Clear Sky Science · en
Integrating liquid biopsy and mutational signatures to advance precision oncology
Why a Blood Test for Cancer’s “Fingerprint” Matters
Cancer is often described as a disease of DNA, but every person’s tumor is genetically different and can change over time. This makes it hard for doctors to choose the right treatment and to know when a cancer is coming back or becoming resistant to drugs. This article explains a promising approach that combines a simple blood test, called a liquid biopsy, with detailed patterns of DNA damage known as mutational signatures. Together, they could allow doctors to track how a tumor behaves throughout a patient’s illness and to tailor therapies more precisely than ever before. 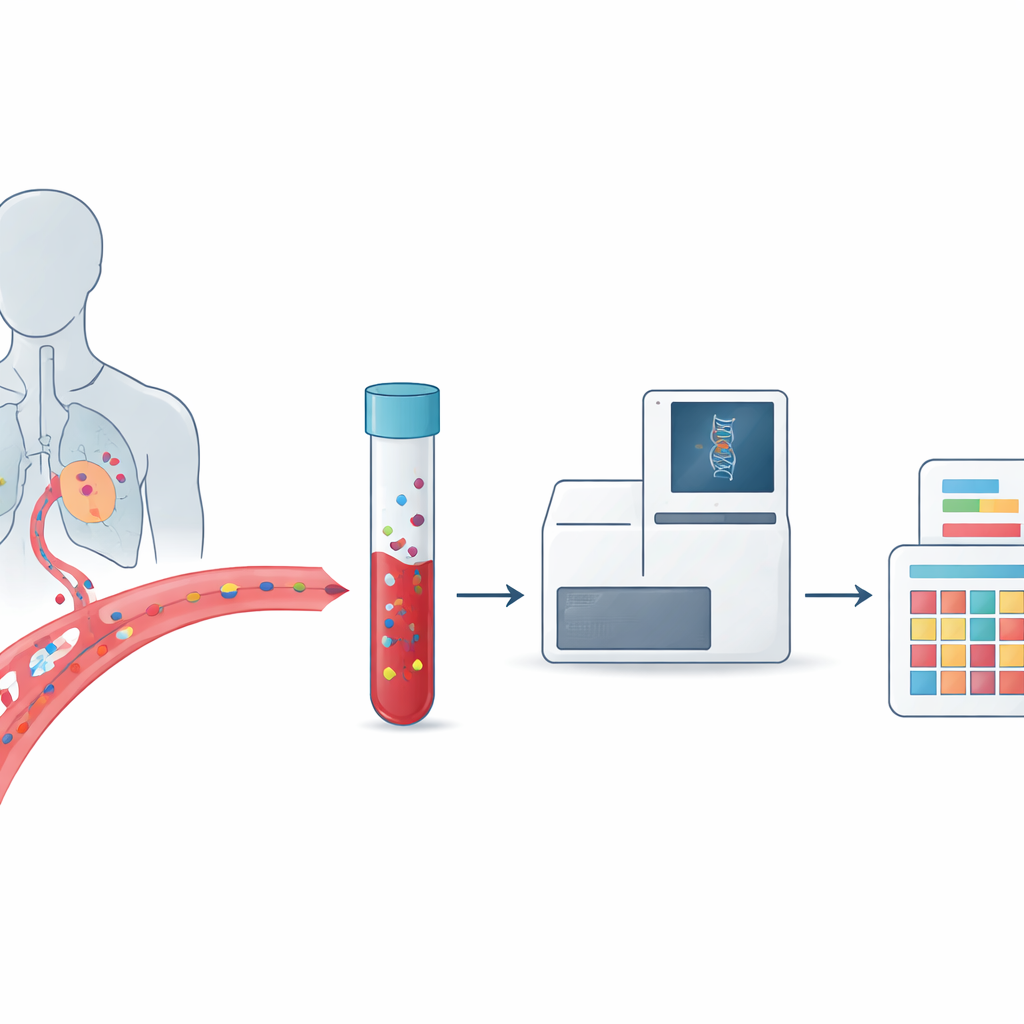
Looking for Cancer Clues in a Tube of Blood
Traditional cancer biopsies require removing a piece of tumor tissue through surgery or a needle, which can be painful, risky, and difficult to repeat. Liquid biopsy takes a different route: it looks for traces of cancer that tumors shed into the bloodstream and other body fluids. These traces include whole tumor cells and short fragments of DNA released when cancer cells die. Because blood samples can be drawn regularly and capture material from multiple tumor sites, liquid biopsy offers a moving picture of how a cancer grows, spreads, and responds to treatment, rather than a single snapshot in time.
From Individual Mutations to Overall Damage Patterns
Most current liquid biopsy tests focus on spotting specific mutations in a short list of genes that guide the use of targeted drugs. However, not all tumors carry such “druggable” changes, and cancers with many different mutations can easily escape fixed gene panels. Over the past decade, powerful DNA sequencing tools have revealed that the mutations in a cancer genome are not random: they form recognizable patterns shaped by factors such as tobacco smoke, ultraviolet light, faulty DNA repair, or chemotherapy itself. These recurring patterns, called mutational signatures, act like fingerprints of the processes that have damaged the DNA over the lifetime of the tumor.
Reading the Tumor’s History and Weaknesses
Mutational signatures can reveal where a cancer may have started and which internal repair systems are broken. For example, lung cancers from smokers often show a pattern linked to tobacco exposure, while some colon cancers carry signatures of mismatch repair failure, a defect that also makes them highly sensitive to certain immunotherapies. Other signatures point to problems in a DNA repair pathway called homologous recombination, which can make tumors especially vulnerable to platinum drugs and PARP inhibitors. Experimental studies in cells and animal models have confirmed that many environmental chemicals and cancer treatments leave distinct mutational footprints that match those seen in patients, strengthening the link between signature and cause. 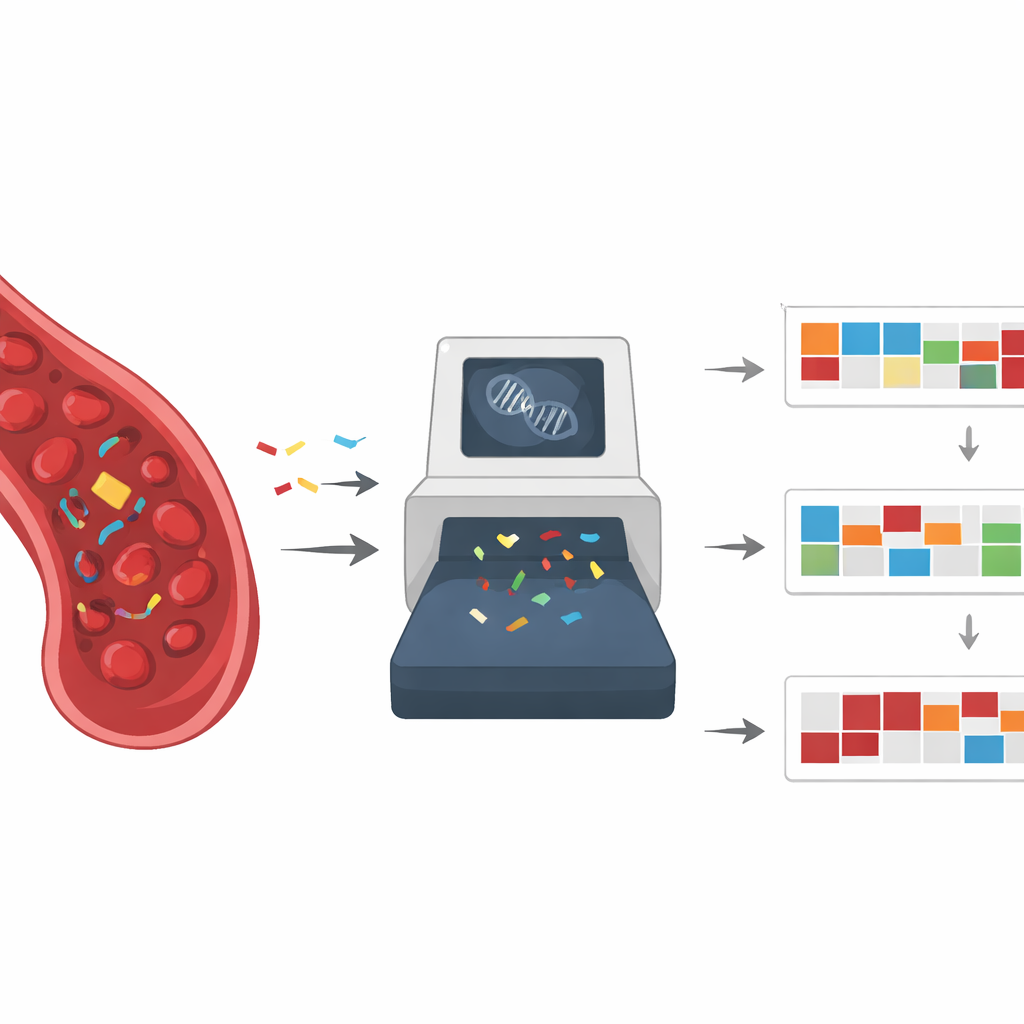
Combining Blood-Based Monitoring with DNA Fingerprints
The key idea of the article is to merge liquid biopsy with mutational signatures into a single framework. By using broad, genome-wide sequencing on tumor DNA fragments in the blood, doctors could simultaneously identify individual mutations and the overall damage patterns that produced them. This could improve early cancer detection, help pinpoint the tissue of origin in puzzling cases, and refine treatment choices even when classic driver mutations are missing. Repeated blood draws over months or years could show when a tumor switches to a new mutational process, such as one triggered by ongoing drug exposure, signaling emerging resistance or the risk of a second therapy-related cancer.
Hurdles on the Road to Everyday Use
Despite its promise, this approach faces practical challenges. Some tumors release very little DNA into the bloodstream, making their signals hard to detect. Many known signatures still lack a clear biological explanation, and some patterns may be partly shaped by technical quirks of different sequencing platforms. Age-related changes in blood-forming cells can introduce their own mutations into blood DNA, confusing the picture further. Current methods to pull out signatures from plasma often rely on complex machine-learning tools that require large training datasets and have mostly been tested in retrospective research studies rather than prospective clinical trials.
What This Could Mean for Future Cancer Care
The authors conclude that integrating liquid biopsy with mutational signatures could eventually transform cancer care from a series of one-time decisions into a continuously updated process. Instead of relying solely on a tissue biopsy taken at diagnosis, clinicians might one day use routine blood tests to follow the evolving “handwriting” of DNA damage in a patient’s tumor. This could sharpen screening, improve diagnosis, guide therapy selection and sequencing, and detect resistance before it becomes visible on scans. Realizing this vision will require standardized methods, rigorous validation, and smarter computer models, but the concept points toward a future where cancer treatment is not just targeted at the tumor’s current mutations, but also informed by the forces that created them.
Citation: Carrasco, R., Dreij, K. Integrating liquid biopsy and mutational signatures to advance precision oncology. npj Precis. Onc. 10, 114 (2026). https://doi.org/10.1038/s41698-026-01337-w
Keywords: liquid biopsy, mutational signatures, cell-free DNA, precision oncology, cancer evolution