Clear Sky Science · en
Detecting isolated REM sleep behavior disorder at home using a lower-back wearable sensor
Why strange dreams can warn of brain disease
Some people act out their dreams—kicking, shouting or flailing in their sleep—without knowing it. This condition, called REM sleep behavior disorder, is one of the strongest early warnings that diseases like Parkinson’s may develop years later. Today, confirming the diagnosis usually requires a night in a hospital sleep lab wired to machines. This study asks a simple question with big consequences: could a tiny sensor taped to your lower back at home spot the same warning signs over several nights?
A quiet disorder with serious implications
Isolated REM sleep behavior disorder (iRBD) happens when the body loses its usual muscle “off switch” during dream sleep. Instead of remaining still, people may thrash around and act out their dreams. More than 80 percent of those diagnosed eventually develop Parkinson’s disease or related brain disorders, often a decade or more later. Catching iRBD early could open a window for monitoring and, one day, for treatments that slow or prevent disease—but the current gold-standard test, overnight video-polysomnography, is expensive, scarce, and captures only a single night that may not reflect a person’s usual sleep.
Bringing sleep testing home
The researchers recruited 73 middle-aged and older adults, 15 with confirmed iRBD and 58 without. Everyone first spent a night in a sleep lab, wired to the usual equipment while also wearing a lightweight motion sensor taped over the lower spine. Then they wore the same sensor at home for up to six additional nights. The device recorded how the trunk moved in three directions all night long. From these recordings the team calculated more than a hundred simple movement features, such as how long a person stayed still, and how often brief twitches appeared during the night and in the hours when dream sleep is most common.
Patterns hidden in nighttime motion
Using these features, the scientists trained several types of machine-learning models to tell apart people with and without iRBD. They carefully avoided “peeking” at the test subjects by rebuilding the model from scratch each time they held one person out for evaluation. Two movement measures stood out: long stretches of immobility and short twitch-like bursts. Compared with controls, people with iRBD tended to show fewer long, quiet periods and more scattered twitch activity, and these patterns varied widely from night to night. Interestingly, when the team looked at all the movement features together, the biggest difference was not between people with and without iRBD, but between nights spent in the lab and nights spent at home—evidence that the unfamiliar lab environment changes how people move in their sleep.
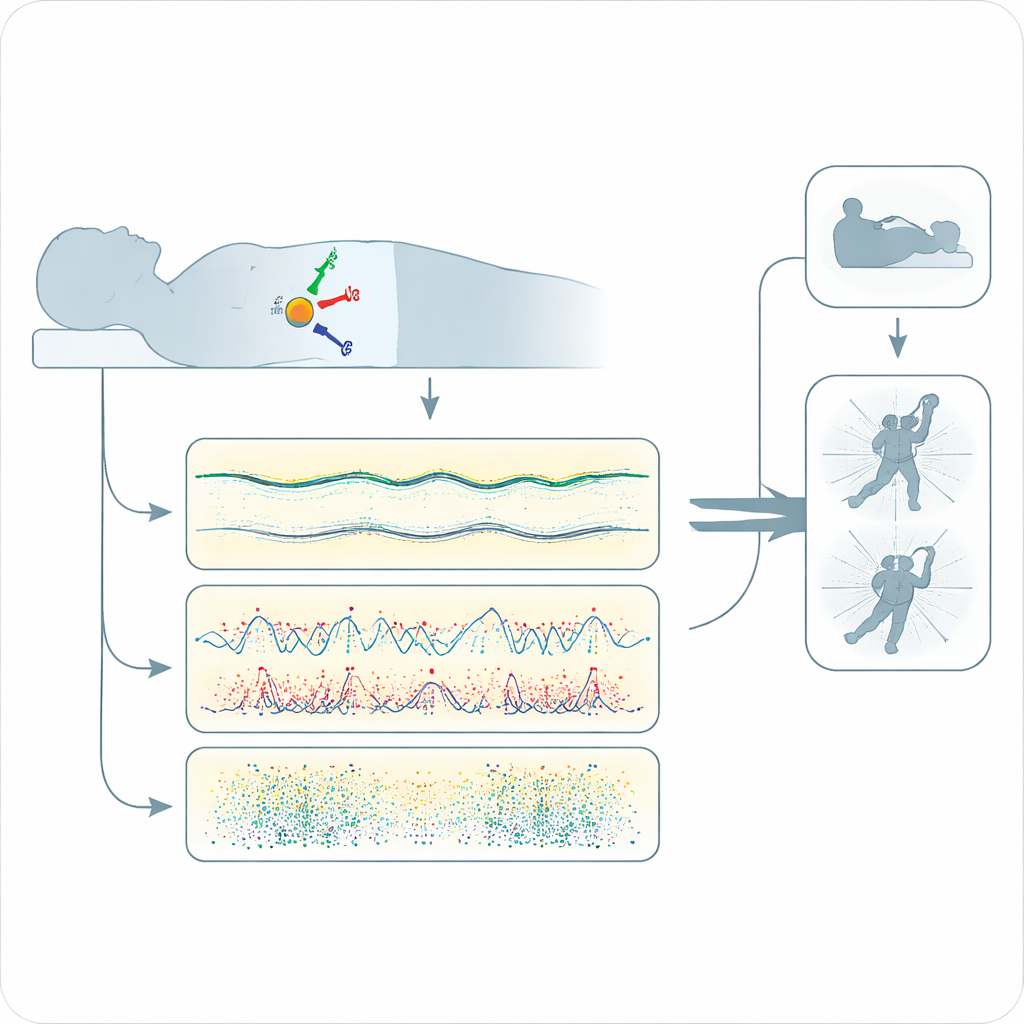
Multiple nights improve detection
The best-performing model, a support vector machine, was especially good at sensitivity—correctly flagging those who truly had iRBD. When trained on data from home nights, it identified about 93 percent of iRBD participants while maintaining moderate specificity, correctly ruling out roughly 72 percent of those without the disorder. Crucially, performance improved as more nights were added: sensitivity climbed and leveled off after about five home nights, reflecting the reality that dream-enactment behaviors do not appear every night. Some of the people flagged as “possible iRBD” by the algorithm actually showed other sleep problems or milder warning signs that also increase nighttime movement and may themselves be linked to future brain disease.
What this could mean for patients
This work suggests that a single, small sensor worn on the lower back at home over several nights can capture meaningful signs of a dangerous sleep disorder. While the method is not accurate enough to replace full sleep-lab studies, its high sensitivity makes it promising as an early screening tool: it could cast a wide net in large groups, identifying people who should go on to more detailed testing. As digital health devices become more common, combining home-based motion tracking with other simple measures, such as questionnaires or brain-wave recordings, may one day allow doctors to monitor both sleep and movement continuously in the real world, catching early hints of neurodegenerative disease long before symptoms appear.
Citation: Tzfoni, T., Tauman, R., Hausdorff, J.M. et al. Detecting isolated REM sleep behavior disorder at home using a lower-back wearable sensor. npj Digit. Med. 9, 210 (2026). https://doi.org/10.1038/s41746-026-02412-z
Keywords: REM sleep behavior disorder, wearable sensors, home sleep monitoring, Parkinson’s disease risk, digital health screening