into ammonia, a key ingredient for life and for fertilizers. At the heart of nitrogenase sits a dense metal “cluster” that does the hard chemistry, but how cells assemble this intricate piece has remained murky. This paper peeks inside that construction process, revealing how a protein scaffold named NifEN acts as a flexible loading dock and conveyor system for the cluster’s metal cargo.
A Molecular Factory Behind Fertilizer and Fuels
Nitrogenase is nature’s counterpart to industrial processes that make ammonia and liquid fuels. Instead of giant reactors, microbes use a compact protein machine powered by cellular energy. Its performance depends on a uniquely complex metal center, built from iron, sulfur, molybdenum, carbon and an organic side group. Assembling this core is too delicate to leave to chance, so cells employ a relay team of helper proteins. One of the most important is NifEN, which receives a nearly finished all‑iron precursor (called the L‑cluster), helps convert it into the mature form (the M‑cluster), and then hands it to the working nitrogenase enzyme. Understanding how NifEN manages this traffic could inform efforts to engineer microbes for cleaner fertilizer production or new carbon‑based fuels.
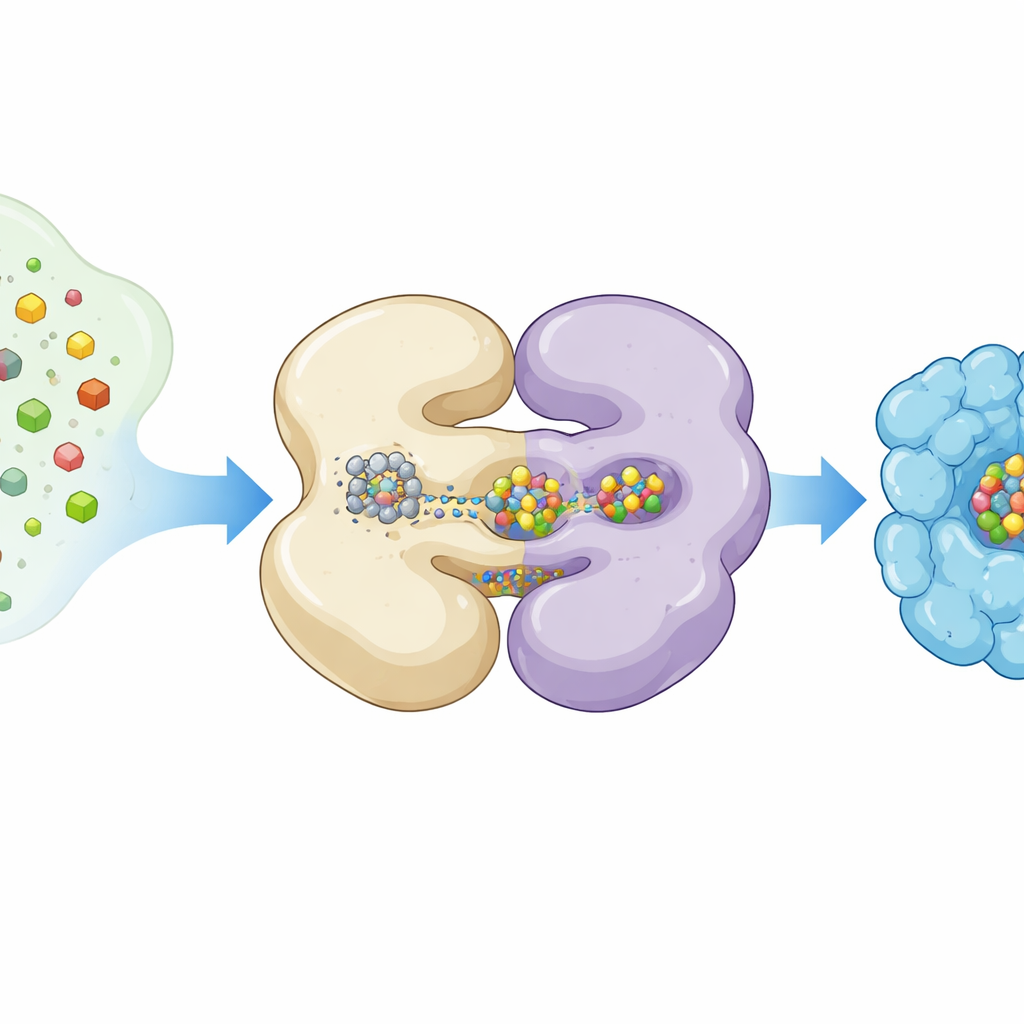
Seeing a Moving Machine with Frozen Snapshots
The authors used cryogenic electron microscopy, a technique that images proteins at extremely low temperatures, to capture NifEN in different working poses. They expressed NifEN in laboratory bacteria in a way that left some protein copies carrying an L‑cluster and others empty. By sorting millions of single‑particle images, they reconstructed two main shapes: an “apo” form lacking the mobile cluster and a “holo” form with the cluster bound at an internal site. Both forms share a core made of four subunits arranged as two pairs, but when the L‑cluster is present, one half of NifEN becomes noticeably more ordered and several helices swing inward, tightening around the metal cargo.
A Hidden Tunnel for Metal Cargo
Comparing the empty and loaded structures revealed a striking feature: a long tunnel running through the NifEN dimer. In the empty state, this passage is wide and open; after cluster binding it narrows as the protein grips its passenger. Earlier crystal structures had shown an alternative “outside” position for the L‑cluster near the protein surface. Putting all views together, the researchers inferred that the cluster can occupy at least two stations on NifEN—one buried inside and one exposed—and move between them along a curved path gated by shifts in a flexible domain. This motion appears to be more relaxed than in the final nitrogenase enzyme, whose active cluster is held rigidly in place, suggesting NifEN is built for handoff rather than long‑term catalysis.
Docking Partners and a Continuous Route
To understand how NifEN connects to its upstream and downstream partners, the team combined their experimental structures with AlphaFold 3 computer models and lower‑resolution electron microscopy of protein complexes. The models suggest that the enzyme NifB, which makes the L‑cluster from smaller iron–sulfur pieces, docks in a valley on one face of NifEN. There, a continuous tunnel can be traced from NifB’s own metal centers directly into the NifEN tunnel and on to the internal L‑cluster station. On the opposite face of NifEN, a different docking site accommodates NifH, the protein that inserts molybdenum and an organic side group to complete the cofactor. In this configuration, the L‑cluster sits at the surface site, ideally positioned for modification. Mutating key amino acids lining the proposed route disrupts cluster loading, movement, or maturation, providing experimental support for this conveyor‑belt model.
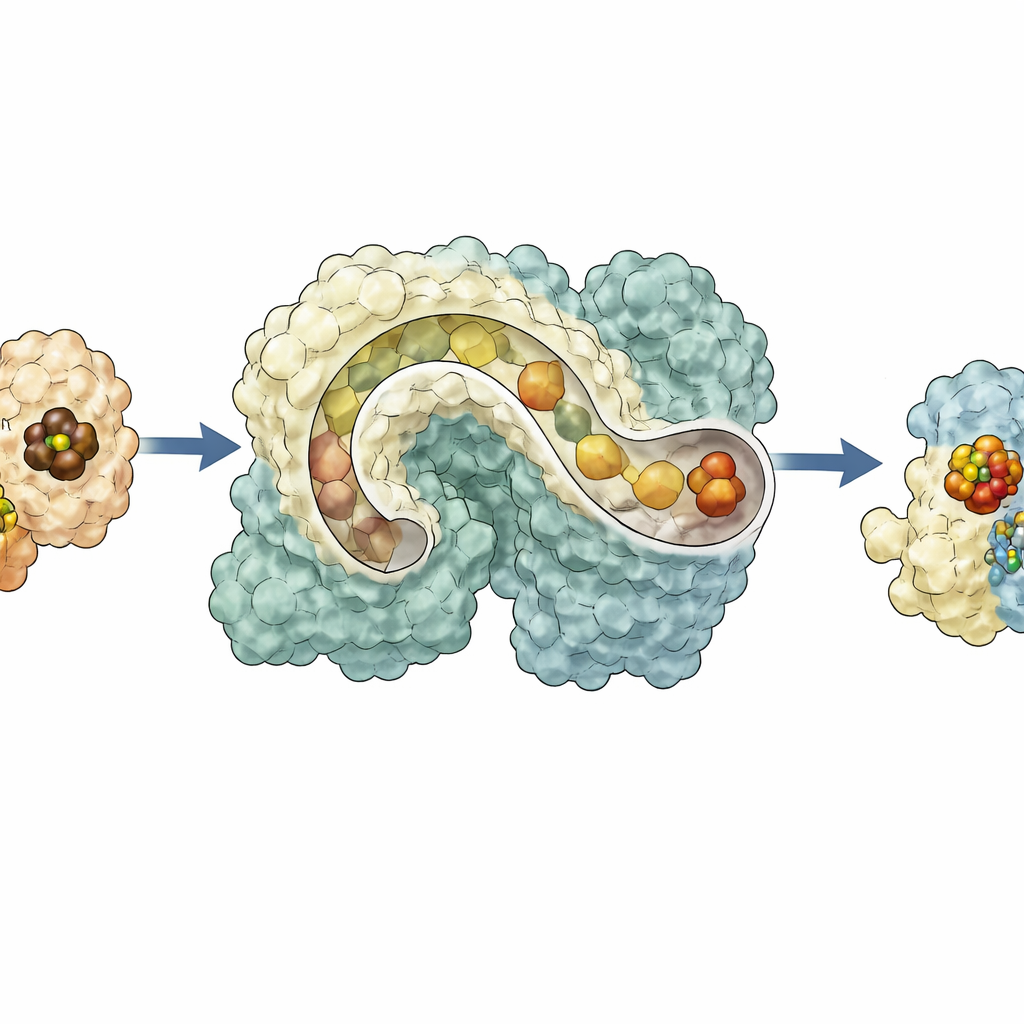
Why a Flexible Scaffold Matters
Taken together, the results paint NifEN as a dynamic hub that receives a metal core from NifB on one side, shifts it into an interior holding bay, then presents it on the opposite surface for finishing by NifH before guiding the mature cluster back inside for delivery to nitrogenase. This conformation‑gated trafficking scheme explains how multiple delicate steps can be coordinated within a single protein framework and hints at how ancient enzymes might have evolved from more flexible scaffolds into today’s highly specialized catalysts. For non‑specialists, the work shows that even at the nanoscale, nature relies on assembly lines, tunnels and moving parts to build the molecular tools that underpin global cycles of nitrogen, food production and, potentially, future green technologies.
Citation: Neumann, B., Brandon, K.A., Quechol, R. et al. Structural insights into metallocluster trafficking in the nitrogenase assembly scaffold NifEN. Nat Catal 9, 281–294 (2026). https://doi.org/10.1038/s41929-026-01489-9
Keywords: nitrogenase, metallocluster assembly, NifEN scaffold, cryo electron microscopy, biological nitrogen fixation