Clear Sky Science · en
Integrating multi-omics and machine learning systematically deciphers cellular heterogeneity and fibrotic regulatory networks in the progression from MASLD to MASH
Why this liver disease research matters
Fatty liver disease linked to obesity and type 2 diabetes is now one of the most common liver problems worldwide. For many people the condition remains mild, but in a sizeable fraction it quietly worsens into an aggressive form with inflammation and scarring that can lead to cirrhosis and liver cancer. Doctors still struggle to predict who will progress and to catch this dangerous stage early without a biopsy. This study uses cutting-edge "multi-omics" tools and artificial intelligence to zoom in on individual liver cells, revealing a newly recognized immune cell type and signaling pathway that appear to drive harmful scarring, and proposing blood-based markers that could one day flag high‑risk patients noninvasively.

From simple fat buildup to dangerous liver damage
The researchers focused on the transition from metabolic dysfunction-associated steatotic liver disease (MASLD), often called fatty liver, to its more severe cousin, metabolic dysfunction-associated steatohepatitis (MASH). MASLD is extremely common, affecting more than a third of adults globally, especially those with obesity or diabetes. Most people with MASLD have fat deposits in the liver but little damage. In MASH, however, liver cells are injured, inflammation rises, and scar tissue forms. About one in five to one in three people with MASH will eventually develop cirrhosis or liver cancer. Current blood tests and scans cannot accurately see which microscopic cell changes are unfolding inside the organ, so they often miss the window for early intervention.
Peering at the liver one cell at a time
To tackle this problem, the team combined several powerful data types from public studies. They assembled single-cell RNA sequencing data from more than half a million individual liver cells, spatial transcriptomics that preserves where those cells sit in the tissue, bulk gene activity profiles from whole biopsies, and chromatin accessibility maps that show which stretches of DNA are open and active. Using sophisticated software, they grouped the cells into seven broad types, including liver cells, immune cells, and support cells called hepatic stellate cells. They then compared healthy livers, MASLD, and early and advanced MASH, and mapped how the mix of cells and their communication networks changed as disease worsened.
Scar-driving immune and support cells working together
A striking pattern emerged: immune cells known as monocytes and macrophages, together with hepatic stellate cells, were much more abundant in MASH than in earlier stages, and they tended to cluster in the same regions of diseased tissue. Delving deeper, the scientists broke stellate cells into five subtypes ranging from resting to fully activated and smooth-muscle-like forms. As livers shifted from MASLD to MASH, quiet stellate cells declined and activated, scar-producing stellate cells rose, tracing a clear path from a resting to a fibrotic state. Spatial analyses showed these activated stellate cells nestled close to the enriched macrophages, suggesting an intimate partnership in building liver scars.
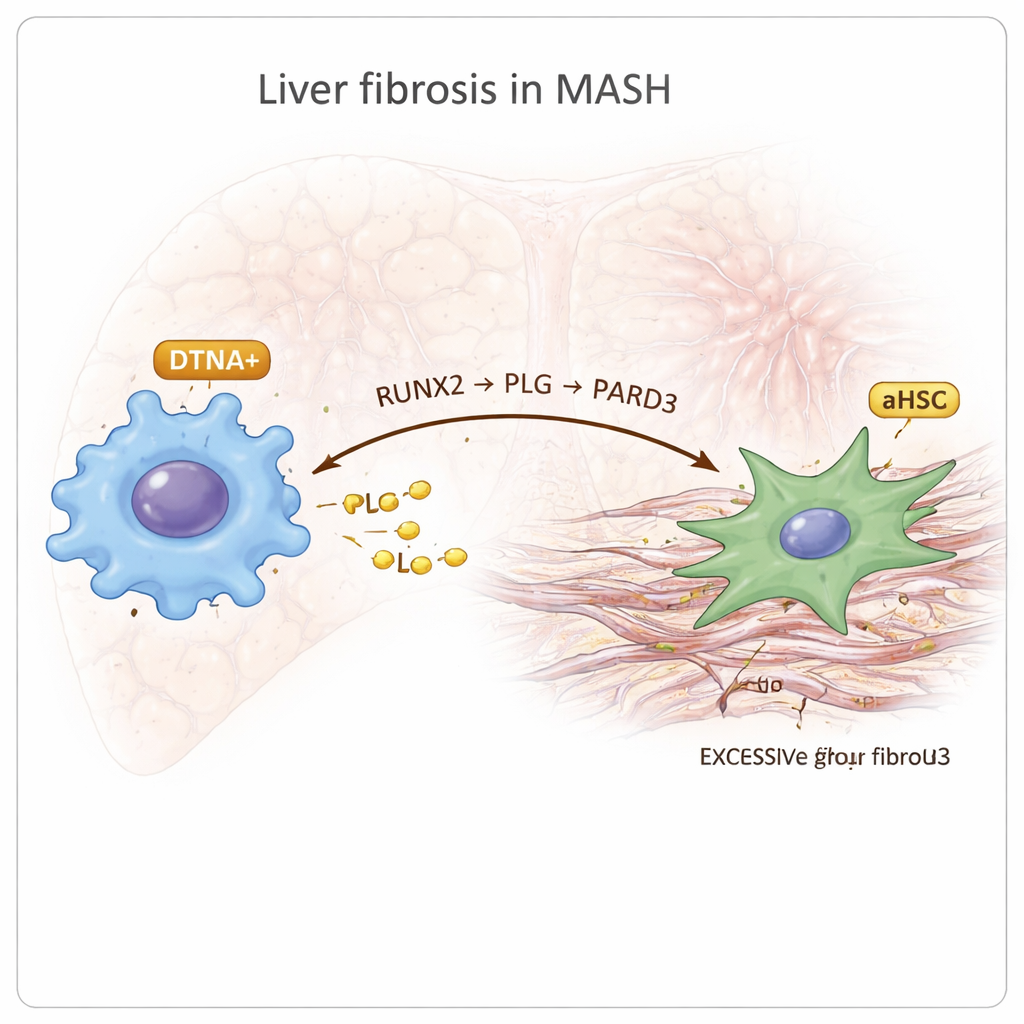
A newly highlighted macrophage type and a fibrotic signaling axis
Among the immune cells, the researchers discovered a distinct macrophage subgroup marked by a gene called DTNA that was specifically enriched in MASH. These DTNA-positive macrophages showed features of so-called M2 polarization, which is often linked to chronic inflammation and tissue remodeling, along with signs of low oxygen and strong inflammatory signaling. By reconstructing developmental “trajectories,” they inferred that these cells arise from resident Kupffer cells in the liver. Regulatory network analysis pointed to a transcription factor called RUNX2 as a master switch in these macrophages. Further cell–cell communication mapping suggested that DTNA-positive macrophages talk to activated stellate cells through a chain they call the RUNX2–PLG–PARD3 axis: RUNX2 boosts production of the protein PLG in macrophages, PLG then signals to stellate cells that express PARD3, and this interaction is linked to more intense fibrosis and altered blood clotting activity.
From cell maps to possible tests and treatments
To see whether these discoveries could help patients, the team trained and tested 113 different machine learning models on large bulk gene-expression datasets from people with MASLD or MASH. An Elastic Net model performed best, and across multiple independent cohorts one gene stood out as the strongest predictor: DTNA. Higher activity of DTNA and its partner genes, including RUNX2, consistently marked patients with MASH rather than simple fatty liver. The authors also screened existing drug databases and found several medicines that may act on the newly described RUNX2–PLG–PARD3 pathway, hinting at repurposing opportunities, though these ideas still require laboratory and clinical testing.
What it means for people with fatty liver disease
In everyday terms, this study shows that not all liver immune cells and support cells are alike, and that a particular partnership between a newly highlighted macrophage subtype and scar-forming stellate cells may be a key engine driving fatty liver toward dangerous scarring. The work suggests that measuring markers like DTNA in blood or tissue could one day help doctors identify which patients with fatty liver are at highest risk of progressing to MASH, without relying solely on invasive biopsies. It also spotlights a specific signaling route, the RUNX2–PLG–PARD3 axis, as a promising target for drugs aimed at slowing or reversing liver fibrosis. While these findings are still at the research stage and need further validation, they provide a sharper roadmap for turning complex cellular data into practical tools for early diagnosis and targeted treatment.
Citation: Wen, W., Liu, Z., Tan, W. et al. Integrating multi-omics and machine learning systematically deciphers cellular heterogeneity and fibrotic regulatory networks in the progression from MASLD to MASH. npj Digit. Med. 9, 167 (2026). https://doi.org/10.1038/s41746-026-02352-8
Keywords: fatty liver disease, MASH, macrophages, liver fibrosis, multi-omics