Clear Sky Science · en
Metallic charge transport in conjugated molecular bilayers
Why this tiny crystal matters
Modern electronics rely on how easily electric charges can move through a material. Silicon—the workhorse of today’s chips—can carry charge extremely well, even at very low temperatures. Organic semiconductors, made from carbon-based molecules, promise flexible, lightweight, even printable electronics, but they usually fall far behind silicon in how fast charges move. This article reports an organic molecular crystal that behaves unexpectedly like a metal across a wide range of temperatures, revealing a design strategy that could bring flexible electronics much closer to silicon-like performance.
Making soft materials act more like metals
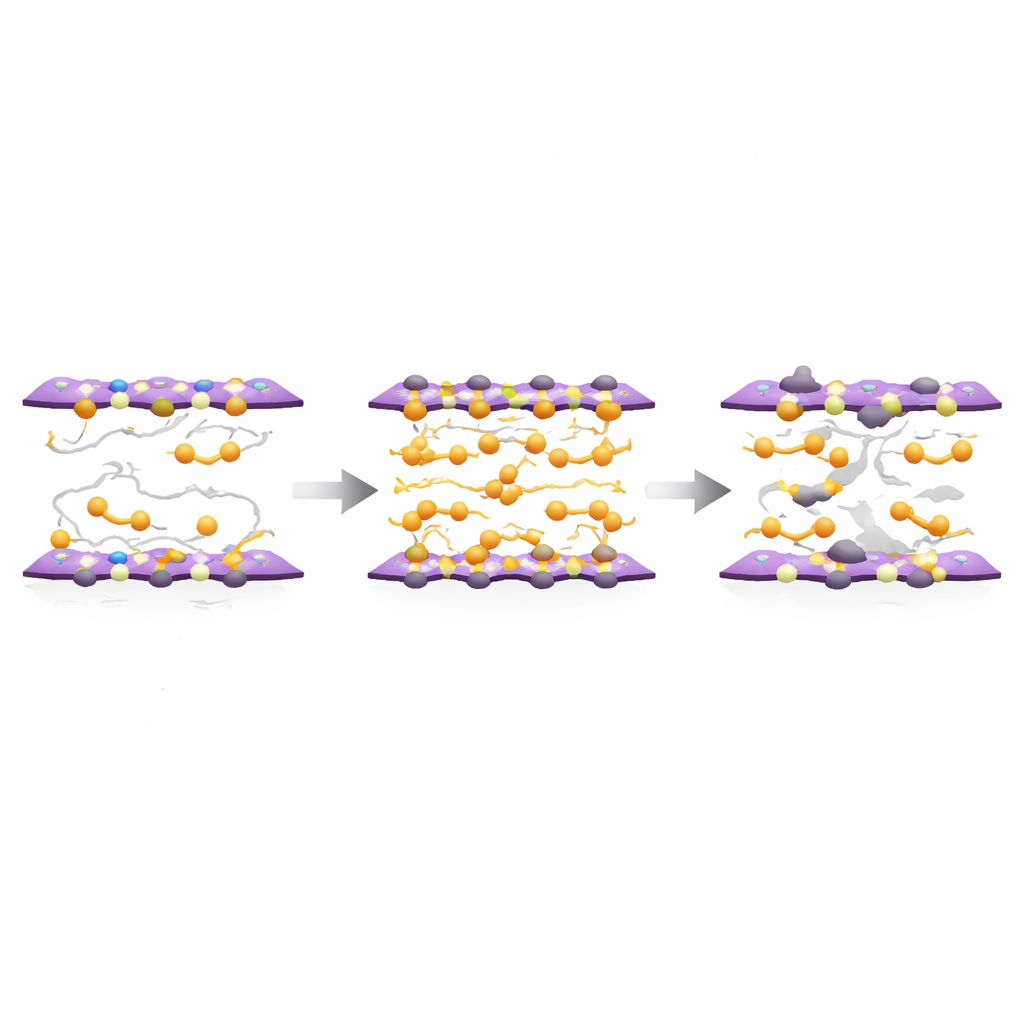
In most organic semiconductors, molecules are held together only by weak forces, so they vibrate and jostle, constantly disturbing the paths that charges try to follow. As a result, charge motion slows down when the temperature drops and eventually becomes trapped, so the material behaves more like an insulator than a metal. The researchers studied a particular molecule called Ph-BTBT-C10 that can form extremely thin, highly ordered crystals just two molecular layers thick. In these crystals, pairs of phenyl rings act as short bridges between the two layers, pulling them close together and making the whole structure more rigid. Theory and computer simulations suggested that these bridges both stiffen the crystal and allow charges to tunnel easily from one layer to the other, creating a more robust, two-layer network for current to flow.

Growing nearly perfect molecular sheets
To test this idea, the team devised a slow, solution-based method to grow large, ultrathin crystals of Ph-BTBT-C10 on silicon oxide. As the hot, concentrated solution cooled on the surface, fluid flows gently swept molecules into place, allowing single-crystal films hundreds of micrometres across to form on top of a thin liquid layer. X-ray scattering and atomic force microscopy showed that the resulting films were extraordinarily flat and ordered, with step heights corresponding exactly to bilayer thicknesses and very few visible defects. This careful growth process proved crucial: it yielded crystals pristine enough that the subtle advantages of the phenyl bridges—stronger layer-to-layer coupling and reduced molecular motion—could play a dominant role in charge transport.
Metal-like current in a flexible crystal
The researchers then built field-effect transistors from these bilayer crystals and measured how current and conductivity changed from room temperature down to just 8 kelvin, only a few degrees above absolute zero. In typical organic devices, conductivity falls off sharply at low temperature as charges freeze in defects. Here, the opposite happened: once enough charge was induced at the crystal surface, the conductivity increased as the device was cooled and stayed high all the way down, a hallmark of metallic behaviour. At the lowest temperature, the organic crystal reached conductivities comparable to some heavily doped inorganic semiconductors and achieved charge mobilities above 100 square centimetres per volt-second—exceptionally high for an undoped organic material. Independent Hall measurements confirmed that the charges moved freely over distances spanning several molecular spacings, consistent with a metal-like state.
Turning metal into insulator on demand
Beyond demonstrating fast charge motion, the team also explored how this metallic state can be disrupted. By deliberately stressing the devices at elevated temperature and high voltage, they introduced controlled disorder—effectively creating extra defects inside the crystal. After this treatment, the same material could be tuned from metallic to insulating simply by adjusting the electric field. At high fields, charges still flowed like in a metal; at lower fields, they became trapped and the resistance rose with cooling. The transition between these regimes followed patterns seen in well-known metal–insulator transitions in inorganic systems, suggesting that this organic crystal can serve as a model platform to study similar physics in soft, molecular materials.
What this means for future electronics
To a non-specialist, the key message is that the way molecules are connected in a crystal can drastically change how well they carry electricity. By engineering strong bridges between layers and carefully controlling crystal quality, the authors transformed a soft, flexible organic material into something that behaves like a metal over a wide temperature range, while remaining undoped and structurally simple. At the same time, they showed that a controlled amount of disorder can switch this metallic state off, hinting at new kinds of memory, sensing, or temperature-stable devices based on organic materials. The work points toward a design recipe—using such molecular bridges—to push flexible electronics closer to the performance of traditional semiconductors, while opening a new playground for studying fundamental electronic transitions in molecular systems.
Citation: Lu, K., Li, Y., Wang, Q. et al. Metallic charge transport in conjugated molecular bilayers. Nat Electron 9, 246–256 (2026). https://doi.org/10.1038/s41928-025-01553-5
Keywords: organic semiconductors, metal–insulator transition, charge transport, flexible electronics, molecular crystals