Clear Sky Science · en
Facile molten salt synthesis of bimetallic NiFe-Ti3C2Tx MXene nano-hybrid as an efficient oxygen evolution electrocatalyst
Turning Water into Fuel with Cheaper Materials
Hydrogen is often hailed as a clean fuel of the future, but making it efficiently and affordably is still a major challenge. This paper describes a new kind of catalyst—built from inexpensive metals nickel and iron on top of an ultrathin material called a MXene—that helps the oxygen side of water splitting run faster, bringing practical, low‑cost hydrogen production a step closer.
Why We Need Better Water-Splitting Helpers
To replace fossil fuels, we can use surplus electricity from wind and solar farms to split water into hydrogen and oxygen. The problem is that the oxygen‑forming half of the reaction, called the oxygen evolution reaction, wastes much of that valuable electricity. Today’s best catalysts for this step often rely on rare and costly precious metals. The authors aim to solve this by combining abundant metals with a highly conductive support so that water can be split efficiently without relying on scarce elements.
A Layered Platform for Active Metals
At the heart of the work is a family of two‑dimensional materials known as MXenes, which resemble stacks of atom‑thin metal carbide sheets. Instead of using the traditional, hazardous hydrofluoric acid route, the team adopts a safer “molten salt” process. They start from a layered compound called a MAX phase and etch out one of its elements using a hot mixture of nickel and iron chloride salts. In one step, this both peels the structure into MXene sheets and deposits a thin metallic alloy of nickel and iron directly onto their surfaces, forming a tightly bonded nano‑hybrid.
Finding the Sweet Spot in Metal Mixing
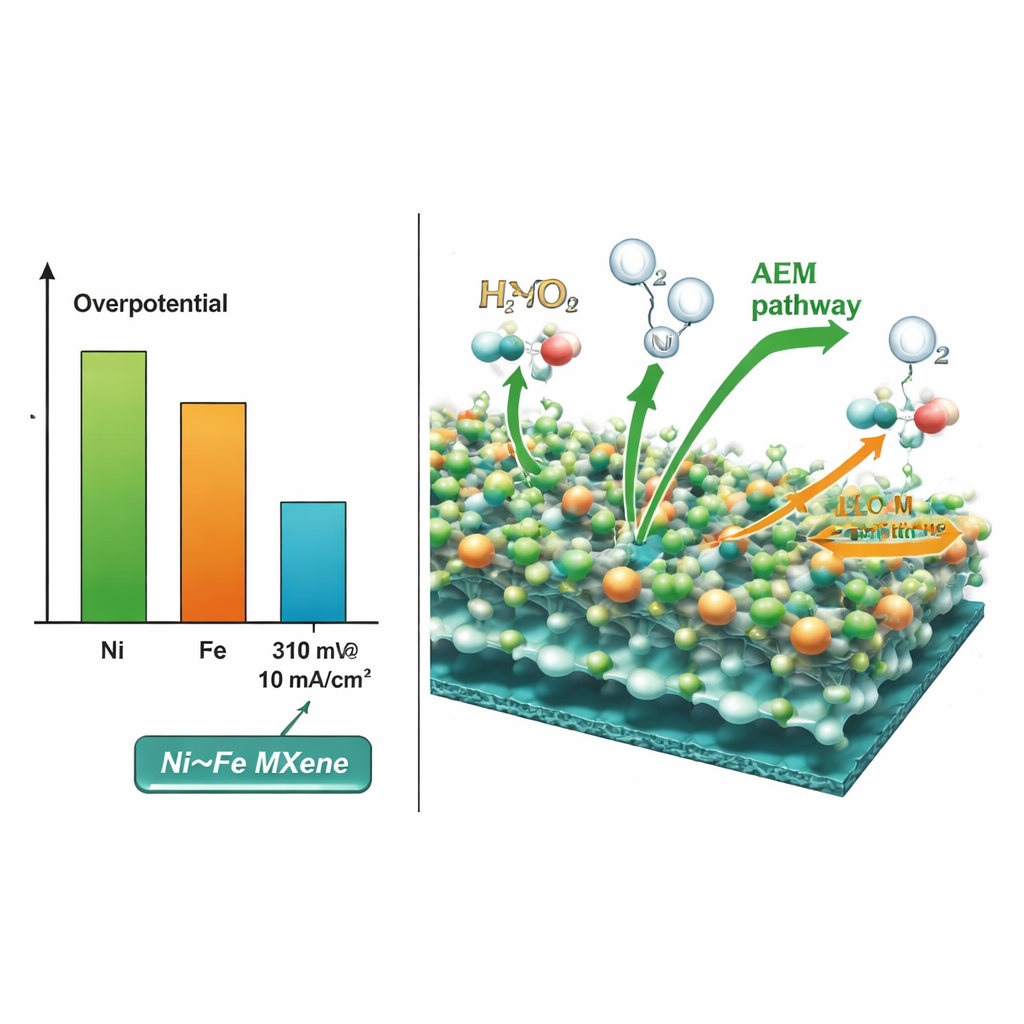
By tuning the ratio of nickel to iron in the molten salt, the researchers create a series of hybrids and test how well each drives oxygen formation in alkaline solution. Detailed measurements show that a 1:1 mixture of nickel and iron gives the best performance: it reaches a useful current with 310 millivolts of extra voltage and has a low Tafel slope, meaning the reaction rate rises quickly as voltage increases. Electron microscopy and X‑ray techniques reveal that this optimal material consists of ultrathin MXene flakes coated at their edges with a nanometer‑scale nickel–iron alloy layer. Electrochemical tests further show that both metals are electrochemically active, but nickel plays the leading role, while iron subtly tunes the behavior of the nickel sites.
Peeking into How Oxygen Is Born
To understand why the 1:1 alloy works so well, the team combines in‑situ infrared spectroscopy with computer simulations. Under operating conditions, the catalyst surface rearranges into nickel–iron oxyhydroxide species and shows clear signs of oxygen‑containing intermediates. Quantum‑mechanical calculations then compare two possible routes by which water molecules can join to form oxygen. They find that a pathway where reaction steps occur mainly on adsorbed species at nickel sites (the “adsorbate evolution” route) requires less energy than a route involving oxygen atoms from the underlying lattice. This helps explain both the superior activity of nickel relative to iron and the overall efficiency of the alloyed surface.
What This Means for Future Clean Energy Devices
In simple terms, the study introduces a relatively safe, scalable way to make a finely tuned nickel–iron coating on a conductive, ultrathin support, and shows that this design significantly improves the difficult oxygen‑forming step in water splitting. While some degradation of the MXene support still occurs over long operation, the work points the way toward robust, low‑cost catalysts that could make hydrogen production from renewable electricity more efficient and more affordable.
Citation: Kruger, D.D., Recio, F.J., Wlazło, M. et al. Facile molten salt synthesis of bimetallic NiFe-Ti3C2Tx MXene nano-hybrid as an efficient oxygen evolution electrocatalyst. npj 2D Mater Appl 10, 24 (2026). https://doi.org/10.1038/s41699-026-00660-x
Keywords: water electrolysis, oxygen evolution catalyst, MXene materials, nickel iron alloy, green hydrogen