Clear Sky Science · en
Distinction between primary and metastatic mucinous ovarian carcinoma from histopathology images using deep learning
Why this matters for patients and doctors
When a person is told they have ovarian cancer, the next crucial question is where the cancer started. For a common subtype called mucinous ovarian carcinoma, tumors in the ovary can either be true ovarian cancers or growths that have spread from the stomach or intestines. These two situations demand very different treatments and carry different outlooks, yet even expert pathologists sometimes struggle to tell them apart under the microscope. This study introduces a specialized artificial intelligence (AI) system that reads digital microscope images and helps doctors distinguish these two possibilities more accurately and efficiently.
Two look‑alike cancers with very different paths
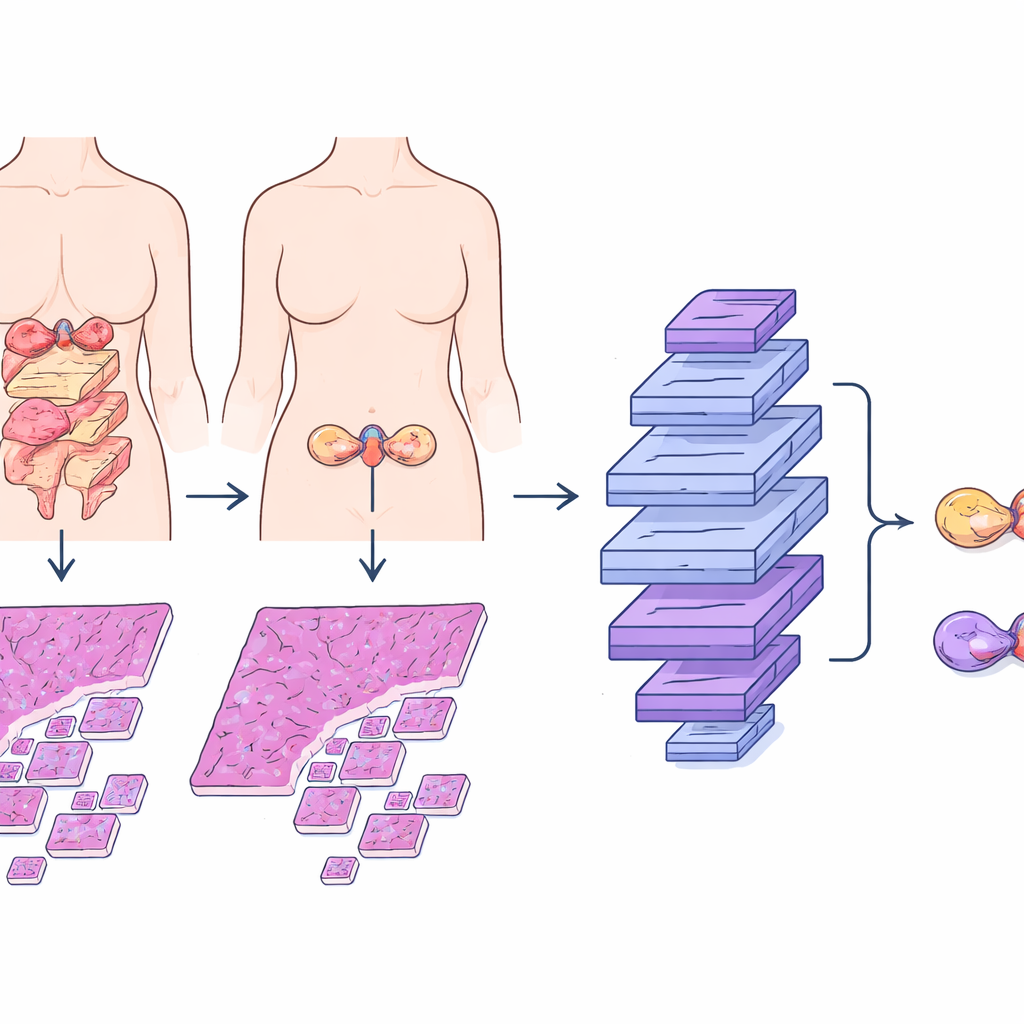
Mucinous ovarian carcinoma often produces large, mucus‑filled tumors that can look remarkably similar whether they began in the ovary or arrived there from the gastrointestinal tract. Traditionally, pathologists rely on a mix of visual clues in stained tissue slides, added laboratory stains, imaging tests, and clinical history. Even with this full work‑up, the diagnosis can be uncertain, especially when small or hidden tumors exist elsewhere in the body. Past research proposed checklists based on tumor size, whether one or both ovaries are involved, and certain cell types, but these rules can fail when metastatic tumors cleverly mimic primary ovarian growths. Mislabeling a metastatic tumor as primary, or vice versa, can lead to the wrong surgery, the wrong chemotherapy, and inaccurate counseling about prognosis.
Teaching computers to read tissue images
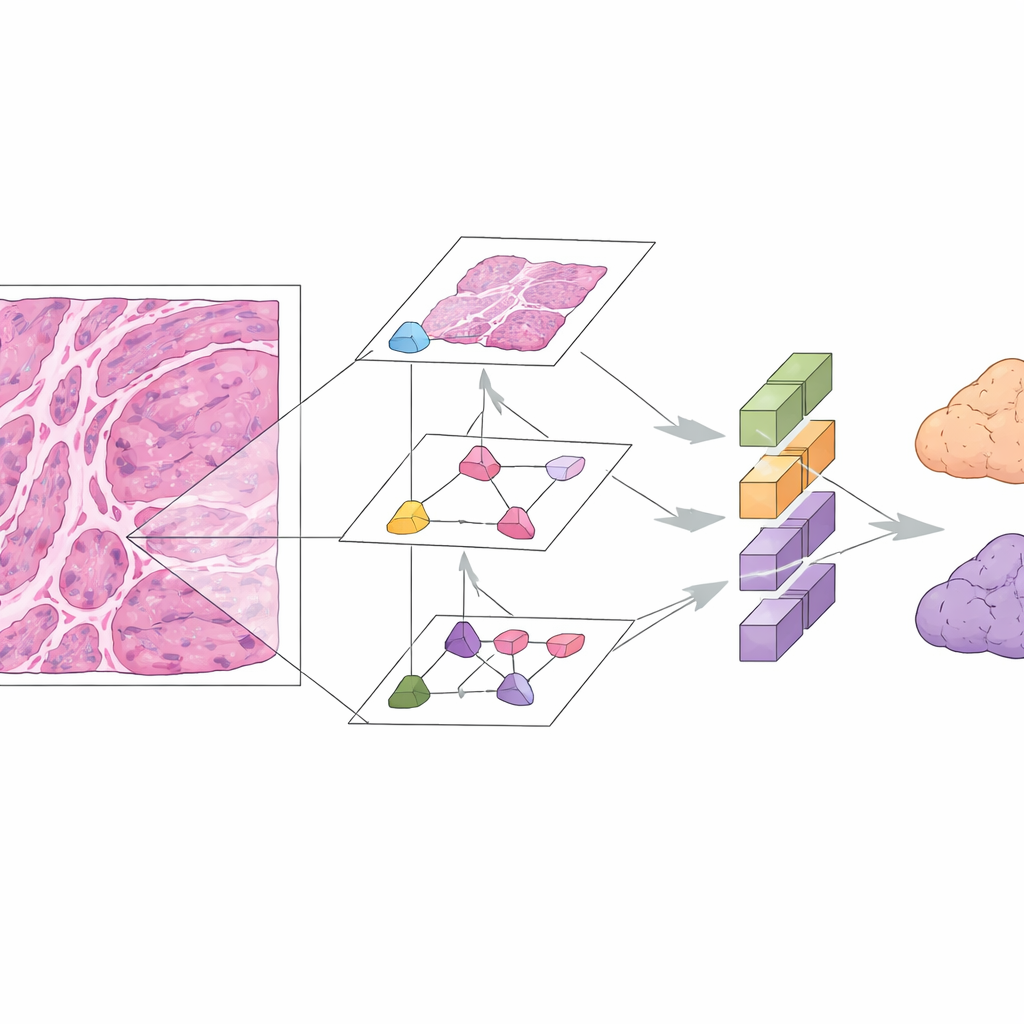
Digital pathology and deep learning offer another way forward. In this study, researchers assembled microscope images from 167 patients with well‑documented mucinous ovarian tumors across three hospitals. They focused on standard hematoxylin‑and‑eosin (H&E) stained slides, which are already part of routine care. The team built the Mucinous Ovarian Carcinoma Origin Prediction Model, or MOCOPM, which automatically breaks each whole‑slide image into many small patches and analyzes them without any added clinical information. They tested several neural network designs and found that a newer graph‑based model, inspired by how pathologists zoom in and out at different magnifications, worked best. This model connects patches across three levels of zoom, allowing it to capture both fine cellular details and broader growth patterns.
How well the AI performs in real‑world settings
MOCOPM was first trained and checked using cases from a large referral hospital, where difficult and unusual tumors are common. In this internal group, it correctly separated primary from gastrointestinal metastatic tumors with high accuracy, reflected by an area under the receiver operating characteristic curve of 0.91. The researchers then challenged the system with images from two additional hospitals, where slides were prepared independently and cases were generally more straightforward. Here, the model performed even better, with an area under the curve of 0.96 and strong scores for precision and recall. It also held up when tested on a separate public dataset of primary mucinous ovarian cancers from another research group, suggesting that the approach is reasonably robust across different sources.
Looking inside the black box
Because AI tools can seem opaque, the researchers used an explanation technique called GNNExplainer to highlight which regions of each slide most influenced the model’s decisions. In the majority of correctly classified cases they examined, the highlighted areas overlapped with features that human experts already use, such as clusters of special "signet ring" cells, patterns of how tumor cells invade surrounding tissue, and mixtures of benign, borderline, and clearly malignant components. This overlap suggests the system is not simply picking up on irrelevant visual quirks, but is keying in on medically meaningful patterns. Importantly, MOCOPM needs only routine H&E slides, potentially saving time and cost by reducing the need for extensive additional staining.
What this could mean for future care
The authors emphasize that MOCOPM is not meant to replace pathologists, but to serve as a decision‑support tool. In busy or resource‑limited settings, it could flag challenging cases, point experts toward suspicious regions on a slide, and provide an additional check before finalizing a diagnosis. The study has limitations: the cancer type is rare, the sample size is modest, and metastatic tumors from non‑intestinal sites were not included. Larger, prospective studies are needed before such a system could be integrated into everyday practice. Still, the work shows that carefully designed AI can assist with one of the trickiest distinctions in ovarian cancer, helping ensure that patients receive treatment tailored to where their cancer truly began.
Citation: Zhang, MY., Liu, B., Qin, ZJ. et al. Distinction between primary and metastatic mucinous ovarian carcinoma from histopathology images using deep learning. npj Digit. Med. 9, 276 (2026). https://doi.org/10.1038/s41746-026-02459-y
Keywords: mucinous ovarian carcinoma, digital pathology, deep learning, graph neural network, cancer diagnosis