Clear Sky Science · en
Physics constrained graph neural network for real time prediction of intracranial aneurysm hemodynamics
Why this matters for brain health
Bulging weak spots in brain arteries, called intracranial aneurysms, can burst without warning and cause life‑threatening bleeding. Doctors know that the way blood flows over these fragile walls affects whether they stay stable or give way, but today’s tools for simulating that flow are so slow and specialized that they are rarely used in day‑to‑day care. This study presents a fast artificial‑intelligence method that can mimic detailed blood‑flow simulations in seconds rather than hours, opening the door to quicker and more personalized decisions about who needs treatment and how.
From static pictures to moving blood
Right now, most aneurysm decisions are based on what can be seen on scans: the bulge’s size, shape, and position, plus basic risk factors like age and high blood pressure. These snapshots miss the invisible forces of flowing blood, such as how hard it rubs along the vessel wall or how much that force changes over each heartbeat. Traditional computer fluid dynamics can calculate these quantities precisely, but requires expert setup and long runs on powerful computers. As a result, many hospitals cannot realistically use them when a doctor needs an answer within a clinic visit or an emergency procedure.
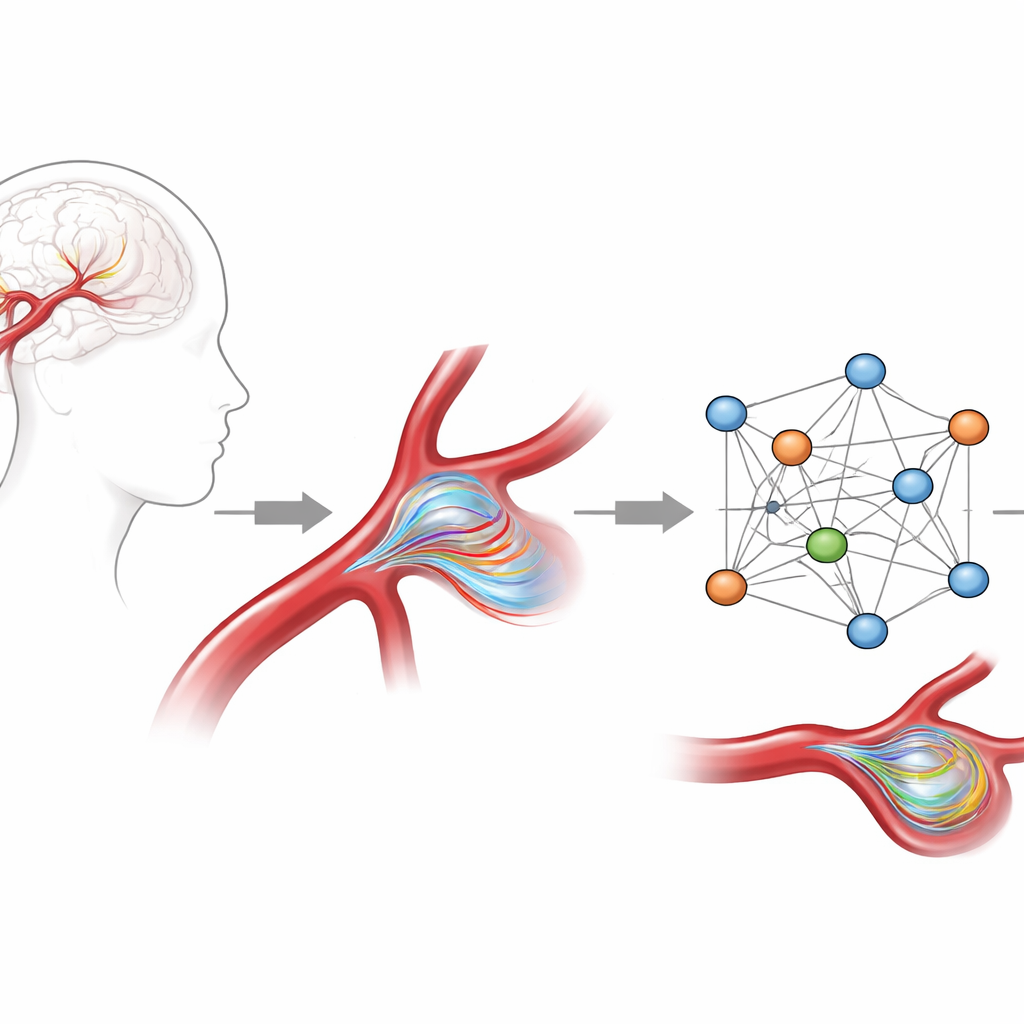
Teaching an AI to respect physics
The researchers built an AI model based on graph neural networks, a type of algorithm that works naturally on branching structures like blood‑vessel meshes rather than square image grids. They trained it on high‑quality computer simulations of blood flow in 105 aneurysm shapes derived from real patient data, focusing on a common location in a key brain artery. Each case included full three‑dimensional velocity and pressure fields over an entire heartbeat. To give the model a strong sense of the underlying physics, they did more than just ask it to copy the simulated speeds: they added extra features that describe how fast blood is entering the artery and how quickly it is accelerating, and they penalized the AI whenever its predictions violated basic fluid laws such as conservation of mass.
Seeing inside a dangerous bulge
After training, the AI could take one frame of blood‑flow information and rapidly roll it forward through time, recreating the swirling patterns and recirculation zones inside the aneurysm sac. Compared with the original simulations, its short‑term errors were already low, but the real test was whether those errors would snowball as it predicted dozens of steps into the future. The enhanced, physics‑constrained version passed this test: over 50 prediction steps, its mistakes were more than six times smaller than those of a simpler baseline, and it tracked the changing flow even during the most turbulent phase of the heartbeat. When the team converted these velocity fields into clinically relevant measures—such as average wall shear stress over the aneurysm dome—the AI stayed within about 10 percent of the reference values and reproduced where the highest stresses appeared.
Generalizing beyond the training set
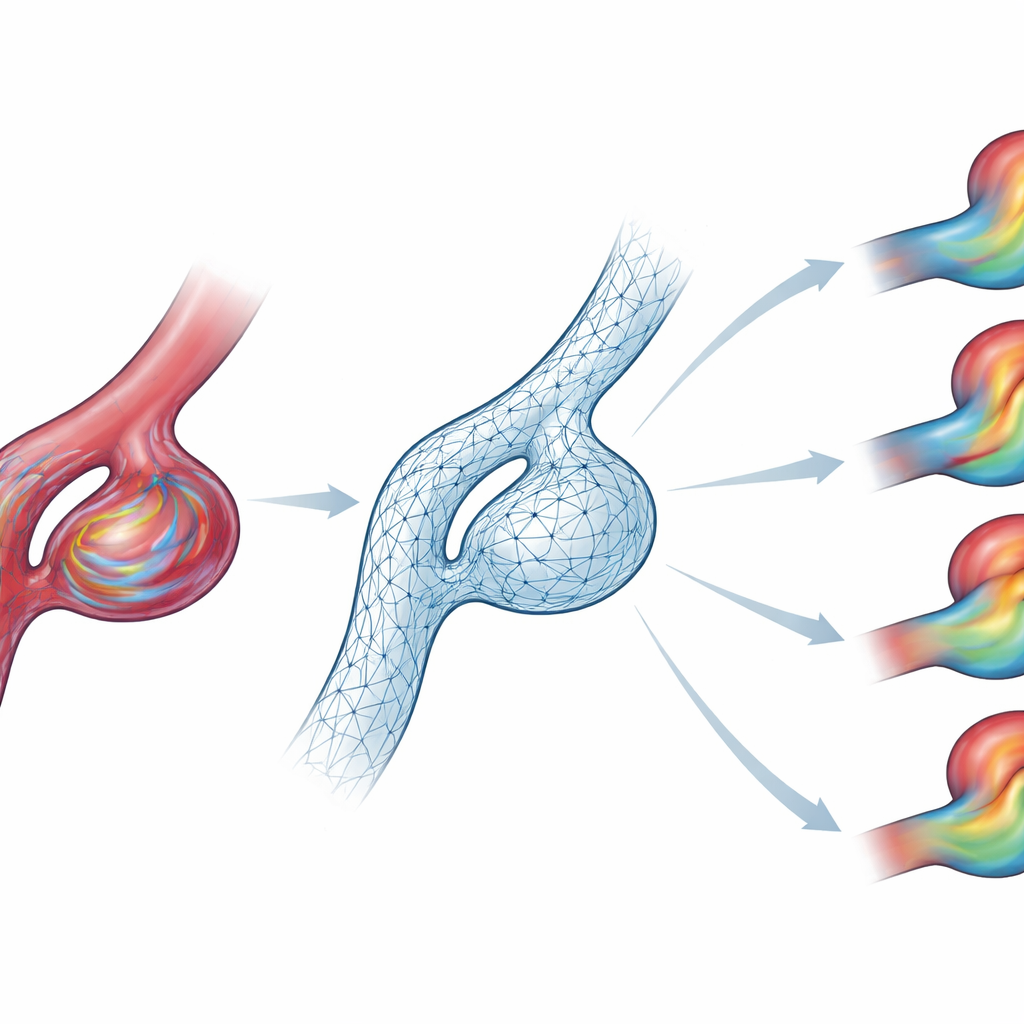
A key question for any medical AI is whether it can handle patients it has never seen before. The team challenged their model with two kinds of unfamiliar situations, without any extra training. First, they changed the inflow waveforms to match patterns measured in different brain arteries. The AI still followed the new timing and strength of each pulse, keeping errors modest and capturing the reshaped flow inside the bulge. Second, they tested it on four fully patient‑specific aneurysm geometries with different sizes, neck widths, and vessel paths. Although the exact speed levels were sometimes off, the model reproduced the main flow structures and impact zones, suggesting it had learned the underlying behavior rather than just memorizing the training shapes.
Faster answers at the bedside
Because this AI runs roughly sixty times faster than the traditional simulations it learned from, it becomes more efficient than standard methods after only a few dozen cases and could eventually deliver full‑field blood‑flow predictions in under a minute. The authors emphasize that their tool is not a stand‑alone rupture predictor; instead, it is a fast engine for generating physically consistent flow maps and wall stresses that other risk models can use. They also release their 105‑case dataset as a public benchmark so that other groups can build and compare improved models. With further training on more realistic vessel shapes and patient‑specific inflow data, this approach could help move sophisticated blood‑flow analysis out of the supercomputer lab and into everyday care for people living with brain aneurysms.
Citation: Lannelongue, V., Garnier, P., Jeken-Rico, P. et al. Physics constrained graph neural network for real time prediction of intracranial aneurysm hemodynamics. npj Digit. Med. 9, 212 (2026). https://doi.org/10.1038/s41746-026-02404-z
Keywords: brain aneurysm, blood flow, graph neural network, computational modeling, digital medicine