Clear Sky Science · en
Rapid prediction of cardiac activation in the left ventricle with geometric deep learning: a step towards cardiac resynchronization therapy planning
Why timing matters for a struggling heart
For many people with severe heart failure, tiny electrical pulses from an implanted device can help the heart’s main pumping chambers squeeze in better harmony. This treatment, called cardiac resynchronization therapy, can ease symptoms and extend life. Yet roughly one in three patients gains little benefit, often because the pacing wire on the left side of the heart cannot be placed in the best possible spot. This study explores whether computer models powered by modern artificial intelligence can rapidly predict how electricity spreads through the left side of the heart and help doctors choose pacing locations tailored to each patient.
From slow simulations to instant predictions
Today’s most accurate computer models of the heart rely on complex equations that mimic how electrical signals travel through heart muscle. While detailed, these simulations can take minutes to run on powerful computers—too slow for routine use during a medical procedure. The authors set out to build fast “surrogate” models that could learn from these heavy simulations and then reproduce their results almost instantly. They focused on the left ventricle, the heart’s main pumping chamber, and on predicting “activation time maps,” which show how quickly different regions of this chamber are electrically switched on during a heartbeat.
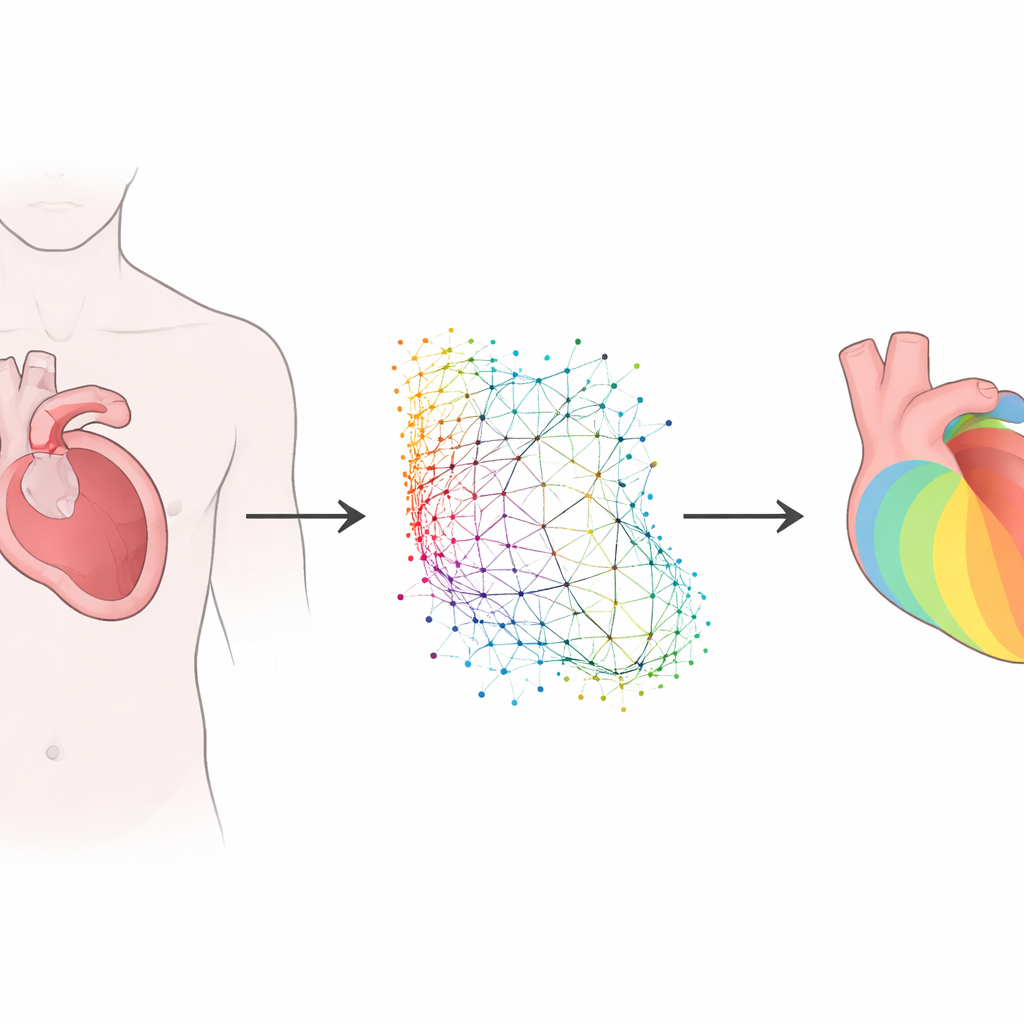
Teaching AI the language of heart shapes
Every person’s heart has a slightly different shape, and these differences matter for how electrical waves spread. Rather than forcing all hearts into a rigid grid, the researchers used a family of methods called geometric deep learning, which can work directly with irregular shapes. They developed and compared two related approaches. One, based on a graph neural network, treats the left ventricle as a collection of points connected like a mesh. The other, called a geometry-informed neural operator, first encodes this irregular shape onto a regular internal grid, processes it, and then maps the result back to the original anatomy. Both models take in the heart’s three‑dimensional shape, the locations where it is stimulated, and how well the tissue conducts electricity, and then predict how activation spreads through the muscle wall.
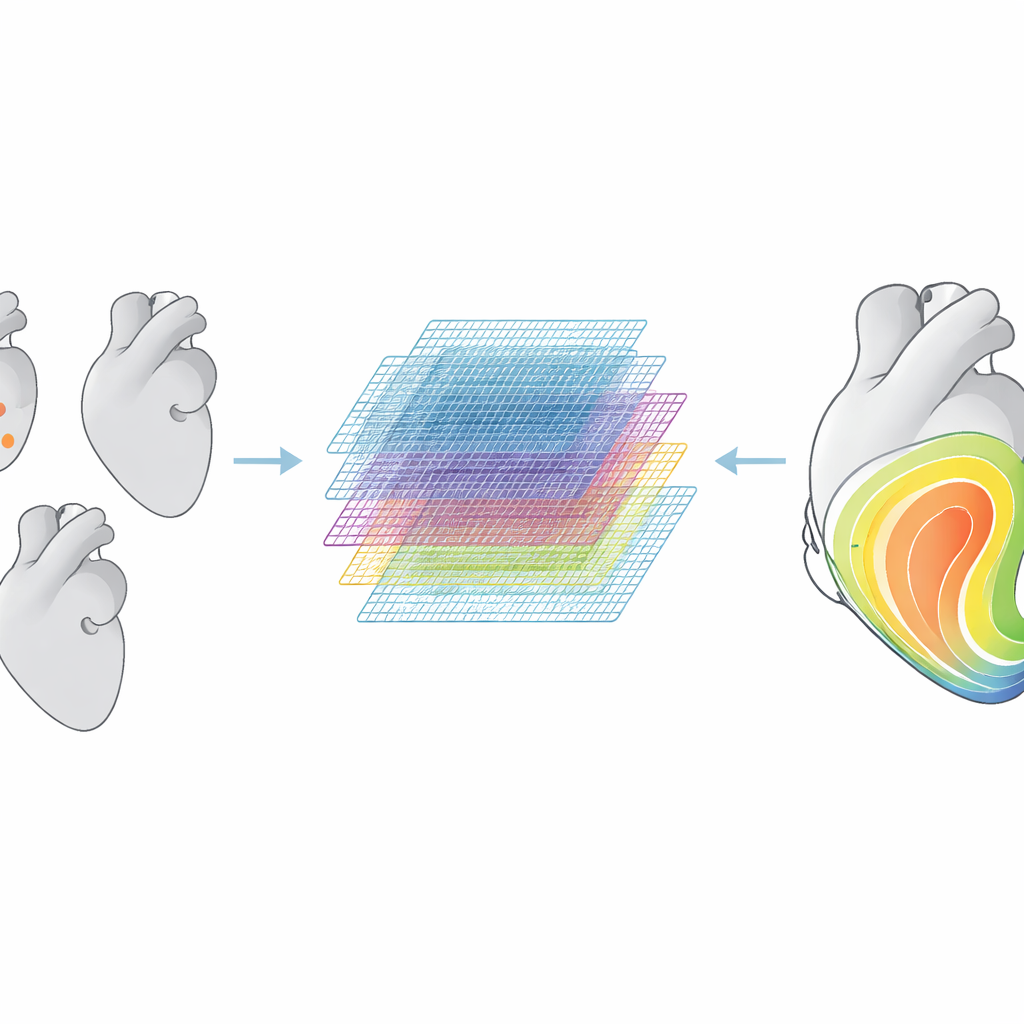
Building a virtual population of hearts
Because large collections of real patient data with full three‑dimensional activation maps are rare, the team generated their own virtual dataset. They started from 75 real left‑ventricle shapes spanning healthy and diseased hearts and used a statistical shape model to create 35,000 synthetic variants. For each one, they assigned realistic muscle fiber directions, chose one or two pacing sites, and varied tissue conductivity across a wide range. Detailed physics‑based simulations produced activation time maps for all these virtual hearts, which were then used to train and test the deep learning models. The models were also challenged with higher‑resolution meshes and with left‑ventricle geometries taken from two independent clinical cohorts to see how well they generalized beyond the synthetic training set.
How well did the models perform?
On synthetic hearts similar to those they were trained on, both models predicted activation maps with small errors, but the geometry‑informed neural operator was roughly twice as accurate as the graph neural network. When the researchers moved to real‑world heart shapes, the error grew for both models, and their performance became comparable. This indicates that the main limitation is not the power of the algorithms, but the gap between simplified training shapes and the full complexity of real patient anatomy. Still, the models could make predictions in milliseconds—much faster than the roughly ten minutes needed for a traditional simulation—making them attractive for tasks that require thousands of repeated evaluations, such as searching over many possible pacing sites.
Testing a virtual planning tool
The team then embedded the trained models into a proof‑of‑concept planning workflow for resynchronization therapy. Starting from a left‑ventricle shape and a noisy activation map meant to mimic clinical measurements, the workflow first worked backward to estimate the patient’s intrinsic pacing site and tissue conductivity. It then searched over the ventricle surface for a second pacing location that would minimize the overall activation time, a quantity linked in previous studies to better therapy response. Both deep learning models were able to recover key subject‑specific parameters from noisy data and to propose pacing sites that substantially shortened activation time, all within tens of seconds on a single graphics processor. The authors also built a web‑based interface where users can upload geometries, explore pacing scenarios, and run this optimization interactively.
What this means for patients
This work shows that carefully trained deep learning models can mimic detailed electrical simulations of the left ventricle across many shapes and pacing setups, and can do so fast enough to be used inside planning tools. While the current models rely on synthetic training data and consider only electrical behavior in the left ventricle, they lay the groundwork for more comprehensive digital twins that include both sides of the heart and its mechanical pumping action. With richer real‑world data and further refinement, such tools could one day help clinicians test many pacing strategies on a computer before entering the clinic, improving the chances that each patient receives a device configuration that truly restores their heart’s rhythm.
Citation: Naghavi, E., Wang, H., Ziaei-Rad, V. et al. Rapid prediction of cardiac activation in the left ventricle with geometric deep learning: a step towards cardiac resynchronization therapy planning. npj Digit. Med. 9, 225 (2026). https://doi.org/10.1038/s41746-026-02399-7
Keywords: cardiac resynchronization therapy, geometric deep learning, cardiac electrophysiology, patient-specific modeling, digital twin