Clear Sky Science · en
Application and prospect of artificial intelligence in diagnostic imaging of prostate cancer
Why Smarter Scans Matter for Men’s Health
Prostate cancer is one of the most common cancers in men, and catching it early can mean the difference between a minor health problem and a life‑threatening disease. This review explains how artificial intelligence (AI) is being woven into modern medical scans to find prostate cancer sooner, judge how dangerous it is, and track whether treatments are working. For readers, it offers a window into how computers are becoming silent partners in the reading room, helping doctors make faster, more consistent decisions while raising new questions about data, fairness, and trust.
From Blurry Shadows to Data-Rich Pictures
Doctors rely on several imaging tools to look for prostate cancer. Ultrasound is quick and inexpensive but struggles to separate cancer from harmless enlargement or inflammation. Magnetic resonance imaging (MRI) gives detailed views of soft tissue and is now the go‑to test for clinically significant prostate cancer, yet it takes time to interpret and even experts often disagree. PET/CT scans, using tracers that bind to a protein called PSMA on cancer cells, excel at finding spread to bones and lymph nodes but can miss very small spots and are costly. AI does not replace these machines; instead, it sits on top of the images they produce. Algorithms first clean and segment the scans, then extract subtle patterns in brightness, texture, and shape. These invisible cues, combined with lab results like PSA blood levels, are used to train models that can flag suspicious areas, estimate cancer risk, and suggest where to biopsy or how to judge treatment response.
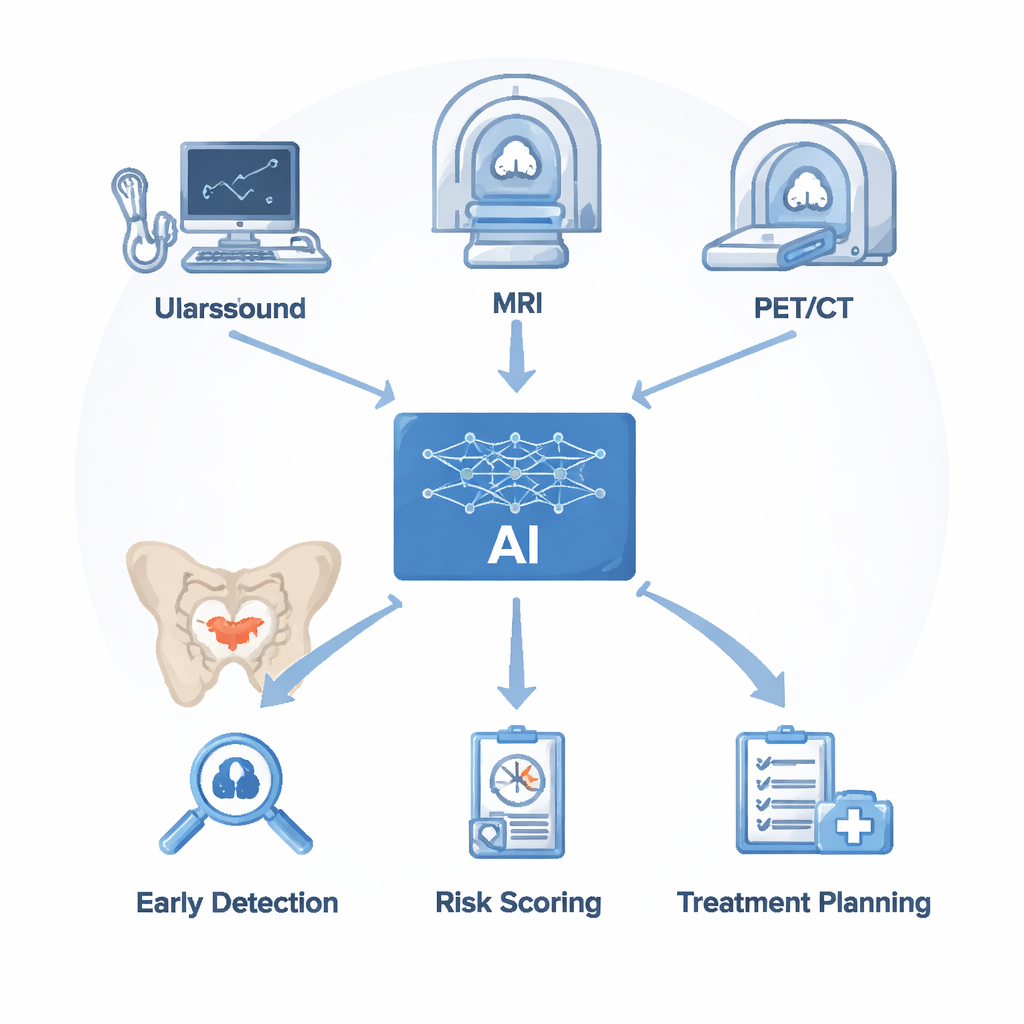
Teaching Computers to See What Radiologists Miss
In ultrasound, AI systems learn from hundreds of examples to spot cancerous regions that might blend into the background. Deep learning models can automatically locate the prostate and highlight likely tumors in real time, sometimes matching or surpassing experienced sonographers while maintaining speed and reducing missed small lesions. Radiomics approaches go a step further, converting ultrasound images into large sets of numbers that capture tissue patterns too complex for the human eye. Machine‑learning models built on these features have clearly outperformed traditional ultrasound and PSA alone, especially for men whose PSA levels fall into the confusing “gray zone,” where older methods often misclassify nearly half of cases. Similar ideas power AI on MRI, where advanced networks automatically outline the prostate and its internal zones, cutting manual contouring from about 20 minutes to barely over a minute and significantly improving agreement between different readers.
Sharper Risk Estimates and Smarter Treatment Tracking
AI shines most when it pulls together information from multiple MRI sequences that show anatomy, water motion, and blood flow. Transformer‑based models, adapted from language processing, fuse these inputs to produce probability maps of clinically important tumors. In testing on large patient groups, such systems have matched or exceeded senior radiologists, particularly for small tumors under a centimeter that humans often overlook. On dynamic contrast MRI, time‑series models can read the changing brightness curves of injected dye to estimate how leaky the tumor’s blood vessels are—an indicator linked to more aggressive disease and higher chances of recurrence. In PSMA PET/CT, three‑dimensional networks trained on full‑body scans automatically detect bone and lymph‑node metastases, measure total tumor burden, and relate it to how long patients stay free of progression. Other AI tools compare scans taken before and shortly after hormone or chemotherapy, predicting several‑month treatment outcomes far earlier than traditional rules based on simple uptake changes.
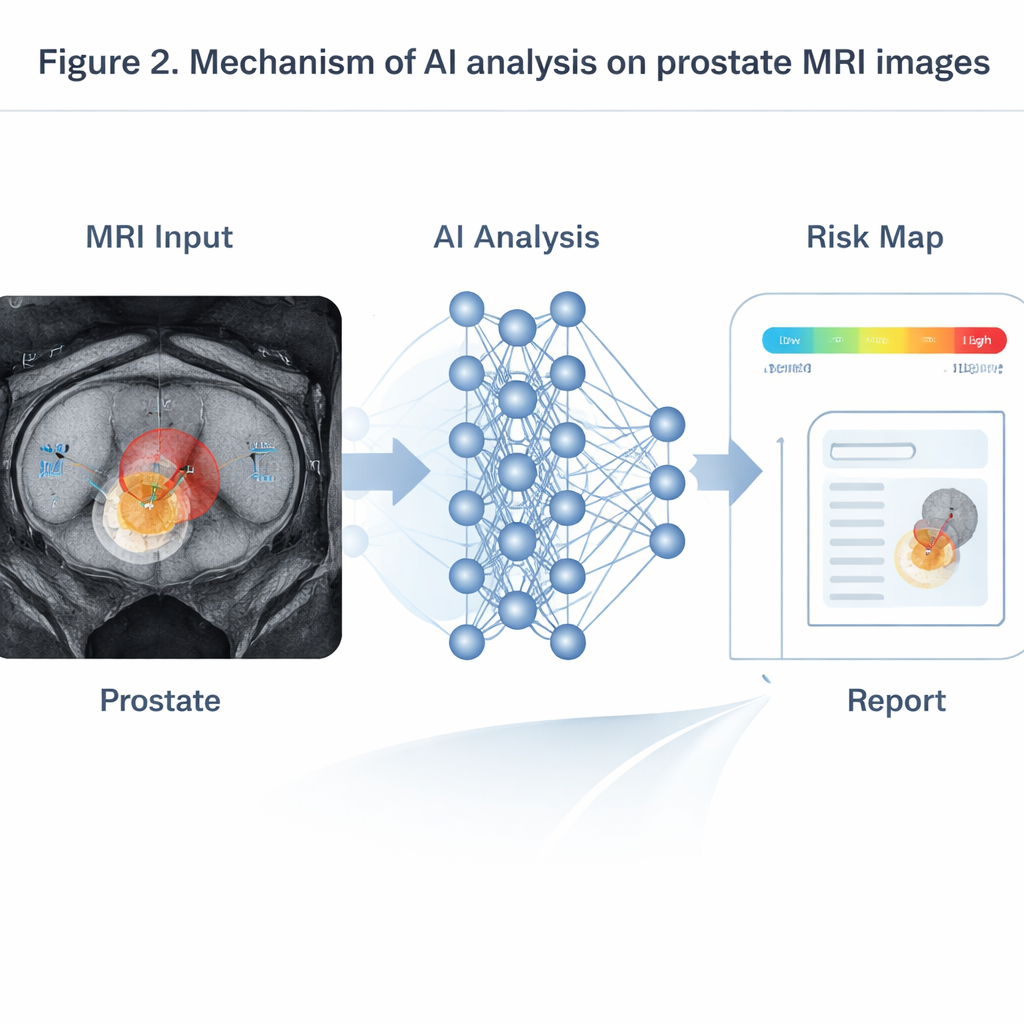
Roadblocks: Data Gaps, Black Boxes, and Fair Use
Despite these gains, real‑world deployment faces serious hurdles. High‑quality, well‑labeled imaging data are still limited and skewed toward large academic hospitals, while scans from smaller centers and diverse populations are under‑represented. Subtle differences between scanner brands, settings, and image quality can cause models trained in one place to stumble in another. Many powerful AI systems operate as “black boxes,” offering a risk score without a clear explanation, which undermines physician trust—especially when the computer disagrees with experience. The review also raises concerns about privacy, data‑sharing restrictions, and the risk that models might work less well for certain groups, potentially widening health disparities if not carefully monitored and corrected.
Building Trustworthy Partners in the Clinic
Looking ahead, the authors envision AI becoming a trusted teammate rather than a mysterious oracle. They outline efforts to build large, shared datasets across hospitals while preserving privacy through techniques like federated learning, where only model updates—not raw patient data—are exchanged. New “explainable AI” tools aim to show which image regions drove a decision and link them to known pathology, giving doctors tangible reasons to agree or disagree. Instead of one‑size‑fits‑all models, tailored systems will focus on specific jobs: screening in busy clinics, guiding biopsies, monitoring treatment, or following up high‑risk patients. Combining imaging with genetic and clinical data could further refine prognosis and personalize therapy. For patients, the bottom line is encouraging: if these technical, ethical, and regulatory challenges are handled well, AI‑enhanced imaging could mean earlier detection, fewer unnecessary biopsies, faster answers, and more tailored treatment plans for prostate cancer.
Citation: Wang, X., Zhong, S., Fang, K. et al. Application and prospect of artificial intelligence in diagnostic imaging of prostate cancer. npj Digit. Med. 9, 168 (2026). https://doi.org/10.1038/s41746-026-02354-6
Keywords: prostate cancer imaging, artificial intelligence, MRI and ultrasound, PSMA PET/CT, radiomics