Clear Sky Science · en
Algorithmic antibiotic decision-making in urinary tract infection using prescriber-informed prediction of treatment utility
Smarter Antibiotics for Common Infections
Urinary tract infections (UTIs) are among the most common reasons people receive antibiotics. Yet choosing the right drug is a balancing act: doctors must cure the infection, avoid serious side effects, and protect society from worsening antibiotic resistance. This study introduces a new kind of decision-making algorithm that aims to support clinicians by combining large-scale hospital data with the real-world priorities of doctors themselves, helping select antibiotics that are both effective for the patient and safer for the wider community.
The Problem with Powerful Drugs
Modern medicine relies on antibiotics, but overuse of broad, “big gun” drugs is driving a global crisis of antimicrobial resistance. The United Nations has set a target: by 2030, 70% of antibiotic use worldwide should come from narrower, first-line medicines known as World Health Organization “Access” antibiotics. In practice, many clinicians lean toward broader drugs, labeled “Watch” or “Reserve,” because they worry that a narrower option might fail in the face of resistant bacteria, especially when patients are very sick. As a result, we often trade long-term resistance risks for short-term peace of mind, without clear tools to judge when a safer, narrower drug would actually work just as well.
Teaching an Algorithm to Think Like a Clinician
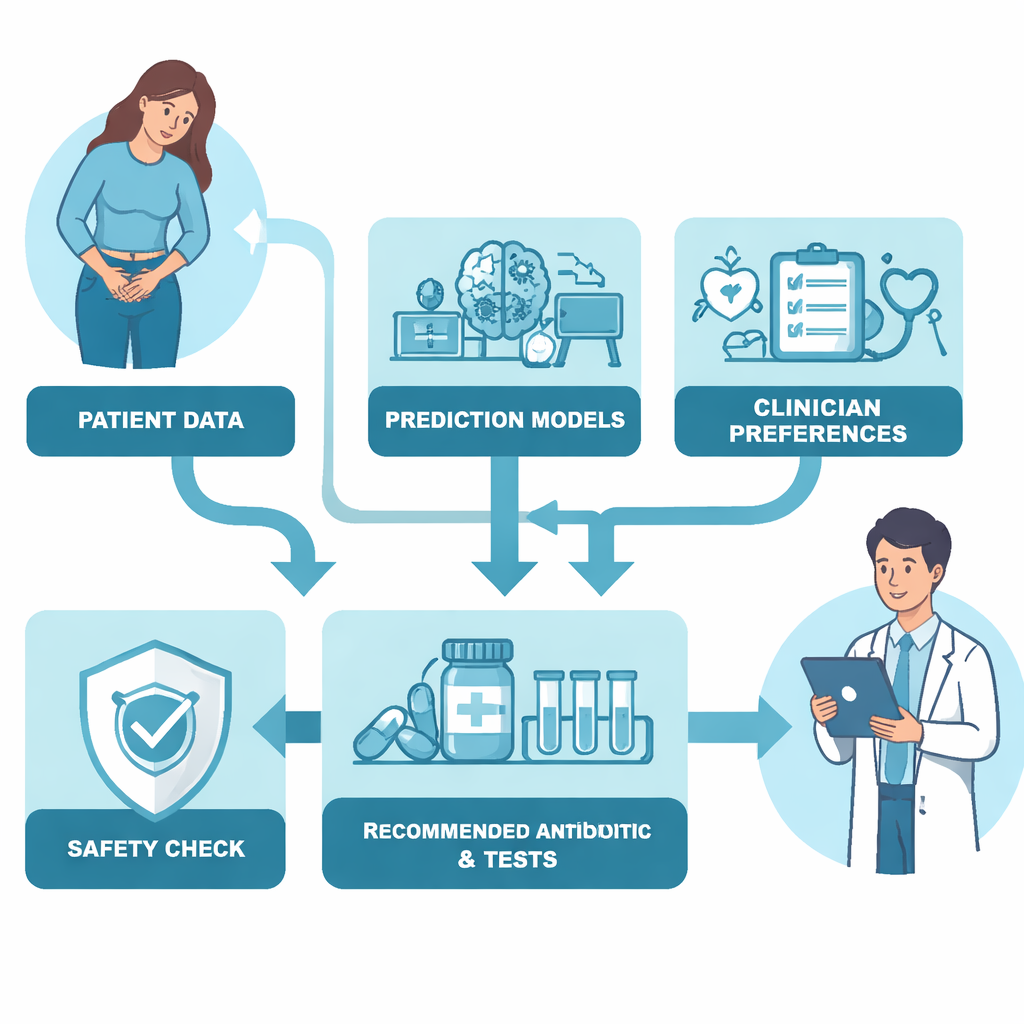
The researchers built an antibiotic decision-making algorithm focused on UTIs using detailed electronic records from nearly 94,000 hospitalized patients in Boston. First, they trained prediction models to estimate, for 13 different antibiotics, how likely the UTI-causing bacteria were to be susceptible, and how likely treatment was to trigger problems such as Clostridioides difficile infection or serious drug toxicity. Next, they asked 49 UK clinicians from multiple specialties to complete an online ranking exercise, choosing between fictional antibiotics that differed in properties like side-effect risk, suitability for UTIs, cost, whether they were Access or Watch/Reserve drugs, and whether they came in pill or intravenous form. By analyzing these rankings, the team quantified how strongly doctors valued each characteristic—for example, how much they preferred UTI-focused, low-toxicity, oral drugs over riskier or more powerful options.
Adding a Safety Net for Very Sick Patients
The final algorithm blended these two ingredients: data-driven predictions and clinician value judgments. For each patient, it calculated a “treatment value” for every antibiotic, taking into account the predicted chance the drug would work, the odds of serious side effects, its Access/Watch/Reserve category, and whether it was oral or intravenous. Crucially, the algorithm also built in a safety mechanism based on how unwell the patient appeared in the Emergency Department. As illness severity increased, the algorithm automatically placed more weight on hitting the infection hard and on having an intravenous option available. In other words, for mildly ill patients it tried to favor narrow, oral drugs, but as patients became sicker it became progressively more willing to use stronger, intravenous antibiotics to protect them from treatment failure.
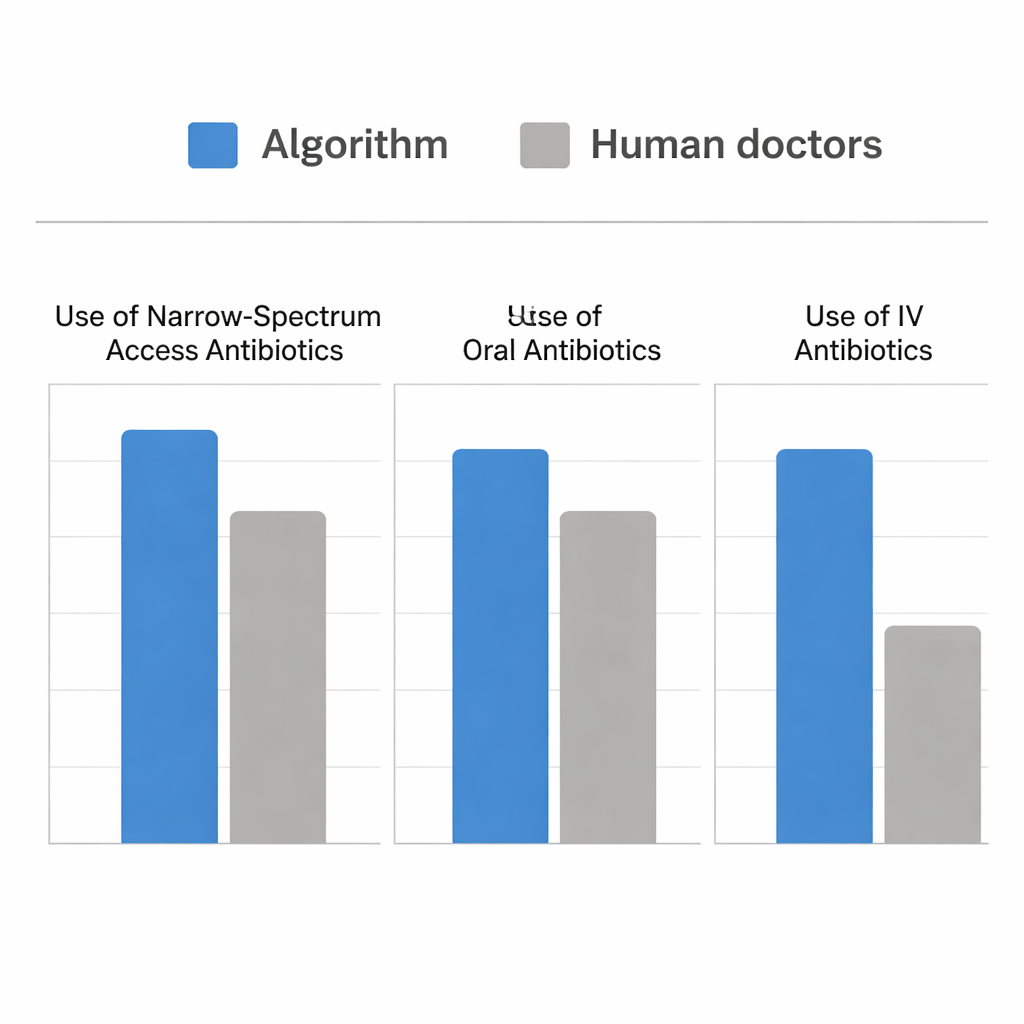
How the Algorithm Stacked Up Against Doctors
The team then ran a simulation using real Emergency Department cases where patients had UTIs and urine cultures. They compared the antibiotics that human clinicians had actually given with the drugs the algorithm would have chosen at the time the culture was sent. Both approaches were similarly good at picking an antibiotic that covered the patient’s bacteria at all. However, the algorithm did so while choosing far more narrow-spectrum Access drugs and far more oral treatments, and while using fewer intravenous antibiotics. For sicker patients, the algorithm behaved much like human prescribers, shifting appropriately toward intravenous and more powerful drugs. Where it differed was in recognizing extra opportunities—especially in moderately ill patients—to safely treat with oral, Access-category medicines such as nitrofurantoin and ampicillin-sulbactam, rather than defaulting to broader options.
What This Means for Everyday Care
For a lay reader, the key message is that this system does not replace doctors; instead, it acts like a calculator for complex risk–benefit trade-offs that clinicians already care about but cannot compute precisely in their heads. By marrying clinicians’ own priorities with large-scale evidence on how antibiotics perform, the algorithm can suggest treatments that are just as effective for individual patients but kinder to the body and to public health—favoring pills over drips when safe, and narrower drugs over last-resort ones whenever possible. If validated in more settings, such tools could help hospitals and health systems move closer to global targets for responsible antibiotic use, without sacrificing the safety of people who arrive critically ill with infection.
Citation: Howard, A., Green, P.L., Zhong, Y. et al. Algorithmic antibiotic decision-making in urinary tract infection using prescriber-informed prediction of treatment utility. npj Digit. Med. 9, 136 (2026). https://doi.org/10.1038/s41746-026-02369-z
Keywords: urinary tract infection, antibiotic stewardship, clinical decision support, antimicrobial resistance, machine learning in medicine