Clear Sky Science · en
Predicting homologous recombination deficiency and treatment responses using a computed tomography-based foundation model: a preclinical study
Why this matters for patients and doctors
Cancer treatments that damage DNA, such as certain chemotherapies and new targeted drugs, can work remarkably well in tumors whose repair systems are already faulty. The challenge is figuring out which tumors have these hidden weaknesses without relying on repeated, invasive biopsies. This study explores whether routine medical-style scans in mice, interpreted by a powerful artificial intelligence model, can reveal how vulnerable a tumor is and how well it will respond to a new experimental drug.
A hidden weak spot in cancer cells
Many tumors carry defects in a key DNA repair pathway called homologous recombination. When this pathway fails, cells accumulate damage in their genetic material, a condition known as homologous recombination deficiency, or HRD. HRD can make cancers more likely to form, but it also turns into an Achilles’ heel: drugs that further damage DNA can push these already stressed cells over the edge, killing them more effectively than healthy cells. Today, identifying HRD usually relies on genetic tests or specialized laboratory assays that are costly, time-consuming, and often require taking pieces of the tumor. The authors asked whether imaging—the same kind of three-dimensional X‑ray scans widely used in clinics—could offer a fast, non-invasive window into HRD.
Using smart scans instead of more biopsies
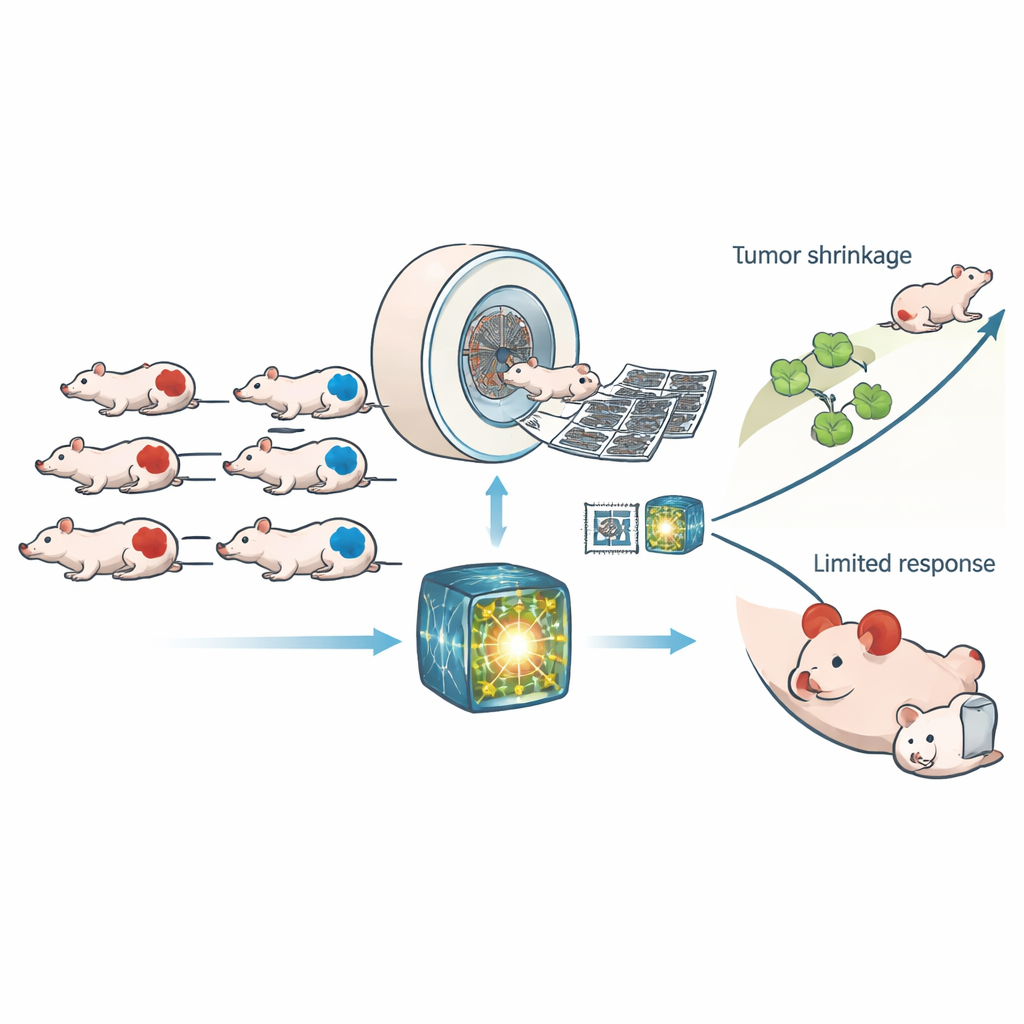
The researchers worked with 307 mice carrying human tumor grafts that were either HRD or normal in their repair ability. All animals underwent high-resolution computed tomography (CT) scans before and after receiving either a control treatment or CP‑506, an experimental drug that becomes active in low-oxygen regions of tumors and crosslinks DNA strands. Rather than relying on a handful of handcrafted image measurements, the team used a modern “foundation model” trained on large human CT datasets to pull out thousands of subtle patterns from each scan. A simple classifier was then trained on these patterns to decide whether a tumor was HRD, and the same learned features were reused to predict how much DNA damage and growth delay CP‑506 would cause.
How well the imaging model did the job
The foundation model clearly outperformed both traditional radiomics and a standard deep learning network that was trained from scratch. On new test scans, it distinguished HRD from non-HRD tumors with an area under the curve of about 0.88, a level of accuracy that held up across different CT energy settings. When both AI approaches agreed on the tumor type, performance rose even higher. The model’s image-based HRD score lined up closely with the true genetic status in several prostate and colorectal tumor lines, though one line with unusual repair behavior remained harder to classify. Importantly, the CT features linked to HRD were not just statistical artifacts: they also predicted how much lasting DNA damage CP‑506 produced 48 hours after treatment and how long it took tumors to regrow to four times their size in a parallel experiment.
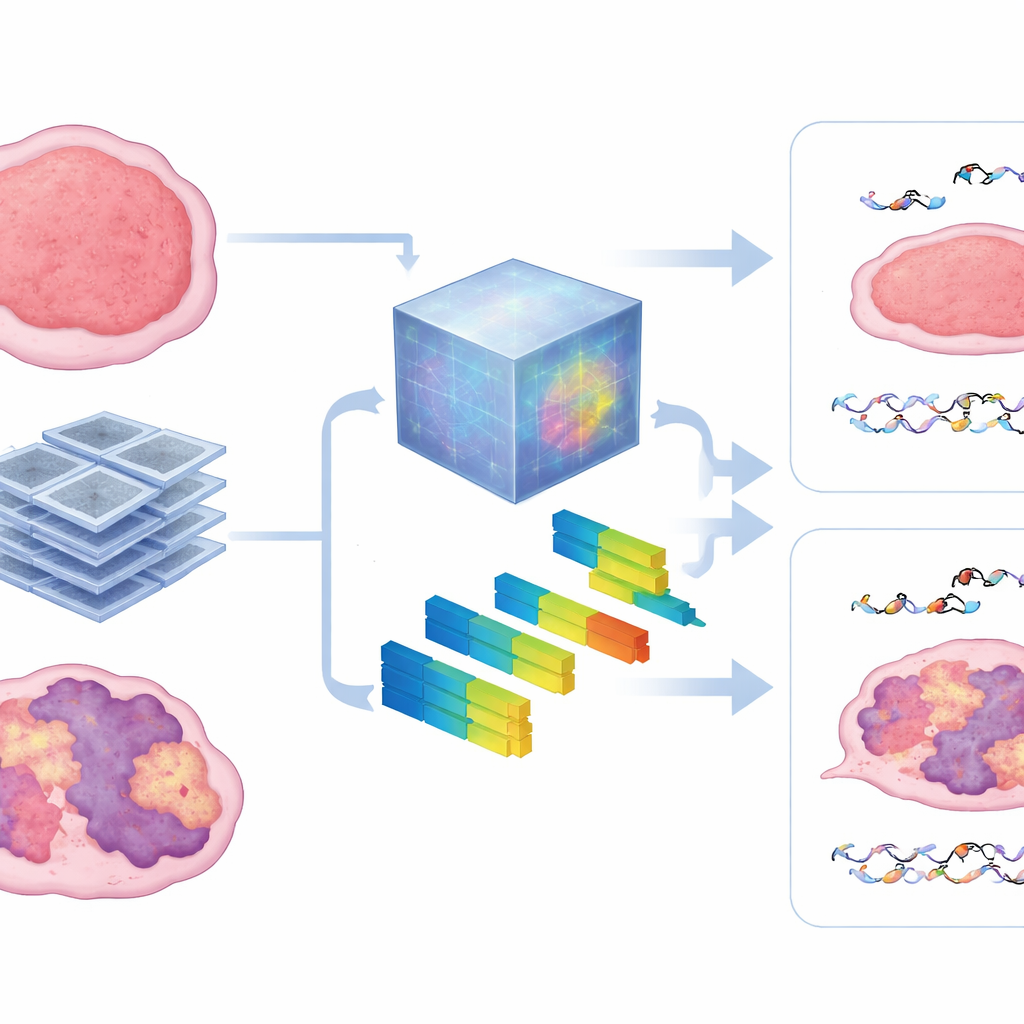
What the scans reveal inside the tumor
To peek inside the black box, the authors compared the patterns learned by the foundation model with standard radiomic features that describe image texture and intensity. They found that the model heavily relied on measures of heterogeneity—how patchy, coarse, or irregular the tumor looked on CT. Tumors with stronger HRD signatures tended to show more disorganized textures, and these same features helped forecast both microscopic DNA damage and macroscopic tumor control. The approach remained robust when less training data were available, hinting that such pretrained models can overcome a major obstacle in animal studies, where collecting large imaging datasets is difficult.
From mouse experiments to future patient care
This preclinical work shows that a single CT scan, interpreted through a powerful, pretrained AI, can capture signals of faulty DNA repair and anticipate how strongly a tumor will respond to a DNA‑damaging drug. For a layperson, the takeaway is that standard-looking scans may soon do double duty—locating a tumor and revealing how fragile it is—without extra procedures. Before this becomes part of routine care, the method must be tested and validated in human patients. If those efforts succeed, CT-based AI fingerprints of HRD could help doctors match people more precisely to treatments like CP‑506 and related therapies, improving outcomes while sparing others from ineffective, toxic drugs.
Citation: Kuang, S., Schuitmaker, L., Wu, M. et al. Predicting homologous recombination deficiency and treatment responses using a computed tomography-based foundation model: a preclinical study. npj Precis. Onc. 10, 121 (2026). https://doi.org/10.1038/s41698-026-01322-3
Keywords: homologous recombination deficiency, cancer imaging, radiomics, foundation models, DNA-damaging therapy