Clear Sky Science · en
Multimodal plasma and urinary cell-free DNA profiling improves risk stratification in newly diagnosed prostate cancer
Why this research matters for men’s health
Prostate cancer is one of the most common cancers in men, yet doctors still struggle to tell which early tumors are harmless and which will turn dangerous. Many men with slightly raised PSA levels face anxious waits, repeated scans, and sometimes painful biopsies that turn out not to be needed. This study explores whether a simple blood and urine test can read tiny traces of tumor DNA to better flag men who truly have aggressive disease, potentially reducing unnecessary procedures and guiding treatment more precisely.
Looking for clues in blood and urine
Instead of cutting out tissue, the researchers focused on “liquid biopsies” – samples of blood plasma and urine that naturally carry fragments of DNA shed from cells, including cancer cells. They studied 73 men with newly diagnosed prostate cancer and 36 cancer-free men who were being checked for the disease. Most patients had tumors confined to the prostate, while a smaller group already had spread to lymph nodes or distant organs. From each sample, the team sequenced the cell-free DNA to examine its overall structure, its large-scale chromosomal changes, and its chemical tags known as methylation marks, which often shift early in cancer development.
Reading multiple signals from the same DNA
The researchers did not rely on a single type of DNA signal. They looked at four kinds of information at once: how scrambled the chromosomes were, how much of the DNA carried prostate-cancer–like methylation patterns, and how the DNA fragments were chopped up by the body in both blood and urine. By first defining cancer-specific methylation patterns in prostate tumor tissue and confirming these in outside datasets, they built a score that summarized how “cancer-like” the methylation was in each liquid biopsy. For chromosomal changes, they estimated how much of the cell-free DNA was likely coming from tumor cells and calculated a separate instability score. They also measured subtle rhythms in fragment lengths in plasma and a narrow peak of fragment sizes in urine that tend to change when tumor DNA is present.
What the liquid biopsies revealed
Each single type of measurement, taken alone, caught tumor DNA in only a minority of patients, especially when the cancer was still localized. However, combining information across both blood and urine, and across all four DNA features, raised the overall detection rate to 45 percent of newly diagnosed patients, including 42 percent of men whose tumors had not yet spread and 56 percent of those with advanced disease. Signals were usually stronger in patients with metastases, who showed greater chromosomal instability and higher cancer-like methylation scores, particularly in urine. Importantly, tumor DNA was found even in many men whose PSA values were below 10 ng/mL, a range often considered a “gray zone” where decisions are hardest.
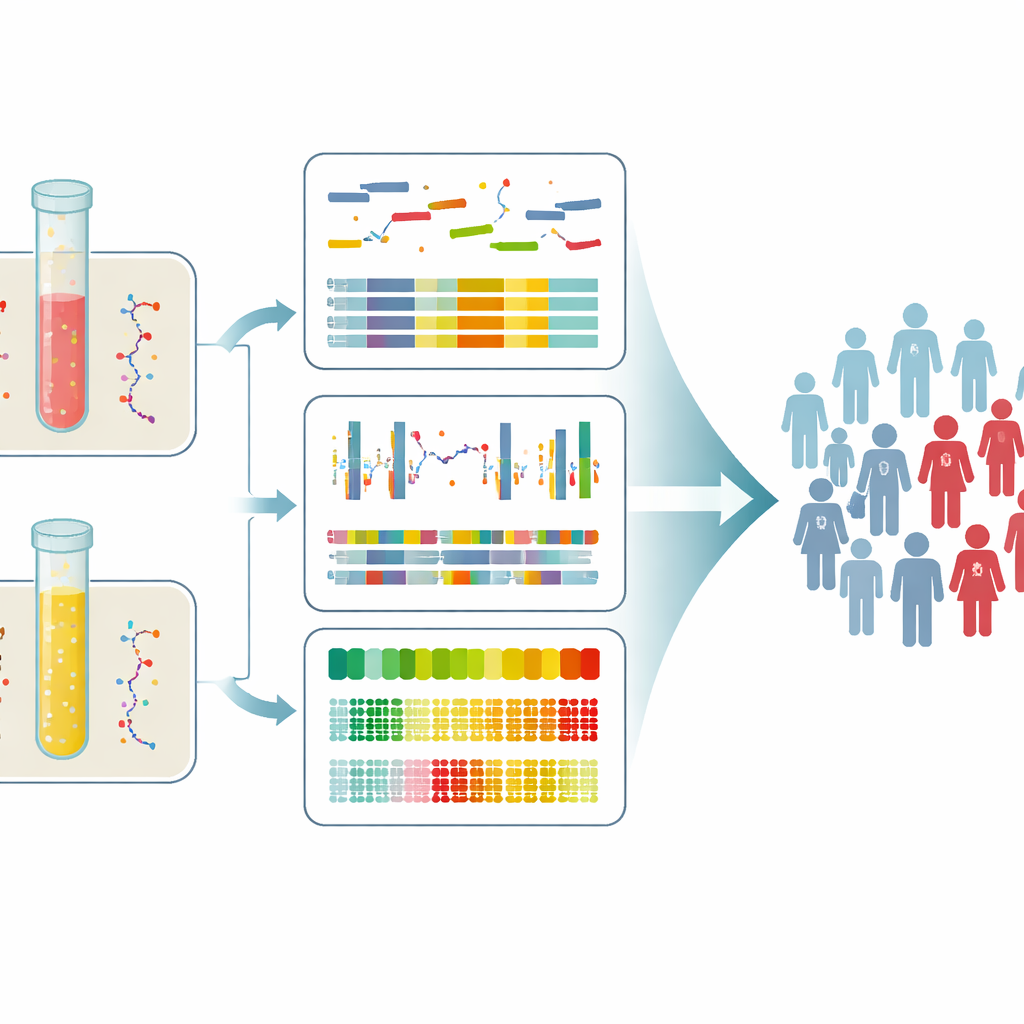
Helping separate low-risk from high-risk cases
The team then tested whether adding these DNA features could improve on PSA alone when trying to distinguish men with advanced cancer from those with disease still confined to the prostate. Among men with intermediate PSA levels, models that used only PSA performed barely better than chance. When the researchers added their combined liquid biopsy signals – the strongest chromosomal, methylation, and fragmentation features from either plasma or urine – the ability to separate localized from advanced disease improved sharply. A similar multimodal model also helped tell cancer patients apart from men with noncancerous PSA elevations, suggesting that this approach could sharpen early screening and reduce unnecessary biopsies.
What this means for patients and clinics
This work is an early proof-of-concept rather than a ready-made test, and not every man with advanced cancer had detectable tumor DNA. Still, it shows that looking at several DNA features at once, in both blood and urine, can reveal a richer picture of prostate cancer than PSA alone. In the future, such multimodal liquid biopsy panels could be added to standard blood tests to better identify men at high risk, especially when PSA results are unclear. That could mean fewer needless procedures for low-risk patients and timelier, more tailored treatment for those whose cancers are poised to spread.
Citation: Riediger, A.L., Eickelschulte, S., Janke, F. et al. Multimodal plasma and urinary cell-free DNA profiling improves risk stratification in newly diagnosed prostate cancer. npj Precis. Onc. 10, 115 (2026). https://doi.org/10.1038/s41698-026-01343-y
Keywords: prostate cancer, liquid biopsy, cell-free DNA, early detection, cancer risk stratification