Clear Sky Science · en
Pressure-tuned plethora of ferroelectric phases in CuInP2S6
Why squeezing crystals can spark new behavior
Most of the technology around us depends on materials responding in useful ways when we press on them, heat them, or run current through them. This study looks at a layered crystal called CuInP2S6, which already behaves like a tiny built-in battery: it has an internal electric polarization at room temperature. By squeezing this crystal to very high pressures, the researchers discovered a surprising sequence of structural changes that first strengthen and then weaken this built-in electric alignment, and eventually drive the material all the way to a metallic state. Understanding this journey from “electric crystal” to “metal under pressure” may help design new switches, sensors, and low-power electronic components.
Stacked sheets that carry a built-in voltage
CuInP2S6 belongs to a family of two-dimensional, sheet-like materials where atoms are arranged in layers with weak gaps between them. At normal conditions, its atoms sit in a slightly asymmetric pattern: positively charged copper ions are shifted off-center between surrounding sulfur atoms. This off-center shift gives each layer a tiny electric dipole, and all of these dipoles add up to a net polarization pointing out of the layers. Because the structure lacks a center of symmetry, the material is ferroelectric, meaning that its internal polarization can in principle be switched by an external electric field. That alone makes it interesting for ultra-thin memory and logic devices.
Probing crystals with light and extreme pressure
To see how this built-in polarization behaves when the crystal is compressed, the team combined several advanced techniques. They shone infrared and visible laser light through and off the sample to monitor how its atoms vibrate (using infrared absorption and Raman scattering), tracked how X-rays diffract from the crystal lattice to follow structural changes, and measured its electrical resistance to watch for the onset of metallic behavior. At the same time, they used first-principles quantum calculations to map the energy of different possible atomic arrangements and to predict how the polarization should evolve with pressure. This combination allowed them to connect subtle changes in vibrational “fingerprints” with specific rearrangements of atoms and with shifts in electrical properties.
A cascade of polar phases as pressure rises
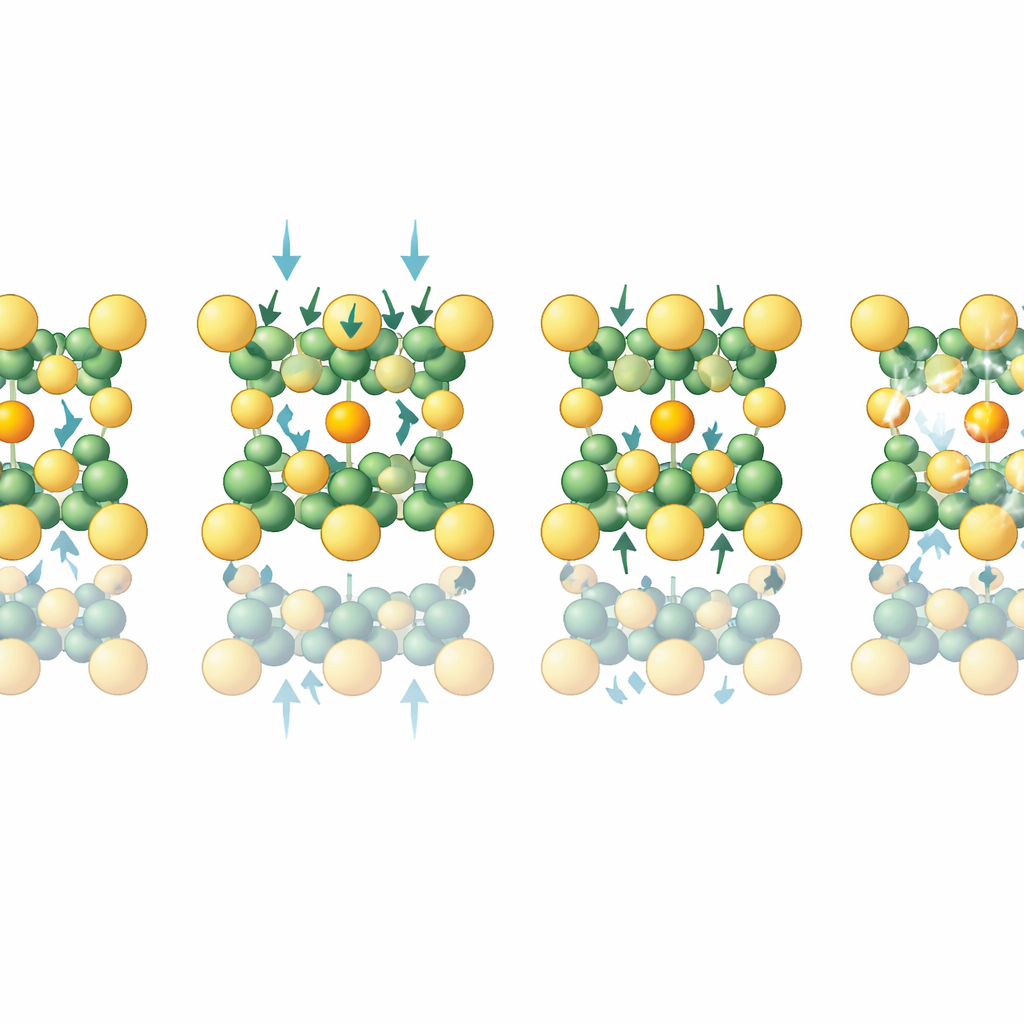
Contrary to the common expectation that compression makes crystals more symmetric and less polar, CuInP2S6 retains polarity through a sequence of phases. Starting from a low-pressure monoclinic structure, the material first experiences a remarkable increase in polarization as copper ions move further off-center. Around a few gigapascals, it then undergoes a structural transition to a higher-symmetry trigonal arrangement. Careful symmetry analysis of the vibrational spectra and X-ray patterns shows that this high-pressure phase still lacks inversion symmetry and belongs to a polar space group. At even higher pressure, a second trigonal phase appears in which the sulfur atoms reorganize from a nearly prismatic to a more octahedral environment around the metal ions. Throughout these changes the material remains polar, but the magnitude of polarization gradually falls as the copper ions’ preferred positions shift.
From insulator to metal in a squeezed crystal
The researchers also traced how the material’s ability to carry charge evolves under pressure. While related compounds in the same family turn metallic at much lower pressures, this crystal stubbornly remains a semiconductor well past tens of gigapascals. Only near about 63 gigapascals—more than 600,000 times atmospheric pressure—does it finally show clear signatures of true metallic behavior. In the infrared spectra, this shows up as a strong, low-energy electronic response that screens and eventually wipes out the sharp vibrational features. The unusually high pressure needed to reach this metallic state is likely tied to disorder and mobility of the copper ions, which complicate the path from an ordered ferroelectric to a simple metal.
Moving ions and the energy landscape
A key piece of the puzzle is the motion of copper ions within and between the layers. Detailed analysis of how vibrational peaks broaden with pressure reveals that certain modes, particularly those involving out-of-plane motion, lose coherence as the copper ions become more mobile and their positions more disordered. Quantum calculations show that small shifts of these ions can move the system between low- and high-polarization states, and that pressure reshapes the energy landscape so that different configurations become favored at different stages. X-ray measurements support this picture, indicating gradual changes in copper site occupancy and hints of local distortions and nanoscale sliding between layers.
What this means for future devices
Altogether, the work establishes a detailed, pressure-driven roadmap for how CuInP2S6 evolves from a ferroelectric insulator through multiple polar crystal structures to a true metal. For non-specialists, the key takeaway is that pushing on this layered material does not simply turn its polarization off; instead, it first enhances, then reshapes, and only at extreme pressures finally erases the ferroelectric behavior. By linking specific atomic motions and structural patterns to changes in polarization and conductivity, the study provides a foundation for engineering related materials where electric states can be tuned by mechanical stress, offering new knobs for future nanoelectronic and energy-efficient switching technologies.
Citation: Shah, S., Mohammadi, P., Singidas, B.G. et al. Pressure-tuned plethora of ferroelectric phases in CuInP2S6. npj 2D Mater Appl 10, 40 (2026). https://doi.org/10.1038/s41699-026-00663-8
Keywords: ferroelectricity, high pressure, two-dimensional materials, ion migration, insulator–metal transition