Clear Sky Science · en
Multicenter evaluation of interpretable AI for coronary artery disease diagnosis from PET biomarkers
Why Heart Scan AI Matters
Coronary artery disease, the buildup of plaque that can block blood flow to the heart, remains a leading cause of heart attacks and death worldwide. Modern imaging tests like PET/CT scans can reveal blood flow, heart function, and calcium deposits in the arteries, but the sheer amount of data can overwhelm even expert readers. This study explores how an interpretable artificial intelligence (AI) model can pull these pieces together into a single, easy-to-use score that helps doctors more accurately spot dangerous blockages—and show clearly which findings are driving its decision.
Bringing Many Heart Signals Into One Picture
When patients undergo a PET/CT heart scan, physicians can see how well blood flows through the heart muscle during rest and stress, how strongly the heart pumps, and how much calcium—an indicator of long-term plaque buildup—is present in the coronary arteries. Traditionally, clinicians look at these measurements one by one and then mentally combine them to decide whether arteries are likely to be narrowed. That mental integration is difficult and sometimes inconsistent, and there is no universally agreed-upon way to weigh blood flow, perfusion defects, and calcium scores together. The researchers set out to build an AI tool that could combine ten routinely available scan-based measurements, plus the patient’s sex, into a single probability that significant artery blockages are present.
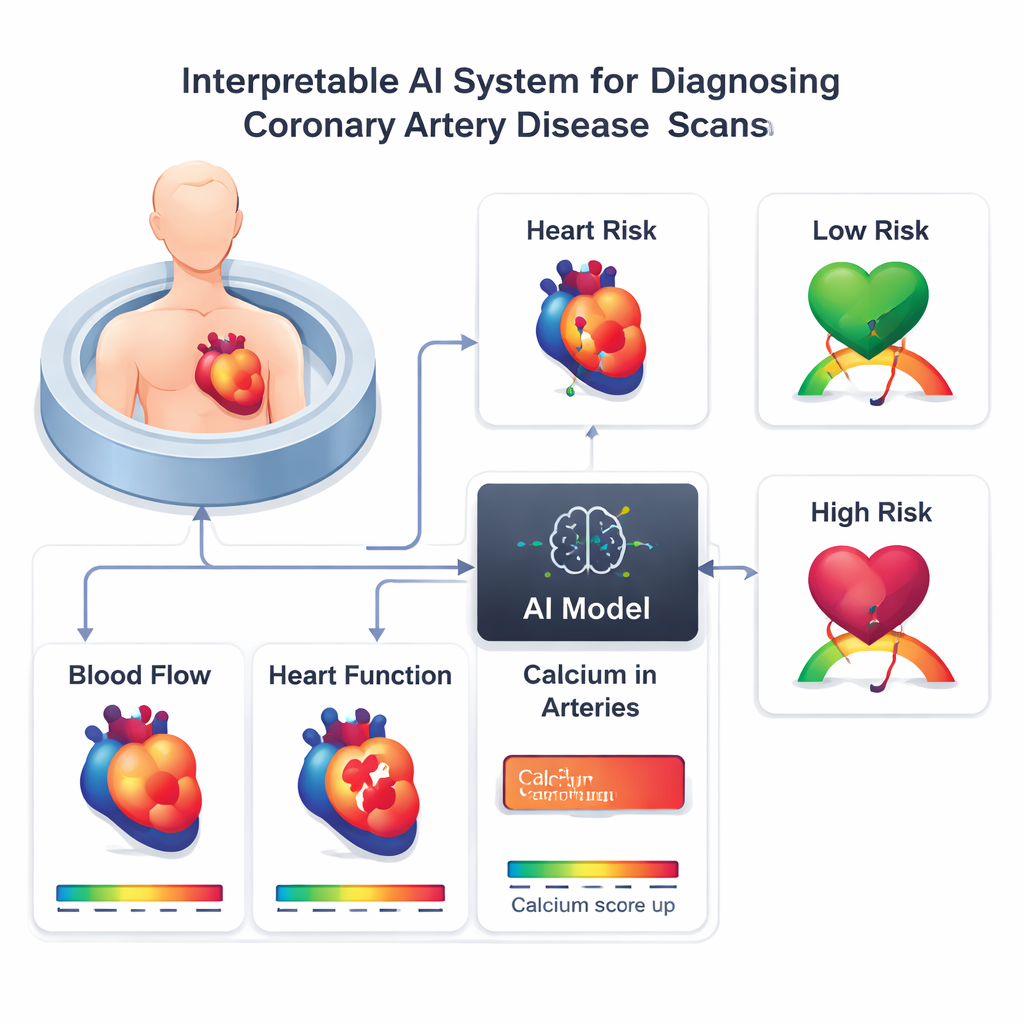
How the Study Was Carried Out
The team drew on a large international registry of 17,348 patients who had PET/CT heart scans. From this group, they focused on 1,664 people from four centers who had no prior heart attacks or bypass procedures and who went on to have invasive coronary angiography, the dye-based X-ray test used as the standard for confirming blocked arteries. One hospital’s data (386 patients) were used to train and fine-tune the AI model, while data from the other three hospitals (1,278 patients) were held back for true “outside” testing. The AI model, based on a machine-learning technique called XGBoost, used ten scan-derived features including blood flow under stress, blood flow reserve, the size of perfusion defects, calcium scores automatically measured from CT images, pumping strength, and a measure of how the heart’s size changes under stress.
How Well the AI Performed
On the external test group, where about half the patients truly had obstructive coronary artery disease, the AI model clearly outperformed both single measurements and experienced physicians. Using a common accuracy metric called the area under the receiver operating characteristic curve, the AI reached a value of 0.83, compared with 0.80 for expert clinical scores, 0.79 for the main perfusion measure, 0.75 for blood flow reserve, and 0.69 for calcium alone. When the researchers adjusted the cutoff so that the AI labeled about the same share of patients as “normal” as traditional thresholds, the AI picked up more high-risk patients with severe multi-vessel disease. Its performance was stable in men and women, younger and older patients, and in people with and without obesity, suggesting the approach is broadly applicable.
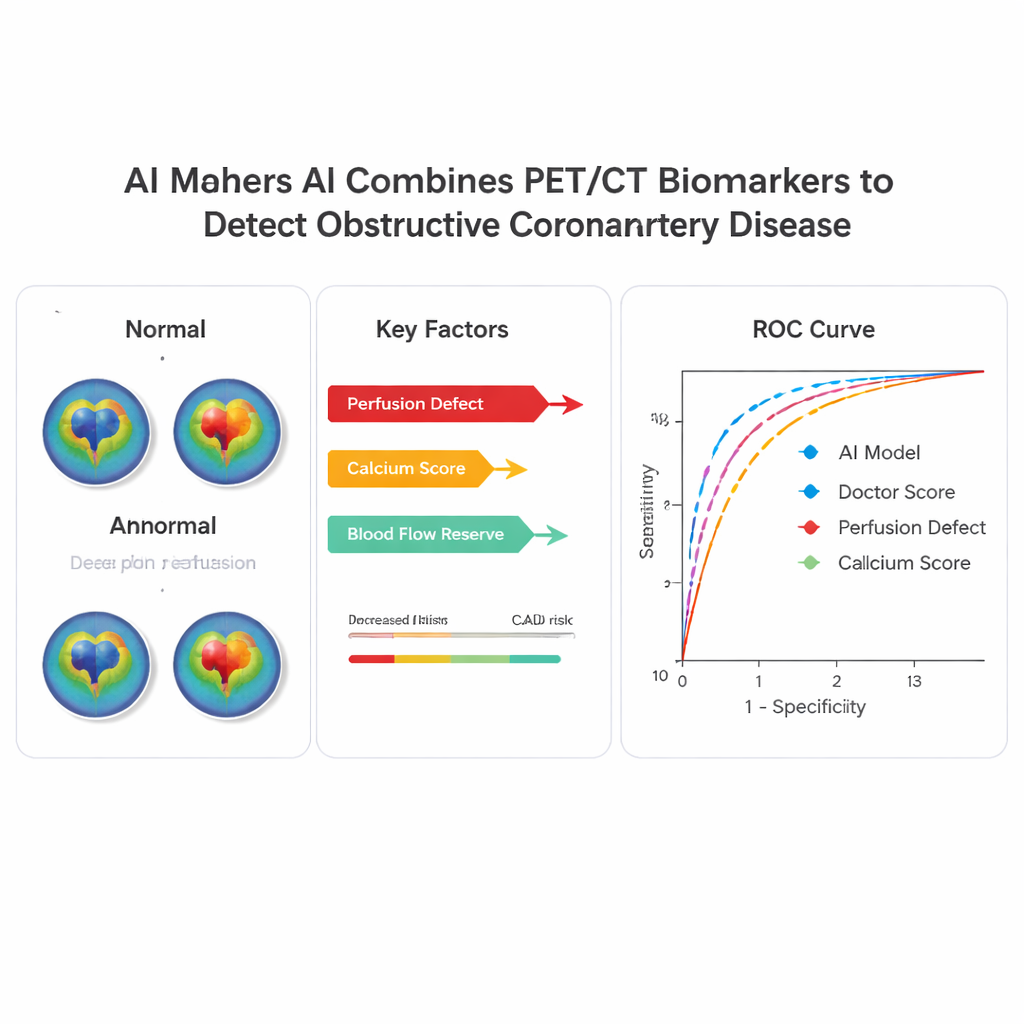
Making the AI’s Logic Visible
A major concern with advanced algorithms in medicine is that they can behave like “black boxes,” offering predictions without explanations. To avoid this, the authors used a technique called SHAP analysis to show which scan features most strongly influenced each individual prediction. Across the study, the most important drivers were the amount of heart muscle with reduced blood flow, the overall calcium burden, and blood flow reserve. For example, in one showcased patient with severely reduced flow and high-risk angiography findings, the AI assigned a high disease probability driven mainly by poor flow reserve. In another patient with borderline perfusion scores but normal blood flow and zero calcium, the AI correctly indicated low likelihood of disease, in contrast to a more alarmed physician reading. Such case-by-case explanations could help clinicians trust and verify AI-supported decisions.
What This Means for Patients
This work introduces the first multicenter, externally tested AI system that combines standard PET/CT heart scan measurements and automated calcium scoring to diagnose coronary artery disease. The model provides a single, interpretable risk estimate that often surpasses the accuracy of expert readers while highlighting the specific scan features behind each judgment. Although the tool is not yet approved for routine clinical use and further prospective studies are needed, it points toward a future in which heart imaging results are summarized into clear, personalized risk scores that help doctors more confidently decide who needs invasive testing or aggressive treatment—and who can safely avoid it.
Citation: Zhang, W., Kwiecinski, J., Shanbhag, A. et al. Multicenter evaluation of interpretable AI for coronary artery disease diagnosis from PET biomarkers. npj Digit. Med. 9, 154 (2026). https://doi.org/10.1038/s41746-026-02338-6
Keywords: coronary artery disease, cardiac PET CT, artificial intelligence, calcium scoring, myocardial blood flow