Clear Sky Science · en
Unveiling active sites and the cooperative role of non-thermal plasma and copper–zinc catalysts in the hydrogenation of CO2 to methanol
Turning Climate Gas into a Useful Liquid
Burning coal, oil and gas releases carbon dioxide (CO2), the main greenhouse gas driving climate change. What if we could not only capture this CO2 but also turn it into something useful, like methanol—a liquid that can serve as fuel, a building block for plastics, and a way to store renewable energy? This study explores a promising approach that uses electrical discharges known as non-thermal plasmas together with copper–zinc catalysts to convert CO2 into methanol more efficiently and under gentler conditions than in today’s chemical plants.
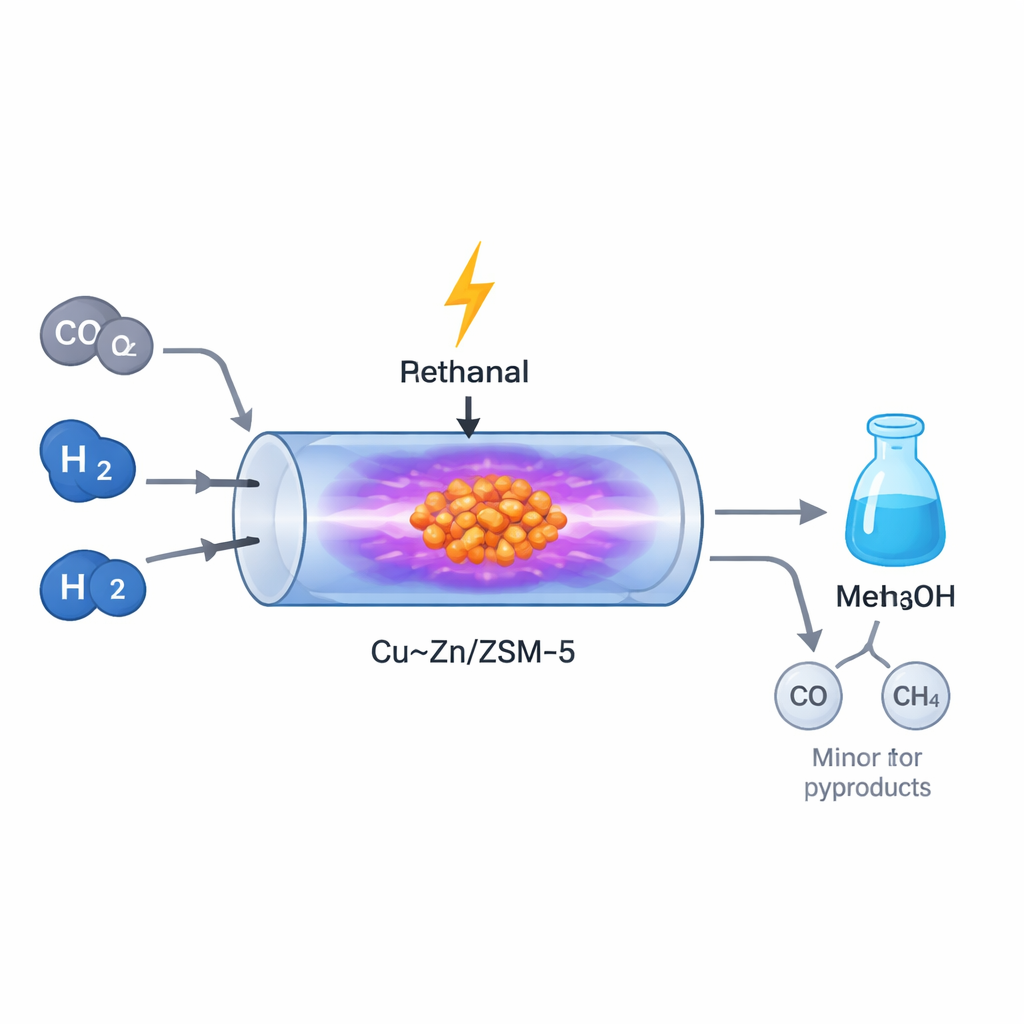
A New Way to Power Chemical Reactions
Traditional methanol plants run at high temperatures and pressures, demanding lots of energy and large, centralized facilities. In contrast, non-thermal plasma relies on strong electric fields to energize gas molecules without heating everything up. In this work, the researchers fed a mixture of CO2 and hydrogen into a small plasma reactor filled with a specially designed copper–zinc catalyst spread on a porous mineral called ZSM-5. The plasma created a flurry of excited and fragmented gas species that interacted with the catalyst surface, allowing methanol to form at around atmospheric pressure and relatively low bulk temperatures. This makes the process potentially well suited to flexible, renewable-powered "micro-plants" that can be located near sources of captured CO2.
Why Copper and Zinc Make a Strong Team
Copper-based catalysts are already used commercially to turn synthesis gas (a mixture of carbon monoxide, CO, and hydrogen) into methanol. However, under plasma conditions with CO2 as the starting point, a standard industrial copper–zinc–alumina catalyst performed poorly, converting only a small fraction of CO2. The researchers therefore redesigned the material: they fixed the copper loading at a low level and systematically varied the amount of zinc on the ZSM-5 support. They found that a particular composition, labeled 2Cu2Zn, struck the right balance. Under non-thermal plasma, this catalyst reached CO2 conversions of around 14–15%, a methanol selectivity near 37%, and a methanol production rate several times higher than that of copper or zinc alone. Importantly, these gains were achieved at far milder conditions than conventional thermal processes.
Peering Into the Catalyst While It Works
To understand why the copper–zinc pair worked so well, the team used a suite of advanced tools while the reaction was actually running. X-ray absorption methods showed that adding zinc helped break copper into smaller, more evenly dispersed particles and made the copper easier to keep in its metallic, active form. Meanwhile, zinc stayed as an oxidized species, forming a close interface with copper rather than mixing into a true alloy. Infrared spectroscopy with adsorbed carbon monoxide revealed that these copper–zinc oxide interfaces created special sites that bind CO differently from pure copper. When exposed to the plasma, these interfacial sites could stabilize reaction intermediates that are crucial stepping stones on the way to methanol, while the overall structure resisted clumping and re-oxidation over many hours.
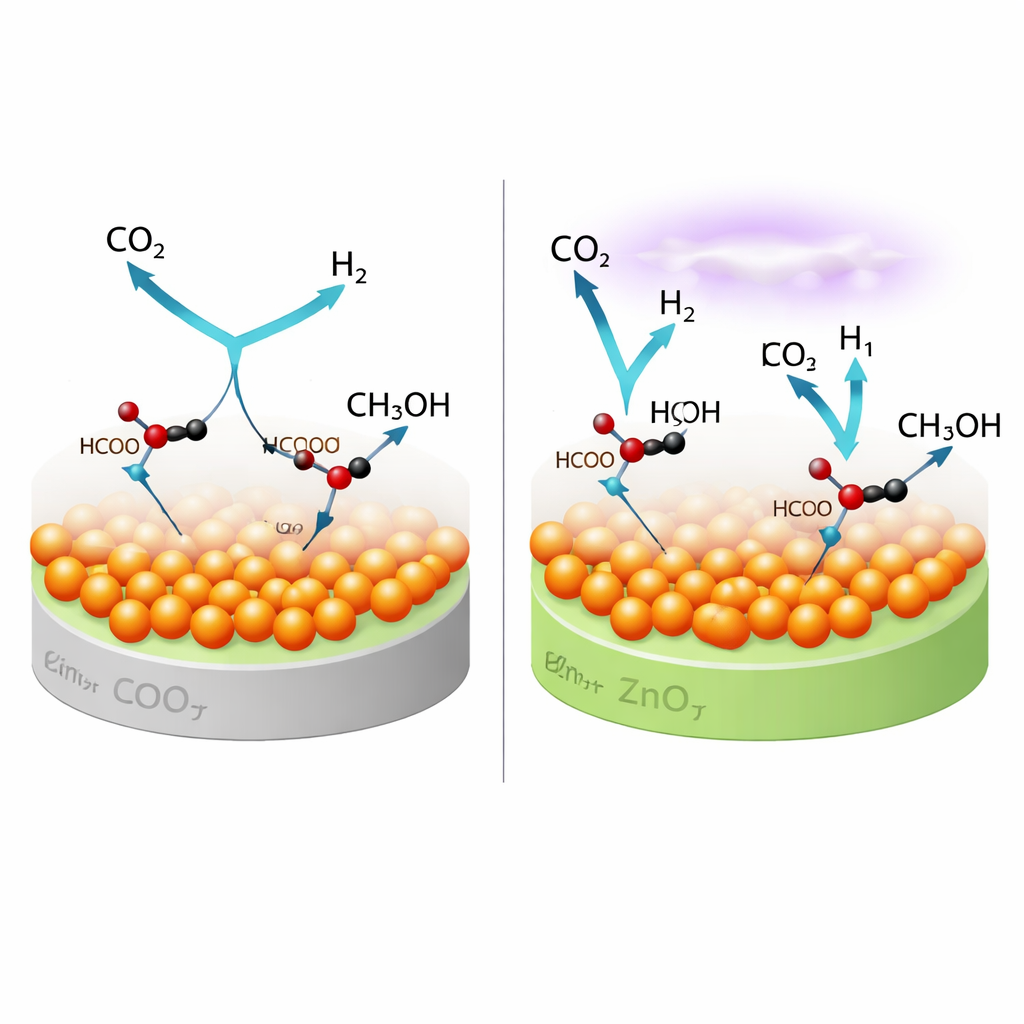
Two Pathways Working Hand in Hand
The study also tackled a key question: what molecular routes actually lead from CO2 to methanol under plasma conditions? Operando infrared measurements, combined with mass spectrometry, showed that on pure copper the reaction mainly proceeds through a "formate" route, where CO2 first attaches to the surface and is gradually hydrogenated. On the optimized copper–zinc catalyst, a second route opens. Here, the plasma breaks some CO2 apart in the gas phase to form CO, which then lands on the copper–zinc oxide interface and is further hydrogenated through a "formyl" intermediate before becoming methanol. Because the plasma continuously generates both CO and reactive hydrogen-containing species, these two pathways can operate side by side, boosting the overall methanol yield.
What This Means for Future Fuels
In everyday terms, this work shows how carefully engineered copper–zinc catalysts, when combined with electrically driven plasmas, can turn waste CO2 into useful methanol more efficiently and gently than traditional heat-based methods. The plasma provides highly reactive fragments of CO2 and hydrogen, while the catalyst’s copper–zinc interface offers the right kind of landing pads to guide these fragments along efficient reaction paths. Because the process runs at low pressure and relatively low temperature, it could be paired with intermittent renewable electricity and modular reactors placed close to CO2 sources. While much engineering remains before such systems can be deployed at scale, the study lays out a clear mechanistic blueprint for designing next-generation, electrified reactors that help close the carbon loop.
Citation: Xu, S., Potter, M.E., Simancas, R. et al. Unveiling active sites and the cooperative role of non-thermal plasma and copper–zinc catalysts in the hydrogenation of CO2 to methanol. Nat Catal 9, 134–147 (2026). https://doi.org/10.1038/s41929-025-01477-5
Keywords: CO2-to-methanol, non-thermal plasma catalysis, copper-zinc catalysts, carbon recycling, electrified chemical processes