Clear Sky Science · en
COL3A1high cancer-associated fibroblasts orchestrate metabolic and immune microenvironments to confer chemoresistance in breast cancer
Why some breast tumors shrug off chemotherapy
Chemotherapy can save lives in breast cancer, yet many patients find that their tumors stop responding or never respond well in the first place. This study looks beyond cancer cells themselves to the neighborhood they live in and asks: which nearby cells help tumors survive treatment, and how do they do it? The answer centers on a special group of support cells that rewire both tumor nutrition and local immunity to shield cancer cells from chemotherapy.
The hidden helpers around the tumor
Breast tumors are more than just cancer cells; they are mixed communities that include blood vessels, immune cells, and connective tissue cells called fibroblasts. When fibroblasts live in a tumor, they are known as cancer-associated fibroblasts, or CAFs, and they can either restrain or promote cancer. By combining several powerful genetic profiling methods on patient samples—single-cell, spatial, and bulk analyses—along with lab and animal experiments, the researchers pinpointed a CAF subgroup that produces high levels of a molecule called COL3A1, a component of the tissue scaffold. Patients whose tumors contained more of these COL3A1-high CAFs were more likely to have poor responses to chemotherapy and worse survival.
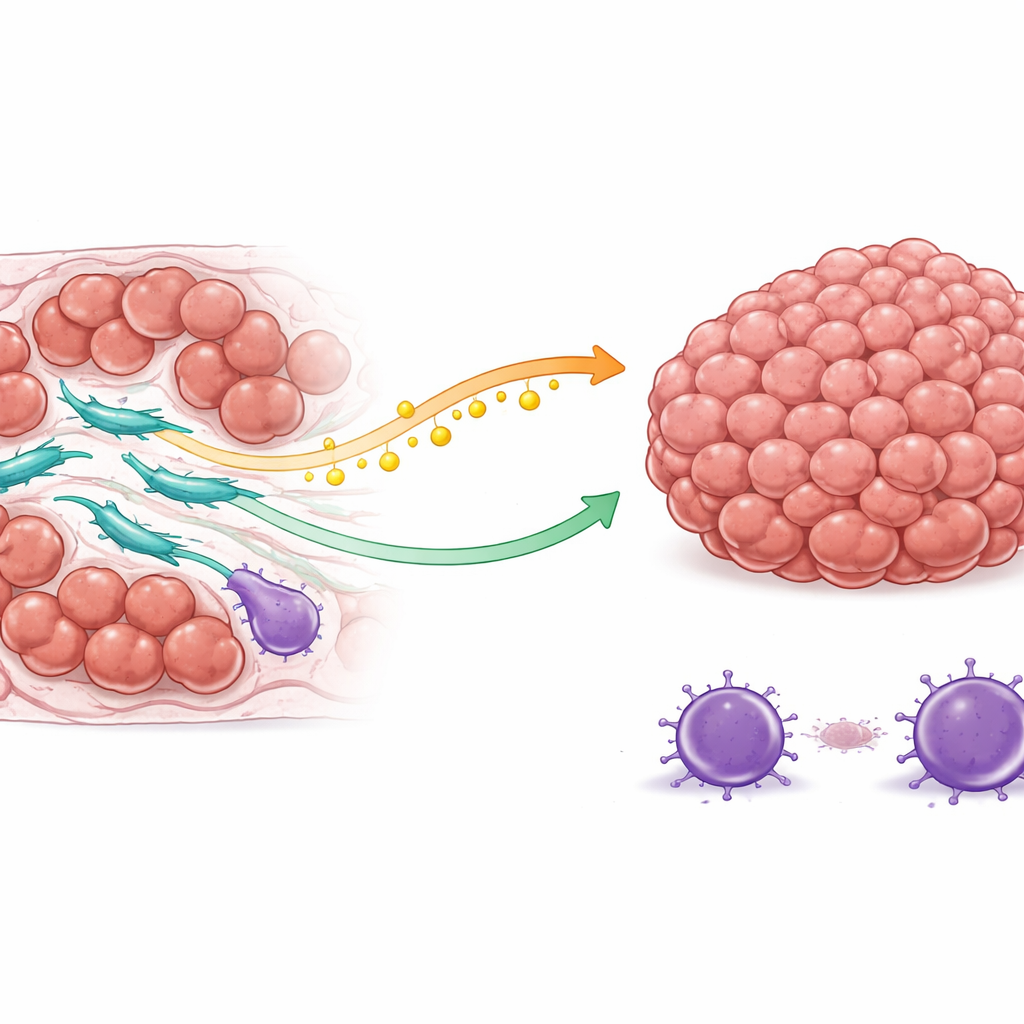
A two-pronged shield: fuel and protection
The team discovered that COL3A1-high CAFs protect cancer cells in two main ways. First, they change how they handle fats. These fibroblasts boost the activity of an enzyme that makes oleic acid, a type of fat. Instead of keeping this fuel, they release it into the tumor neighborhood. Cancer cells pick up this oleic acid through a surface protein and, in response, switch on an internal survival pathway known for helping cells resist death signals. As a result, when chemotherapy tries to trigger self-destruct programs in cancer cells, those cells are better equipped to resist and keep dividing.
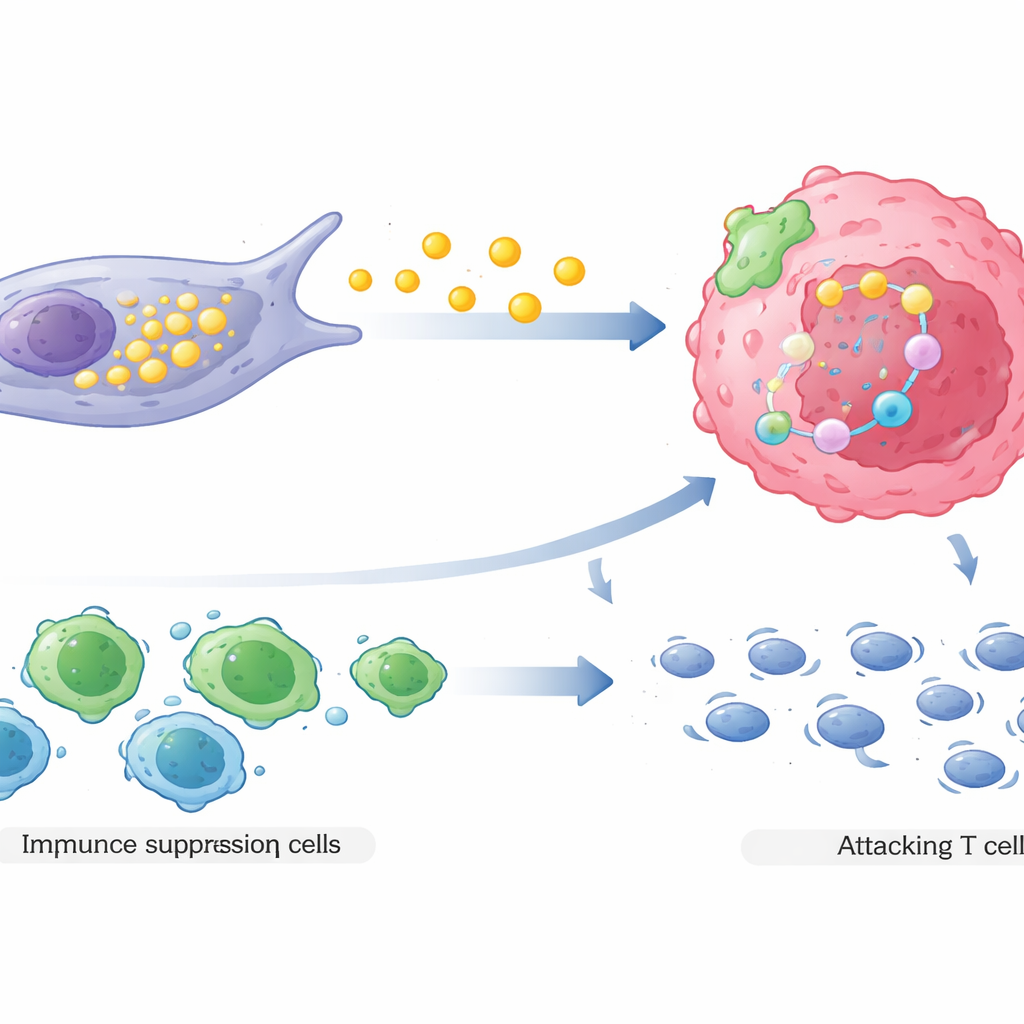
Shaping an unfriendly immune neighborhood
Second, COL3A1-high CAFs reshape the local immune landscape in favor of the tumor. Through complex signaling interactions, they attract regulatory T cells, a type of immune cell that dampens immune attacks, and at the same time discourage or weaken killer T cells that would normally attack cancer. Spatial mapping of real patient tumors showed that these fibroblasts tend to sit close to both drug-resistant cancer cells and suppressive immune cells, while effective killer T cells are pushed farther away. This arrangement creates a sheltered zone where cancer cells face fewer immune threats just as they are also becoming harder to kill with drugs.
Proof from cells, mice, and patients
To test cause and effect, the researchers reduced COL3A1 in CAFs grown in the lab. When breast cancer cells were grown alongside these altered fibroblasts, they formed fewer invasive structures, were more easily killed by the chemotherapy drug paclitaxel, and showed more signs of programmed cell death. In mice implanted with mixtures of breast cancer cells and CAFs, blocking COL3A1 in the fibroblasts slowed tumor growth under chemotherapy and lowered levels of the fat-making enzyme and the cancer-cell receptor linked to oleic acid. In a group of 72 patients treated with chemotherapy before surgery, higher levels of COL3A1 and its fat-metabolism partners strongly matched poorer treatment responses and shorter time before the cancer returned.
What this means for future treatment
Put simply, the study shows that a particular set of support cells in breast tumors acts like a bodyguard unit: they feed cancer cells a protective fat-based diet and call in immune allies that blunt the body’s defenses. Together, these actions make chemotherapy less effective. Measuring the abundance of COL3A1-high fibroblasts improves doctors’ ability to predict who will or will not respond to treatment, and the pathways they use—fat production, survival signaling inside cancer cells, and immune dampening—offer new targets for drugs. In the future, therapies that disarm or reprogram these fibroblasts could be paired with standard chemotherapy to prevent tumors from developing resistance.
Citation: Jiang, P., Li, X., Wang, Z. et al. COL3A1high cancer-associated fibroblasts orchestrate metabolic and immune microenvironments to confer chemoresistance in breast cancer. npj Precis. Onc. 10, 139 (2026). https://doi.org/10.1038/s41698-026-01338-9
Keywords: breast cancer, chemoresistance, tumor microenvironment, cancer-associated fibroblasts, lipid metabolism